TempliPhi products from Cytiva, based on phi29 DNA polymerase, are widely used in genomics research. A range of TempliPhi products can accommodate low or high throughput needs, and deal with larger DNA constructs such as bacterial artificial chromosomes (BACs). As the science of genomics has advanced, however, researchers are increasingly utilizing TempliPhi amplification kits in a host of new applications. Interestingly, TempliPhi has jumped sequencing generations and gained a foothold in several next-generation sequencing (NGS) workflows, providing a suitable template for library prep and even facilitating new techniques such as Mobilome-seq, Cider-seq and MitoSV-seq.(1,2,3,4)
What underlies the usefulness of TempliPhi amplification kits in genomics applications?
phi29 DNA polymerase, well characterized by Margarita Salas and her group is a highly processive enzyme derived from the phi29 bacteriophage, capable of synthesizing long strands of DNA greater than 70 kb per binding event.(5,6,7) phi29 polymerase possesses very strong strand displacement capabilities at 30°C, which enables rapid DNA replication from multiple sites without the need for temperature cycling as is required with polymerase chain reaction (PCR).
From nanograms of starting material, phi29 DNA polymerase rapidly produces consistent microgram yields of DNA that is ready for direct use in a range of downstream analyses, including sequencing and genotyping (Fig 1).
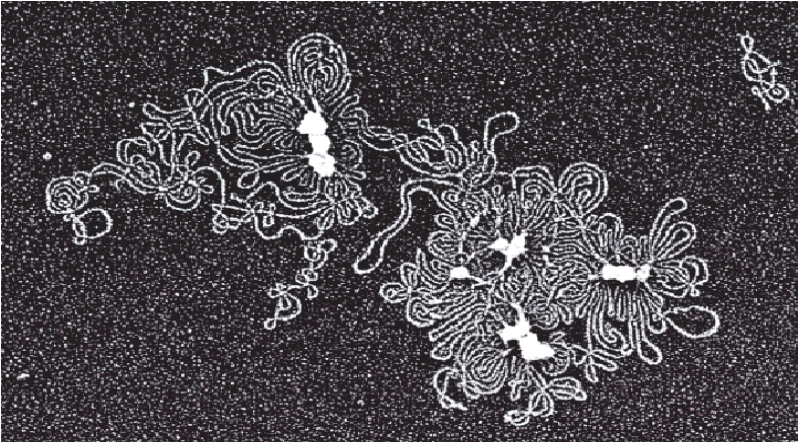
Fig 1. Electron micrograph of nondenatured TempliPhi-amplified product. A single nanogram of plasmid DNA (pUC19) was amplified with the TempliPhi DNA Sequencing Template Amplification kit. For size comparison, two copies of pUC 19 DNA appear in the top right section of the image
The one-tube, one-temperature format simplifies the DNA preparation process, without the need for specific priming sequences. TempliPhi DNA Amplification Kits are designed to prepare DNA directly from plasmid, glycerol stocks or bacterial colonies, removing the need for overnight culture and subsequent DNA isolation prior to dye terminator DNA sequencing. The elimination of these steps significantly reduces the time and complexity of template preparation, enabling higher throughput and process cost reductions compared to conventional methods (Fig 2). The streamlined workflows also facilitate automation for high-throughput sample amplification.
TEMPLIPHI BENEFITS
- Efficient: Prepares templates for cycle sequencing, cloning, and transformation from circular DNA starting material without the need for purification.
- Simple protocol: Reduces time, labor and consumables needed for template preparation, and workflow allows for easy automation.
- Quick: Protocol allows amplification of 96 samples from bacterial colonies with less than 20 minutes of hands-on time.
