The Figurate automation platform implements the control and management of flexible processes, variable batch sizes and production locations on the Cytiva FlexFactory platform. Agile, platform-based automation permits users to rapidly adjust capacity across facilities, sites and even geographies. Automated single-use facilities enable faster changeovers while maintaining safety and regulatory compliance, with additional staff utilization benefits. A Cytiva FlexFactory single-use processing platform with Figurate automation is the optimal solution for biomanufacturers who wish to initiate or expand manufacturing capacity quickly and cost-effectively. Wonderware automation, part of the Figurate portfolio, offers an economically attractive option for start-up biomanufacturers and those who do not have an alternative automation platform in place.
A highly integrated automation approach is a prerequisite for production efficiency and operational visibility across the biomanufacturing process. With centralized control and collection of information and data, process intensification and productivity can be maximized. Compliance and consistency are critical in a heavily regulated manufacturing environment. The goal of automation within a fully integrated single use/hybrid bioprocessing facility is to create applications that enable the delivery of products (such as mAbs or viral vectors) at the highest possible quality and lowest cost.
The Cytiva FlexFactory platform addresses the increasing demand for an integrated and flexible manufacturing platform solution for biotherapeutics. The FlexFactory offering combines equipment, integrated automation, connectivity solutions and supporting services in a turnkey platform, designed and delivered within a controlled project schedule. The delivered FlexFactory is production-ready, set up and qualified to meet the facilities manufacturing needs.
Automation enables and optimizes flexible biomanufacturing
When combined with other enabling technologies such as single-use processing equipment, centralized automation delivers powerful benefits to biologics manufacturers.
- Streamlines the time and effort required to implement a new process, enabling flexible capacity with high utilization.
- Supports increased efficiency and reduced operating costs.
- Provides control and consistency critical in a heavily regulated environment.
- Offers process transparency, minimizing operational and financial risks.
FlexFactory automation with Wonderware is pre-configured and verified to enable efficient design of production environments for development and manufacturing of biopharmaceuticals. The automation platform is the foundation of the flexible, configurable, and scalable FlexFactory biomanufacturing solution.
Standardized approach accelerates time-to-revenue
FlexFactory implements a standardized approach to streamline operations, drive down costs, enhance productivity, reduce risk and minimize the burden of compliance, while delivering consistent high-quality product.
Leveraging a pre-configured and verified equipment, software and integration design, FlexFactory provides rapid access to cGMP biomanufacturing capacity. A FlexFactory automation platform integrates all bioprocessing unit operations.
- Standardizing unit types within a facility creates flexibility to scale up or scale out production capacity.
- Use of standard protocols and networks reduces time to implement, document, test and validate, and facilitates compliance
- Consistent data interfaces help reduce human error, training time and operator expense if all platforms look and feel the same.
- Standardized data interfaces permit flexible implementation of MES and ERP layers.
- Standardizing across facilities enables rapid deployment.
Open system design based on hardware and software standards greatly reduces design and engineering efforts, saving time and money. While customization is possible, it will require additional design, engineering, reviews and approvals, with attendant costs.
