The possibility to separate molecules by size under native conditions is powerful in protein analysis. With over thirty years in protein science, Åke Danielsson, Research Director at Cytiva, recently held a webinar on protein characterization using size exclusion chromatography (SEC). Here, I share three important takeaways from this webinar.
Importance of chromatography system configuration
One thing to remember is that the configuration of your chromatography system has a huge impact on resolution in SEC analysis. You can easily diminish resolution of a column by having large internal volumes in your system prior to the detector. Therefore, it is of high importance to keep tubing as short as possible and using the smallest inner diameter allowed without getting issues with high backpressures. As shown in Figure 1, the larger the tubing diameter, the poorer the resolution.
In general, the smaller the bed volume of your column, the larger the impact of internal system volumes is on resolution. Hence, you should ensure that all components of your system have as small internal volumes as possible, not just the tubing.
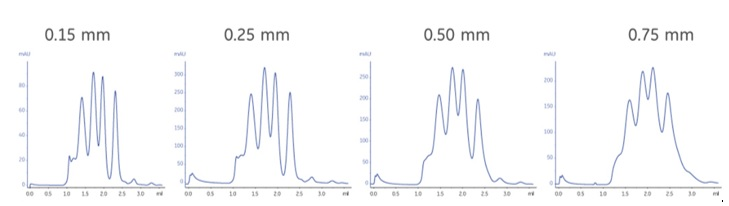
Fig 1. Example chromatograms from using different tubing diameters (0.15, 0.25, 0.50, and 0.75 mm) on the same system setup including a Superdex 200 5/150 GL column.
Impact of sample loop filling on sample loss and reproducibility
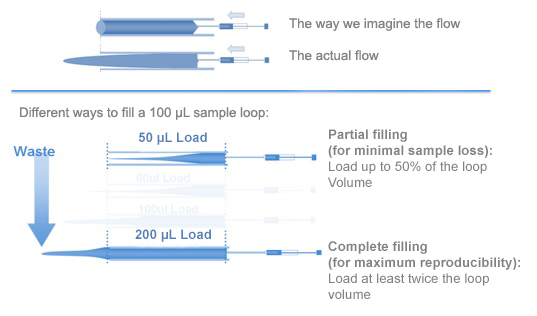
To minimize sample loss, you should fill the sample loop only partially, with maximum 50% of the loop volume. For maximal reproducibility, however, you should fill the sample loop with at least twice the loop volume. Figure 2 illustrates the fluid dynamics involved.
Why agarose-based SEC resins are important complements to silica-based resins
There are mainly two types of SEC resins available: silica-based resins and agarose-based resins. Properties of silica-based and agarose-based resins are listed in Table 1. The main difference, which has a practical impact, is that agarose-based resins have much higher pH stability and can thus be cleaned under harsh conditions. Standard protocols for cleaning of agarose-based resins include NaOH. Silica-based resins, on the other hand, decompose at high pH, preventing the use of NaOH in cleaning of these resins. In addition, while agarose has little, if any, tendency to adsorb proteins, silica-based resins might contain residual silanols that can adsorb proteins.
Traditionally, agarose-based resins are more commonly used in preparative SEC, while silica-based resins are more used in analytical SEC applications. Today, however, there is a new generation of agarose-based resins available, with smaller, more rigid beads. These resins offer a good complement to the silica-based resins for protein analysis. Superdex 200 Increase, Superose 6 Increase, and Superdex 75 Increase are all agarose-based resins suitable for analytical applications. All three resins are available in three different column formats: 10 × 300 mm, 5 × 150 mm, and 3.2 × 300 mm.
If you are using a silica-based resin for your SEC analysis today, consider trying an agarose-based SEC resin next time.
Table 1. Properties of silica-based and agarose-based SEC resins
| Property | Silica beads | Agarose beads |
|---|---|---|
| Chemical composition | SiO2 | Polysaccharide (D-galactose-3,6-anhydro-L-galactose)n |
| Surface coating | Essential to block silanols to minimize protein adsorption | Not required |
| pH stability | Typically pH 2—8 | Typically pH 3—11 (short term pH 1—12) |
| Mechanical stability | Rigid, high-pressure stability | Rigidity achieved by chemical cross-linking |
| Porosity | Can be controlled to achieve the desired separation range | Can be controlled to achieve the desired separation range |
Did you miss Åke’s webinar followed by a live Q&A session? Listen to the on-demand version of the webinar Protein characterization using size exclusion chromatography (SEC).