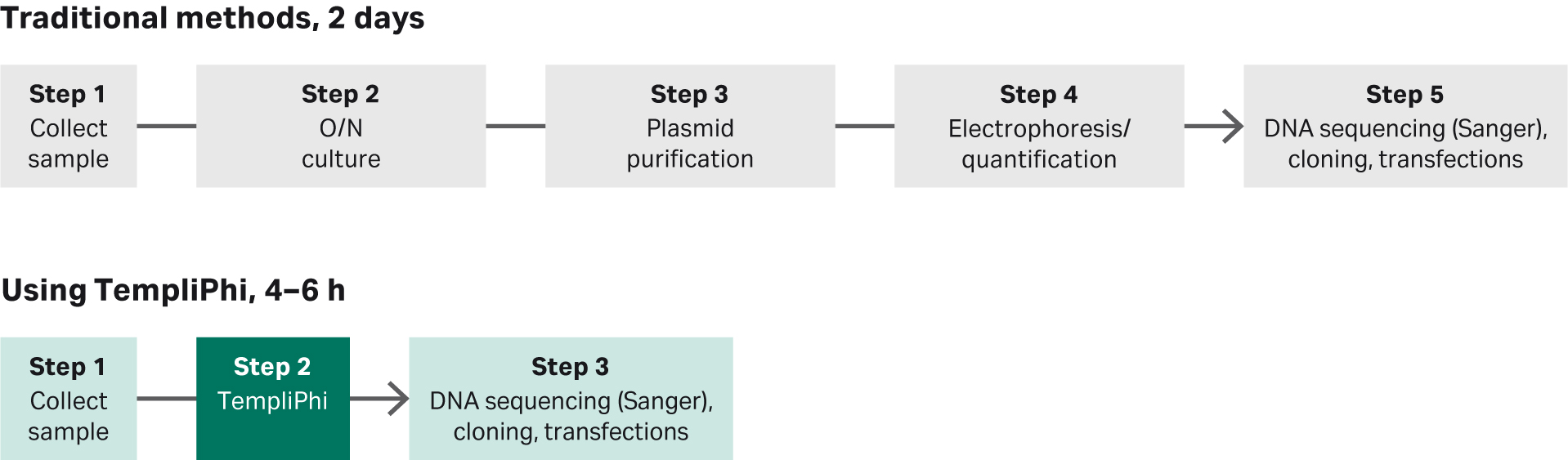
Fig 2. Schematic comparing traditional sequencing template preparation versus TempliPhi DNA amplification
The TempliPhi technology uses isothermal rolling circle amplification (RCA) for the exponential amplification of circular DNA enabled by phi29 DNA polymerase.(10) Random hexamer primers anneal to the circular template DNA at multiple sites and phi29 DNA polymerase extends from each of these primers. When the polymerase reaches a downstream primer, that strand is continually displaced by the polymerase as synthesis progresses. The displaced strand is rendered single-stranded and available to be primed by more random hexamer primer. The process continues, resulting in exponential isothermal amplification (Fig 3).
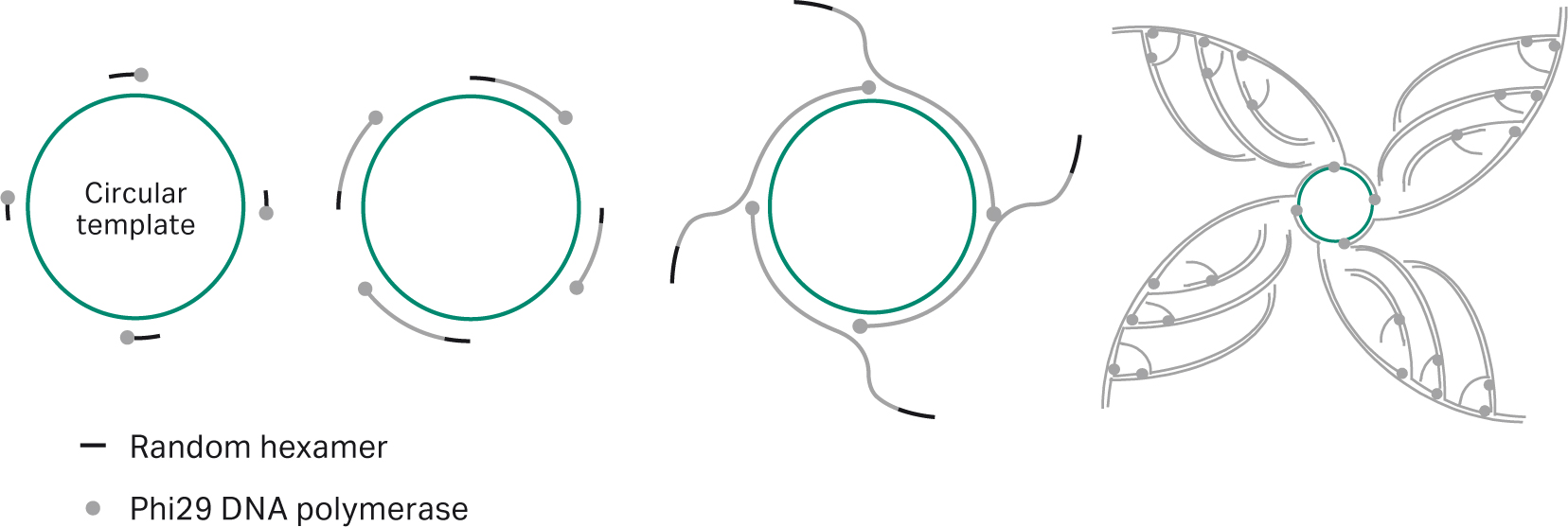
Fig 3. Rolling circle amplification (– Random hexamer; • phi29 DNA polymerase). Random hexamers bind to the circular template and are extended by phi29 DNA polymerase. Strand-displacing activity of phi29 DNA polymerase causes nascent strand to be displaced, exposing new recognition sites for the hexamers in a process known as branching.
Each TempliPhi formulation is designed to ensure that the final yield in each reaction is similar, no matter how much input material is used. This normalization effect means there is no need to quantify each reaction prior to setting up sequencing reactions. The amplified DNA can either be purified or, more commonly, a small amount used directly in sequencing and library construction without any purification, simplifying the process and reducing hands-on time without compromising on downstream success.