Bioprocess automation infrastructure
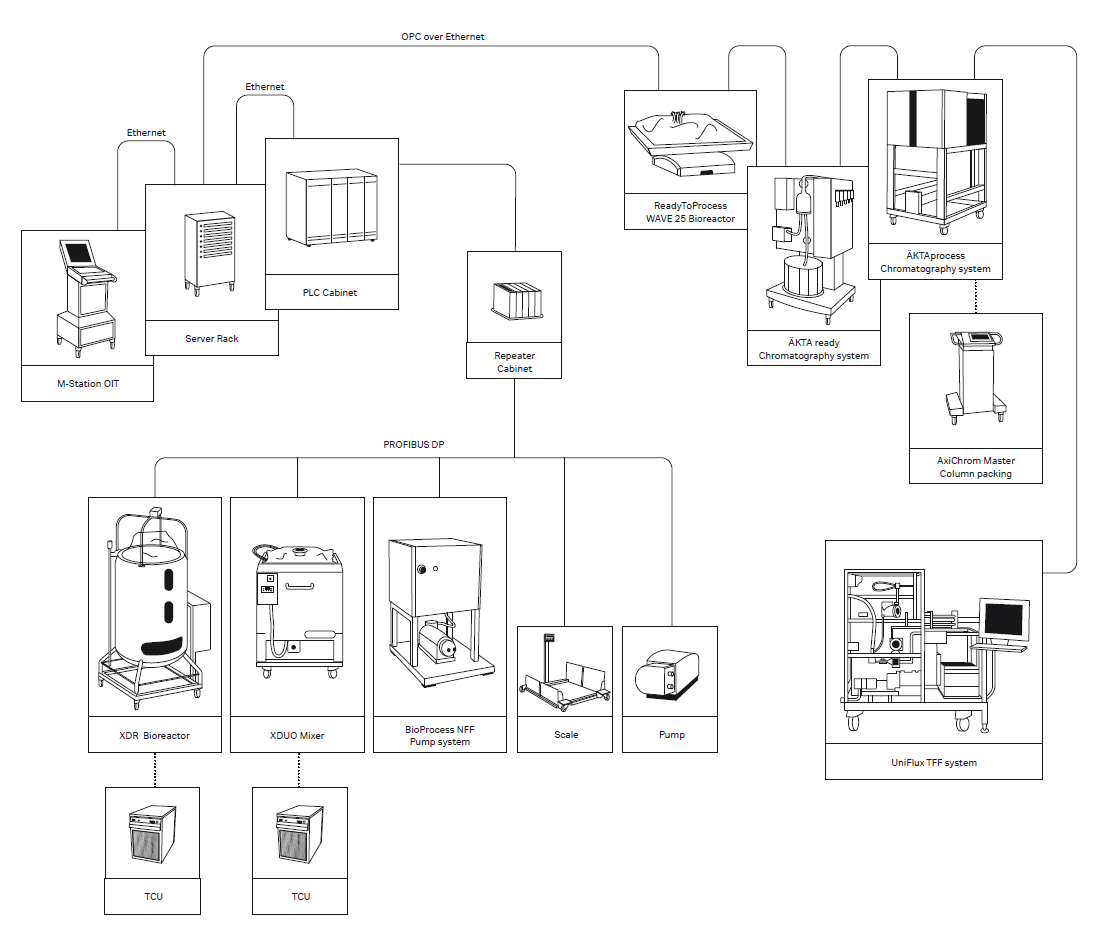
All process equipment is run by the centralized automation platform that includes control, data acquisition and storage, reporting centralized user management, common HMI and recipe control.
The automation infrastructure includes the hardware and software required to run the plant with the necessary level of redundancy to meet the high availability demands and data integrity requirements of biologics manufacturing. Wonderware automation, as implemented in the FlexFactory, is highly reliable and fault tolerant. The standard configuration offers a cost effective solution that can quickly and easily scale up, scale out and transition to new products.
The Wonderware automation platform includes the HMI, historian and data connectors necessary to provide a complete centralized automation solution. Both UNICORN software and Rockwell programmable logic controllers (PLCs) are integrated under the Wonderware HMI. UNICORN software provides unit-level control for ÄKTA and WAVE systems, and Rockwell PLCs provide the physical interface between the Wonderware software and the other unit operations equipment. Redundancy is built into the automation architecture to mitigate the risk of losing the process or batch data in case of a server failure.
Bioprocess operations using standard automation Human-machine interface (HMI)
The human-machine interface (HMI) is the central point of interaction for the overall FlexFactory. Each HMI allows an operator to monitor and control all the individual unit operations by providing a window into the process through a common user interface. Operators can access all FlexFactory automation features, including unit operations systems and historical data in the form of trends and reports. The level and type of access for individual operators is secured through role-based user accounts. (AWP)
Alarm strategy
Alarms notify the operator of abnormal situations which require timely operator action, directing their attention so that they can take corrective action and prevent an undesired consequence. The operator is the focal point of the alarm system.
