Virus-like particles (VLPs) are a promising molecular format for the development of vaccines targeting emerging viruses and cancers. Their size and shape enable an easy uptake by antigen-presenting cells (APCs) resulting in an effective induction of an adaptive immune response.
VLPs are formed by the recombinant expression of viral structural proteins. A method for the large-scale production of VLPs is production in suspension cell culture. Extracting the VLPs from the cell culture supernatant is a time and labor-intensive process. The development of a purification strategy is a mainly experimental approach where parameters are optimized via Design-of-Experiment (DoE) studies. Dozens of experiments are needed to screen a process design space that was manually chosen.
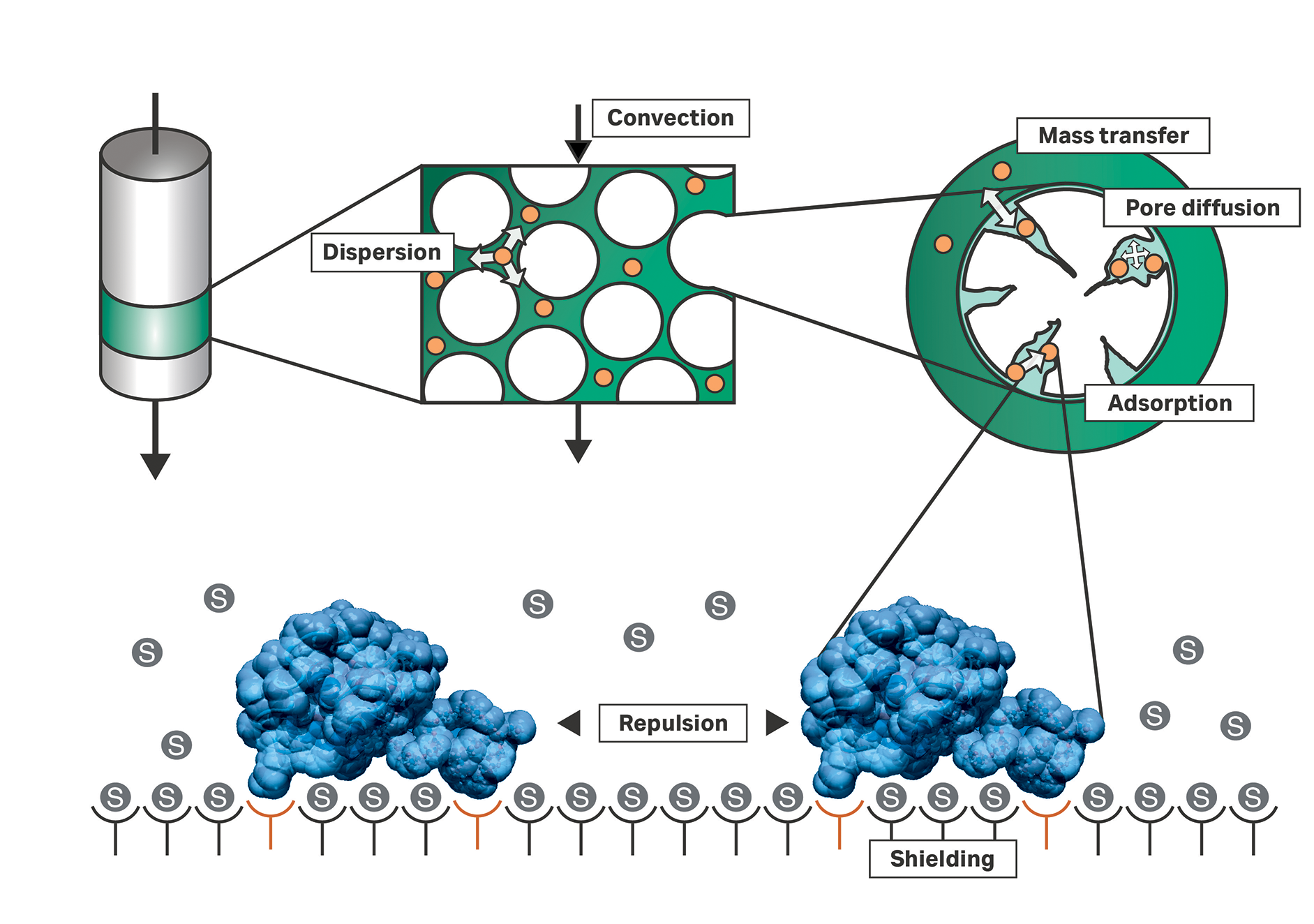
In contrast, mechanistic models describe the transport of sample components using fluid dynamic and thermodynamic principles (Fig 1). Further, they allow for efficient process optimization in silico. In this article, GoSilico™, now part of Cytiva, and CEVEC Pharmaceuticals present the results of an innovative approach for the development of a purification process for VLPs.