phi29 DNA polymerase exhibits proofreading activity which results in 100-fold higher fidelity compared to Taq DNA polymerase(8,9) and with strong displacement capabililities, can be leveraged to resolve the sequence of difficult templates, helping to overcome sequencing artefacts caused by secondary structures (Fig 4). Overall, TempliPhi amplification produces significant yields with high accuracy, quickly and efficiently.
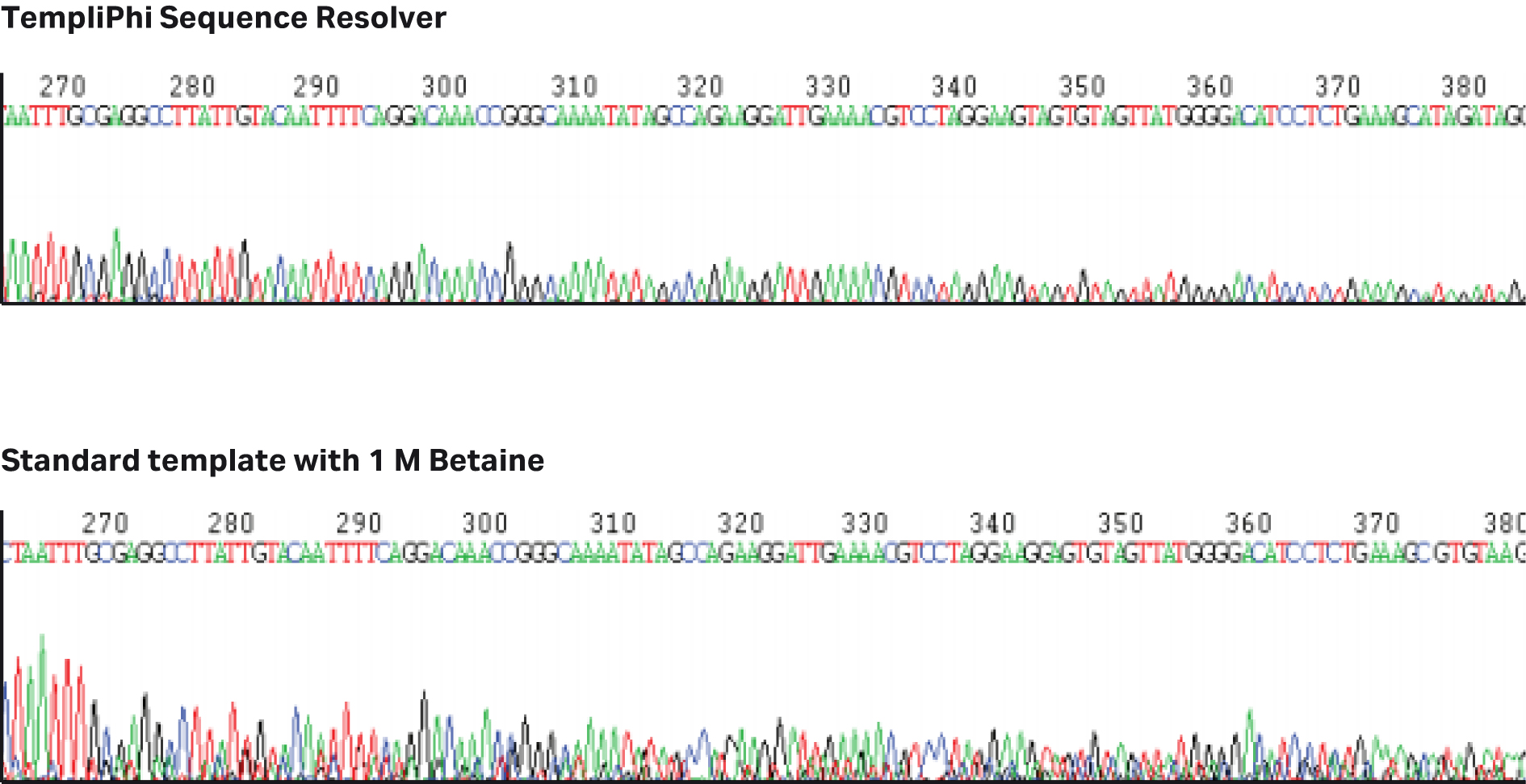
Fig 4. Template prepared using TempliPhi Sequence Resolver improves sequencing of difficult regions that other commonly used methodologies do not resolve
New applications for new times
The ability of RCA to preferentially amplify small circular DNA has opened new opportunities in recent years where TempliPhi has gained recognition outside of its original design brief and continues to be utilized in solving new problems. Researchers have found the ability of RCA to create large amounts of DNA from small amounts of input to be useful in both plant and animal studies, with TempliPhi products being extensively referenced for enrichment of viral genomes, amplification of complete mitochondrial genomes and whole community genome amplification for complex metagenomic studies. The ability to generate sufficient DNA template from artificial DNA constructs is even finding use in larger scale production of mRNA. Away from genomic analysis, TempliPhi-based methodologies have been developed to simplify cell-free protein expression, bringing rapid small-scale screening and production of proteins within reach.