The automation uses a tiered approach to classify process or equipment failures. Personnel safety is the highest tier of failure handling and alarming. Other alarms are designed to protect the equipment and process. Users are able to configure process alarms to correctly notify their operators of process excursions or failures relevant to their process.
At the highest level where personal or machine safety is a concern, interlocks are used to protect people, equipment and the facility for each FlexFactory unit operation. The design of interlocks minimizes the opportunity for unintended change to interlock trip points, logic, or status. A unified approach to interlock configuration, monitoring, and response enables consistent and effective interlock implementations within the FlexFactory. This also promotes understanding of and response to interlocks by operators and engineers working with the equipment.
All FlexFactory unit operations include emergency stop (ESTOP) pushbuttons for personnel safety. ESTOP interlocks are hard wired to shut down the equipment immediately upon activation of an ESTOP pushbutton, and the ESTOP push buttons operate independently of any logic in the automation configuration. ESTOP push buttons are monitored, and the automation will alarm and force setpoints and batch logic to safe states when an ESTOP pushbutton is activated.
Table 1. Interlock descriptions
| Failure |
Example |
Required Response |
| Personnel safety | Emergency stop (ESTOP) | ESTOP interlocks are hard wired and response to shut down equipment does not depend on the controllers. The automation will alarm on ESTOP and will force setpoints and batch logic to safe state. |
| Equipment, environmental protection | Shut off gas flow to SUB on high bag pressure | Automation software interlocks continuously monitor and protect equipment and the environment, irrespective of batch status. Interlock logic will activate on sensor failure. The automation will alarm and will force setpoints and batch logic to safe state |
| Product quality process efficiency | Control jacket temperature | The automation continuous, batch, and alarm logic is designed to ensure product quality and process efficiency. |
Recipes, batch control
The Wonderware central automation platform utilizes both Wonderware InBatch and UNICORN software to create and manage recipes and batches for the control of unit operations. All unit operations are implemented according to S-88 guidelines. The InBatch, batch execution engine and UNICORN-based functions are accessed seamlessly through the HMI. The features include:
- Create, edit, modify or delete a recipe or method;
- Schedule, initialize and execute a batch, operation or method.
As part of our wrap-around services, Cytiva can work with you to create and configure your recipe within the automation platform.
Data connectivity
The Wonderware automation platform utilizes, industry standards-based architecture to enable integration with applications such as enterprise historians, MES, LIMS and ERP systems.
The FlexFactory automation provides the necessary data flows and connectivity between units, skids and servers via the Profibus and ethernet infrastructure. The system is configured to ensure the security and reliability of the data transfer and storage.
The Wonderware historian aggregates and stores data from all units and operations, providing an auditable, tamper-proof record of all batches. The Wonderware historian server stores all batch historical data for use in reporting, including time-stamped batch events such as start, hold, restart, abort and operator interaction such as prompting and recipe parameter changes. The data is accessed by the Dream Report™ software for report generation.
Fig 1. Wonderware data connectivity schematic
Reporting
The FlexFactory Wonderware implementation utilizes Dream Report software (Ocean Data Systems, Ltd.), a powerful, flexible reporting package widely used in industrial environments. Dream Report connects directly to the historian server. Reports can be configured for different types of users, allowing operators, supervisors and managers to access the information that is relevant to their roles. Sample reports are provided to guide report configuration.
The reports generated by Dream Report support manufacturing in a regulated environment, documenting the validated audit trail for each batch and process. Integration and verification of the reporting software with the automation platform reduces the opportunity for user errors in data handling.