Fig 1. Fluid dynamic and thermodynamic principles of chromatography processes used by mechanistic models.
Materials and methods
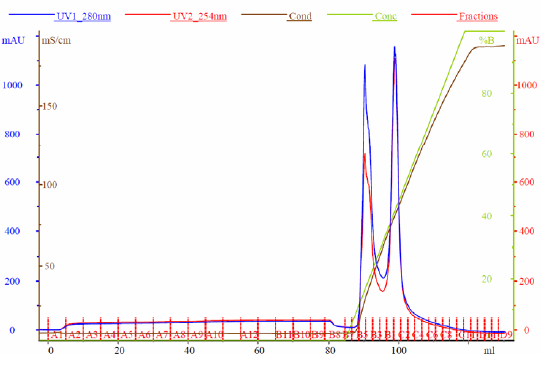
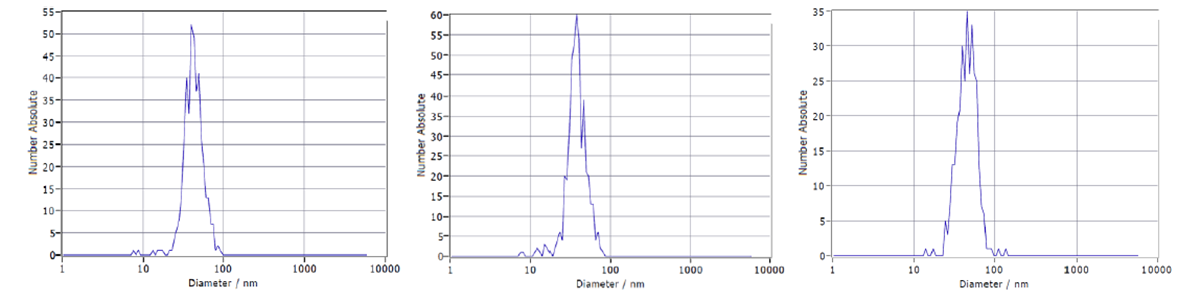
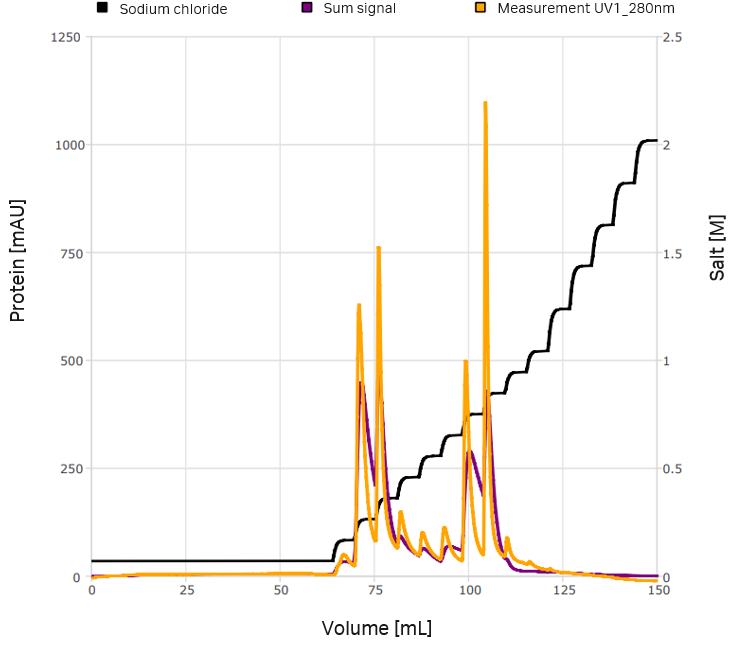
VLPs are assembled by recombinant expression of viral structural proteins. A method for the large-scale production of VLPs is the expression in suspension cell culture. CEVEC's CAP®Go platform was employed to produce a flavivirus VLP of ~ 40 nm size. As a first purification step, tangential flow filtration was used. The selected mode of separation was anion exchange chromatography. Chromatography experiments were conducted with an ÄKTA™ system. Chromatograms and additional fraction analysis were imported to GoSilico™ Chromatography Modeling Software for modeling and optimization. The initial reference chromatogram can be seen in Figure 2. A nanoparticle tracking analysis revealed two different VLP variants (Fig 3).
Fig 2. Initial reference chromatogram exported from UNICORN™ software.
Fig 3. Nanoparticle tracking analysis of feed (left), first elution peak (center), and second elution peak (right) showing two VLP variants.