Enrichment of circular viral DNA
The study of viral genomics in disease can be very difficult because viruses are typically found in the presence of host, and as such any genomic analysis can be masked or overwhelmed by host genomic DNA. Viruses that contain circular DNA are an ideal target for enrichment by RCA, yielding big gains in copy numbers. Indeed, the use of TempliPhi in enrichment of circular viral genomes has enabled investigations that would otherwise have been far more difficult. Researchers across the biology spectrum are finding interesting applications for this capability.
- In humans, novel human papillomaviruses have been discovered in oral rinse samples from patients with head and neck cancer and sequenced by NGS following enrichment amplification with TempliPhi.(11)
- Several polyomaviruses, typically hosted by birds and mammals such as raccoons, have recently been isolated from humans, with some of them clearly being associated with diseases such as cancer.(12)
- In the animal kingdom, detection and classification of tumor-causing polyomaviruses in cattle, and studies characterizing novel ssDNA viruses associated with honeybees offer further examples of RCA adoption.(13,14)
- In plants, TempliPhi amplification is used in the study of viral diversity and the impact of Geminiviridiae infections such as begomoviruses that cause significant economic damage to many important crops such as tomatoes, beans and cotton each year.(15,16,17) Here, our understanding is critical to reducing the burden on future food crop demands.