Regulatory compliance for FlexFactory with Wonderware automation
Verification services and GAMP™ 5
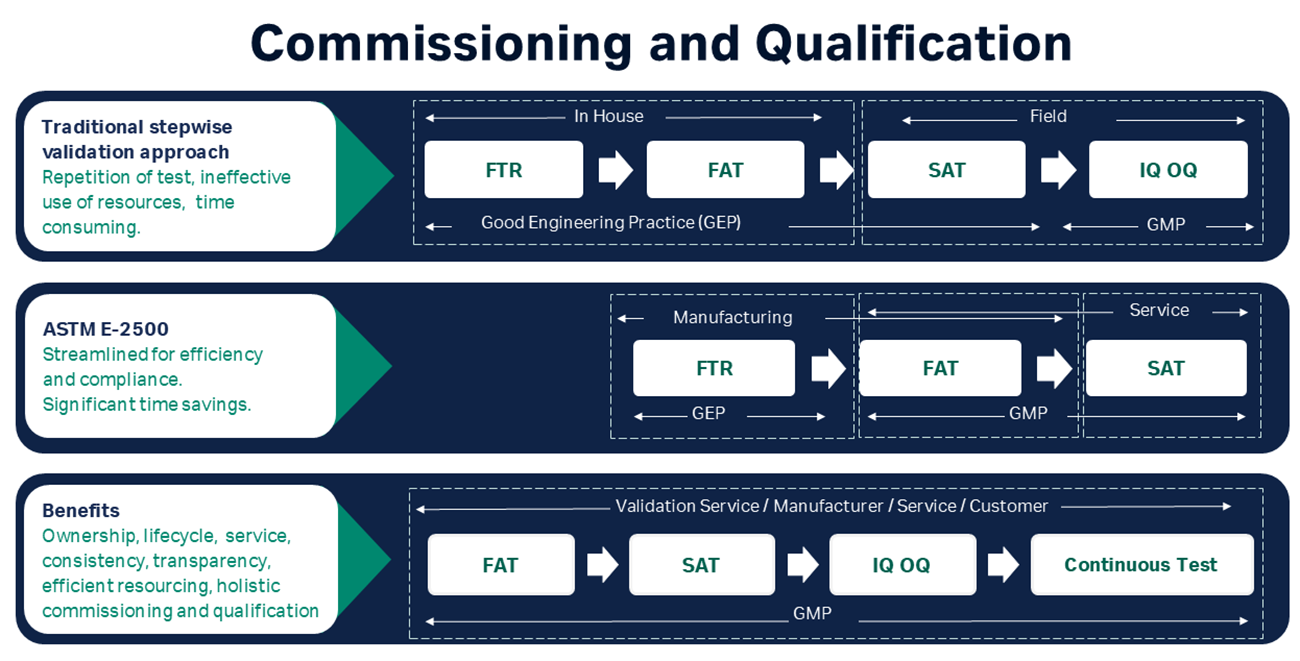
To enable compliance with regulatory requirements, FlexFactory automation is designed according to GAMP 5 guidelines. The automation implementation is assembled, tested and verified in the Cytiva factory. On arrival at the customer site, installation of the FlexFactory verifies that the equipment has not been damaged in transit and has been installed and calibrated (if required) correctly.
IOQ and Witnessed FAT & SAT services can be optionally purchased, either following a traditional regulatory approach (distinct FAT, SAT, IQ, OQ steps) or combined under a risk-based approach following ASTM-E2500 guidelines. Documentation is provided to support regulatory submissions.
Fig 2. Wonderware datafile graphics
Industrial control system (ICS) cyber security
In terms of information technology assets, a FlexFactory is a complex, highly integrated control and data management system comprising Windows®-based servers, switches, controllers and other ancillary controls equipment, all of which have been targets of cyberattacks in the industry.
Cytiva ensures that the systems, as shipped, are patched to the latest versions from our hardware and software suppliers. Once the FlexFactory is installed and commissioned, the backup and disaster recovery guidelines must be followed. Cytiva at this time does not recommend that the FlexFactory be permanently connected to business (level 3 or above) networks for any purpose without threat modelling and risk assessments, which must be performed in conjunction with the owner’s IT and OT staff.