Model calibration
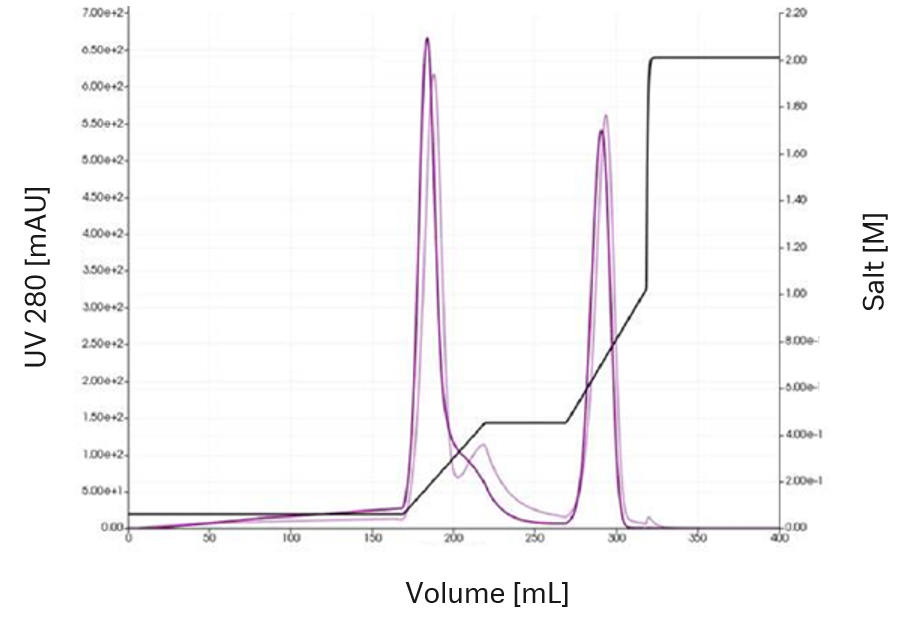
Initially only two chromatograms, one gradient and one stepwise elution, were recorded (Figs 4 and 5). Using only spectral analysis, 6 different feed components were identified. A radial-flow column model was chosen together with the steric mass action (SMA) model for ion-exchange chromatography. Model calibration was accomplished using the built-in peak detection and parameter estimation methods of GoSilico™ Chromatography Modeling Software. After uncertainty quantification (see Model Quality below), a first in silico optimization was done using a combination of a step and a gradient.
Fig 4. The first experiment using a gradient elution shows two VLP peaks.
Fig 5. A stepwise elution was performed to identify a suitable level for peak separation.
Model-based process optimization
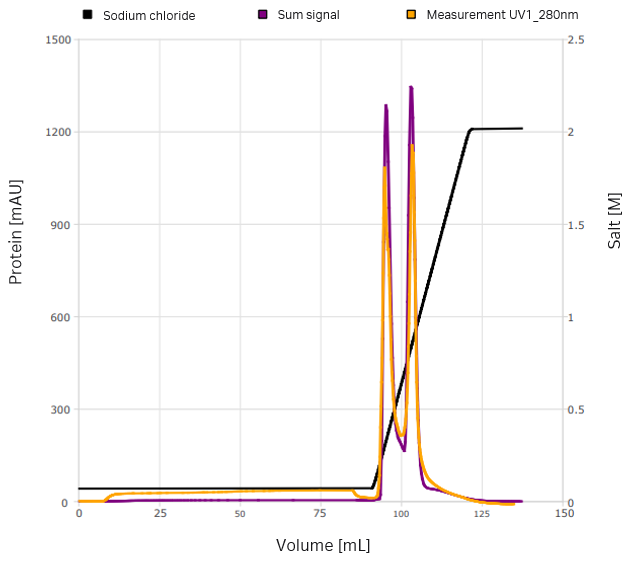
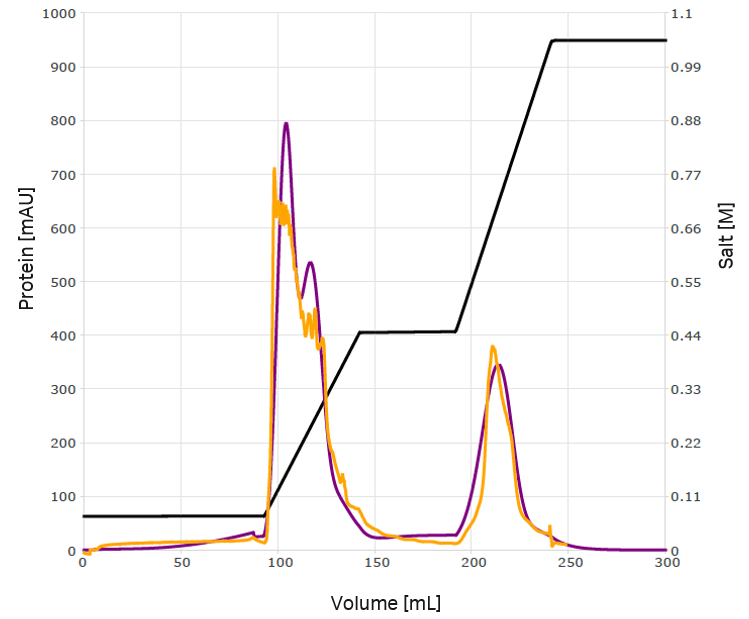
Robust elution conditions were developed based on the preliminary model. To improve model quality while aiming for baseline separation, the first step was replaced by a gradient (Fig 6). Most of the host cell proteins could be removed and >95% of the VLPs could be recovered. Furthermore, the elution conditions achieved the desired base-line separation. Alternatively, a flow-through of the first peak was considered. A validation run was performed with this setup and was in agreement with the model predictions (Fig 7).