Another area of interest utilizing the power of RCA lies in identification and characterization of entire new viral and microbial populations via whole community genome sequencing, which has become commonplace with advances in both sequencing and bioinformatics technologies. These metagenomic studies shed light on the variety of organisms present in different environmental niches, with the potential to characterize the make-up and monitor changes over time. Generating meaningful data can still be challenging and requires enrichment of DNA due to very low abundance or high sample volume, as when looking for novel viruses in treated waste-water.(18) Analyses can be further compounded due to similarities in sequence that can cause errors in assembly, especially with short read sequencing technologies. Both TempliPhi and GenomiPhi, a phi29-based whole genome amplification solution from Cytiva, have been used to identify and characterize these sequences, often from unculturable sources(19), generating sufficient DNA for full metagenomic analysis. An interesting methodology that tries to resolve such issues is termed CIDER-seq (Circular DNA Enrichment Sequencing) which provides assembly-free long-read sequencing with SMRT technology from PACBIO™ (3). In this method, RCA is used to enrich DNA to generate sufficient template for sequencing.
Cell-free protein expression
In recent years, cell-free protein expression has become a genuine alternative to culture-based methodologies, with recent technological advances allowing adoption beyond the purely research environment. With decreasing costs and increasing yields, cell-free protein expression is starting to deliver on the early promise to take protein production to new areas.(20) For example, where larger numbers of proteins need to be processed in parallel, cell-based methods are cumbersome and time consuming. Some proteins can also be difficult, if not impossible, to express in cell-based systems, for example, when they are toxic to the host cell. In such cases, cell free methodologies that utilize cell-free extracts are the only way to achieve progress. These methods can significantly reduce processing times and enable expression of multiple proteins at the same time.
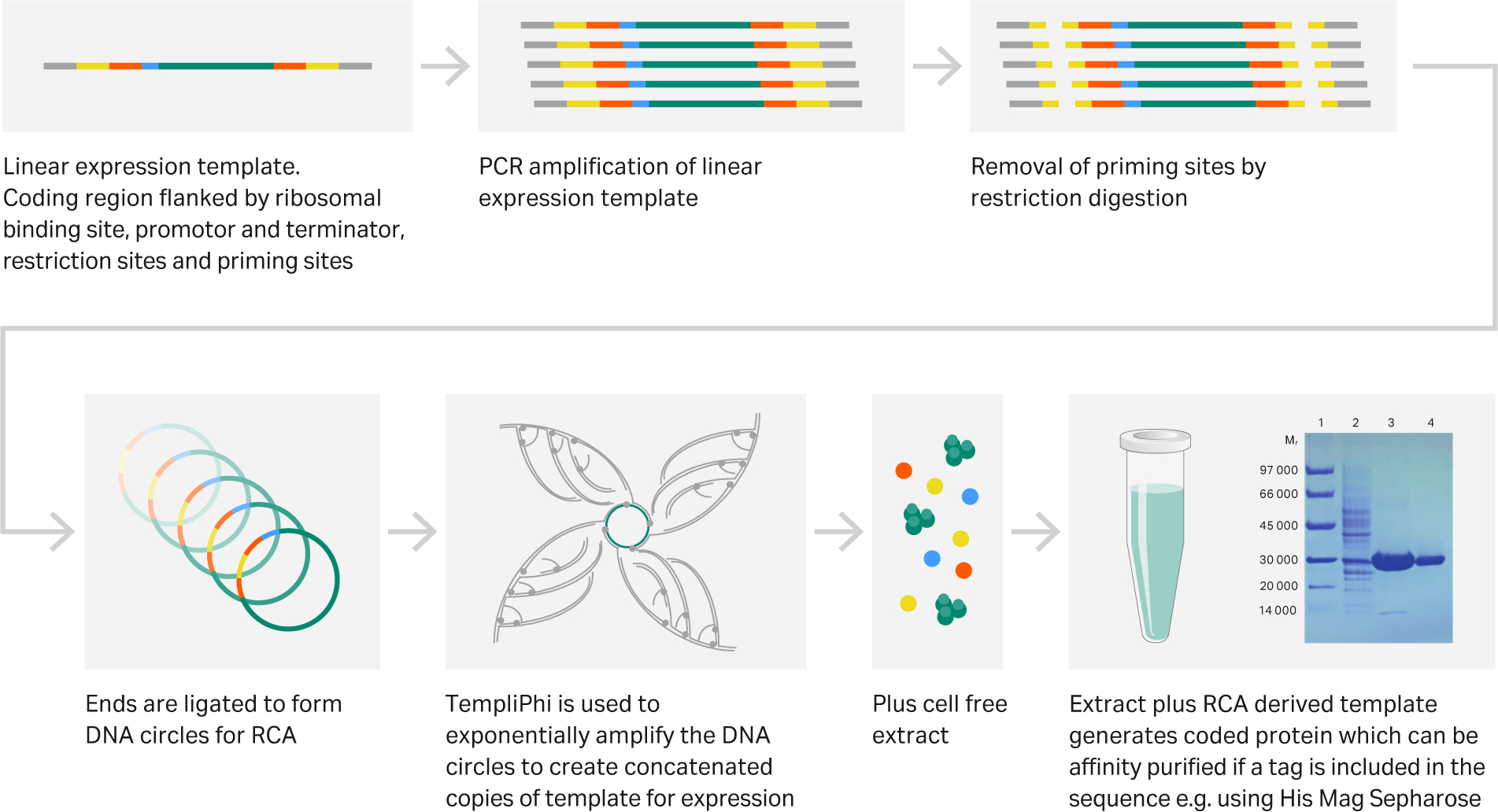
Fig 5. Workflow illustrating production of designer protein, from purchased expression template to functional protein in a single day