Project management
A dedicated FlexFactory project manager (PM) from Cytiva is the single point of contact from initial configuration and specification of equipment, to delivery of the final documents following completion of activities at the manufacturing site. The project manager organizes all responsible parties within Cytiva, communicates project progress and status to the end-user, and provides information required for facility preparation and coordination of installation, commissioning and qualification, and training. Wonderware automation for the FlexFactory is delivered within the FlexFactory project management methodology and timeline, and a dedicated automation project manager is a member of the FlexFactory project team.
FlexFactory services
Complete commissioning and qualification (C&Q) documentation for the automation is included in the FlexFactory C&Q documentation package. Cytiva’s qualified engineers and support staff will execute and document integrated factory acceptance tests (FAT) (at the Cytiva site) with central automation and all major equipment.
Cytiva will install the FlexFactory platform at the customer site after transport, and will execute and document site acceptance testing (SAT) to demonstrate that the systems, including automation, meet their specifications and are fully functional and ready for formal qualification.
Installation qualification (IQ) and operational qualification (OQ) protocols are also provided and executed by Cytiva personnel at the customer’s site. Appropriate IQ/OQ protocols are generated to provide documented evidence to demonstrate that that all key aspects of the installation adhere to the approved design intentions, manufacturers’ recommendations, cGMP, GEP, user and process requirements, and that the system operates as intended throughout all its anticipated operating ranges.
Operator training is provided as part of the FlexFactory hand-over process. Operator training includes the set-up and operation of all the FlexFactory systems, including the automation software and HMI workstation usage. This practical course provides both hands-on experience and theoretical background knowledge for optimum performance of the FlexFactory platform.
The automation software is covered by the standard 1-year FlexFactory warranty, included with the FlexFactory purchase.
In addition to the included standard services, optional services are available to complement the FlexFactory offering. These include a range of FastTrak training services, lifecycle services and process development services.
Supported bioprocessing equipment
Most Cytiva bioprocess equipment can be integrated into the FlexFactory. Standard configurations are available to integrate supported equipment with the FlexFactory automation platform, while utilizing the standard sensors and controllers that are integral to the operations units. Third party equipment can be integrated on request. Cytiva project teams work closely with each customer to identify the optimal design for each FlexFactory.
Supported Cytiva bioprocessing equipment in standard design
- Xcellerex Stirred Tank Bioreactors XDR50 to XDR2000
- Xcellerex Mixers XDUO 50 L to 2500 L
- WAVE 25 Rocking Bioreactor
- Normal Flow Filtration (NFF) Skid for Harvest and Viral Filtration operations
- ÄKTAprocess and ÄKTAready chromatography skids
- UniFlux Tangential Flow Filtration (TFF) Skid.
Automation equipment
The automation platform comes with the following pre-defined automation hardware and software system components.
| Automation software |
• Wonderware 2014 software suite on Windows Server 2014 R2
|
| Server infrastructure | • Eight (8) Dell R640 servers with RAID 10 storage and redundant power supplies
• KVM and KVM Switch (e.g. Raritan™) • Uninterruptable Power (UPS) and Power Distribution (PDU) |
| Network infrastructure | • Redundant ethernet switches
• Ethernet wiring |
| Operator interface infrastructure | • HMI (human machine interface) mobile workstations/OITs (operator interface terminals) |
| Control and IO Infrastructure | • Two (2) Rockwell ControlLogix™ in full cross-over redundancy mode providing robust, secure operations to the FlexFactory and upstream unit operations
• UNICORN servers providing control and HMI for the UNICORN-controlled devices. |
| Control network infrastructure | • Profibus repeater panels
• Ethernet switches and routers |
| Additional software | • Dream Report
• Optional Win 911: Remote alarm notification |
Ordering information
Contact your Cytiva representative
Cytiva is a leader in end-to-end bioprocess manufacturing solutions, including FlexFactory, for a single or multiple production trains, and KUBio, a complete, modular biomanufacturing facility. These turnkey solutions leverage single-use bioprocess equipment and standardization to achieve rapid implementation, accelerating time-to-market and return on investment.