Fig 6. A robust optimization was performed that shifted component four in the first peak. The conductivity trace is indicated in black, while purple and lilac show two possible simulated outcomes of the optimization.
Fig 7. The validation run was in good agreement with the model-based prediction. The conductivity trace is indicated in black. The simulation is depicted in purple, the validation measurement is shown in orange.
Model quality
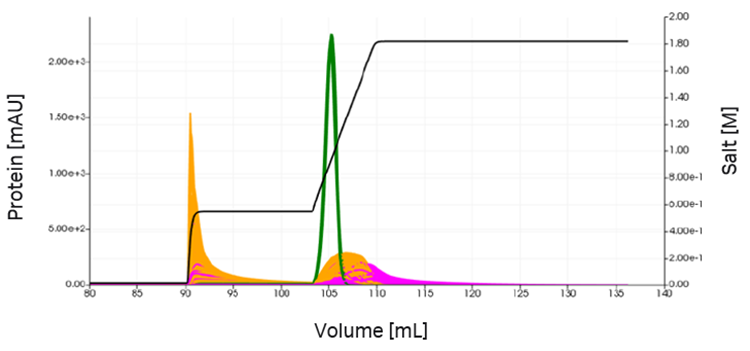
While the visual model fits were very good, only the calculated 95% confidence intervals provide information on if the parameters were identified. The uncertainty of the estimation of components 4 (orange) and 6 (pink), which elute close to the product (green), leads to a possible co-elution. The resulting chromatograms and yield distribution for a 1000-point Latin Hypercube Sampling (LHS) is shown in Figure 8.
Fig 8. Uncertainty in the model parameters of component 4 (orange) and 6 (pink) leads to a variety of chromatograms at optimized process conditions (first VLP peak not shown). The product is depicted in green.
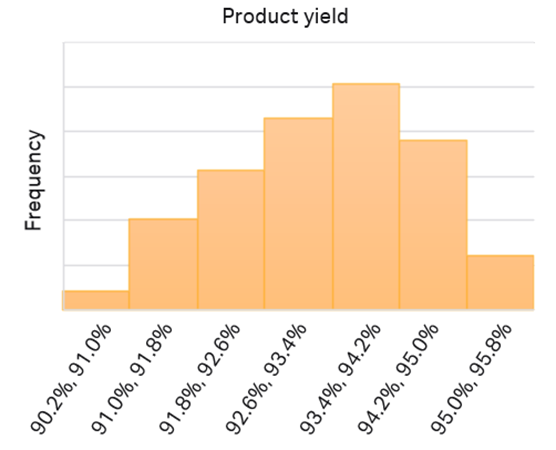
Fig 9. The predictivity of the initial model was evaluated in a 1000-point LHS sampling. The average yield is predicted to be 93.3% with 95% confidence interval of ± 0.1%.
Conclusion
The combination of a limited set of experiments and a mechanistic model enabled efficient process optimization and design space characterization for VLP purification. This novel approach could be used whenever a downstream platform process currently doesn’t exist. The finalized model created a greater understanding of product quality attributes and their relation to process parameters. This is a requirement of the Quality-by-Design guideline set forth by the European Medicines Agency (EMA) and Food and Drug Administration (FDA). Mechanistic model’s ability to predict the outcome of a scaled-up process is very valuable in early process development for novel pharmaceuticals, since there is typically only a limited amount of sample available to work with
This case study was conducted in close collaboration with CEVEC Pharmaceuticals GmbH. Experimental data presented in this article is used with courtesy of CEVEC Pharmaceuticals.