Rapid prototyping of proteins within 24 hours using TempliPhi rolling circle amplification has been described by Dopp, et al.(21) Here, the authors demonstrate successful production of designer proteins using a fast and effective approach whereby protein-specific linear expression templates are purchased, circularized and subjected to rolling circle amplification using TempliPhi RCA (Fig 5). Amplified product is then fed into the expression reaction as substrate for the cell-free protein production. In this way, a large number of sequences can be screened for post translational efficacy. Even proteins with disulphide bonds are now amenable to cell free protein expression using simple homemade E.coli extract.(22)
In another example, Hadi et al. demonstrated rapid generation and screening of 16 hydrolase enzymes from DNA minicircles using RCA-enabled cell free protein expression.(23) In this way, they were able to reduce processing time by more than 50% over traditional methods, with an overall time saving of 10 days.
Direct transformation or transfection
As with cell-free protein expression, increasing capabilities for manipulation, error free production, and assembly of nucleic acids are driving growth of synthetic biology. Many of these techniques rely on use of circular DNA constructs to maintain and manipulate sequences, and it is here that adoption of TempliPhi methodology has evolved to become an enabler for streamlining such workflows. The ability of TempliPhi products to efficiently amplify circular DNA is of course extremely well-suited for template preparation prior to sequence verification. It is also a valuable tool for manipulation and synthesis of the constructs themselves. Here, its high-fidelity amplification and processivity, together with the ability to directly transform or transfect the product of TempliPhi reactions into cells without further manipulation, not only saves significant time and effort but may open up new opportunities. It is this direct transformation that is surprising to some investigators, but in fact, long repeat copies of a DNA sequence produced by rolling circle amplification can be effectively taken up by different cell types such as yeast (100 fold less than supercoiled plasmid) and mammalian (similar to supercoiled plasmid) depending on promotor (Fig 6).(24) Bacteria can also process RCA DNA, albeit with significantly reduced efficiency, which can still be effective due to plentiful supply of RCA derived DNA.
Transfection efficiency with supercoiled plasmid vs RCA-derived DNA
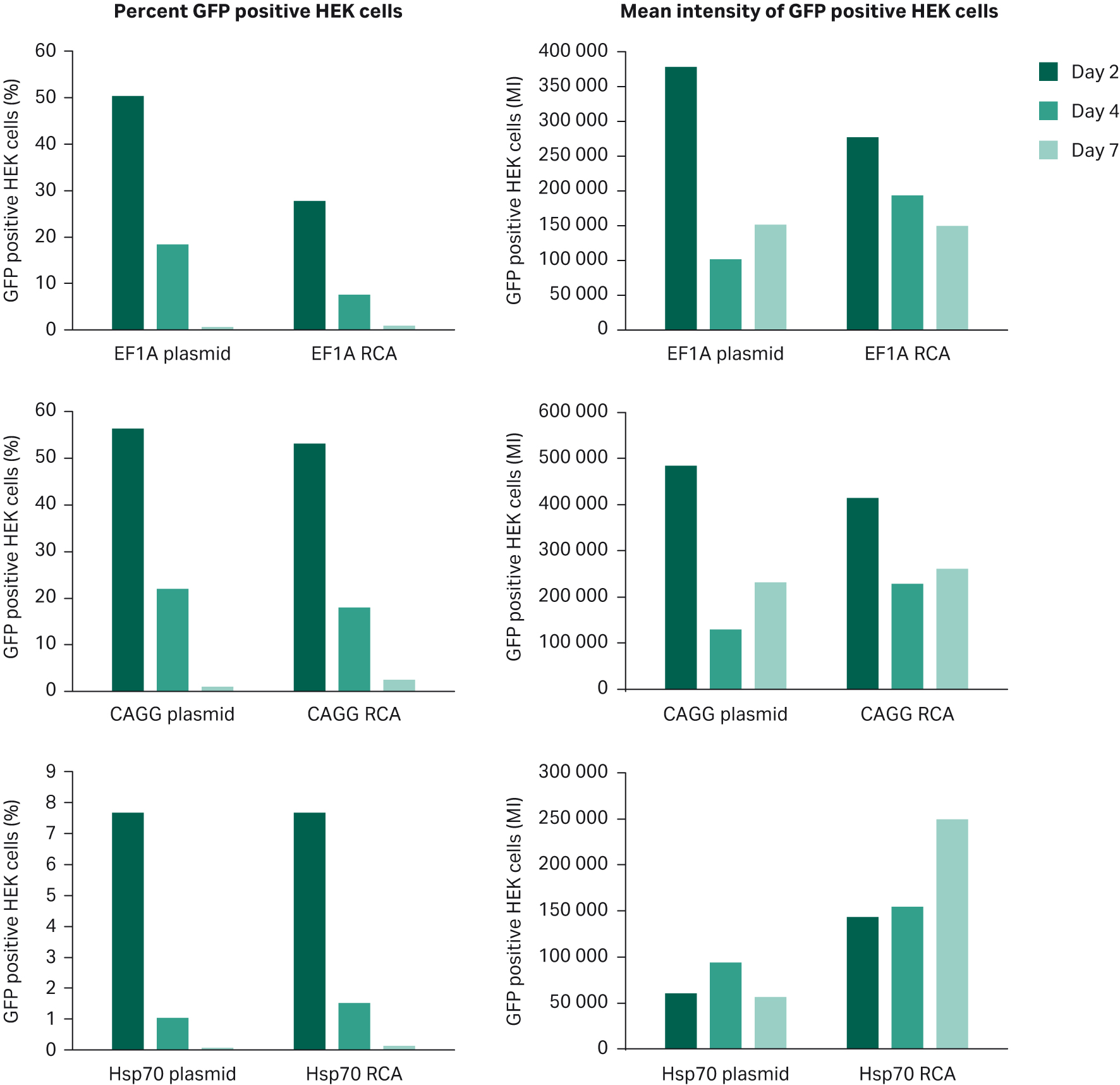
Fig 6. Efficient transfection into mammalian cells can be achieved directly from RCA derived product. Supercoiled plasmid and RCA derived product, both carrying the GFP gene with either EF1A, CAGG or Hsp70 promotors were each transfected into HEK293 cells using Dharmafect™ transfection agent. Analysis was via flow cytometry with IN Cell analyzer. It can be seen that promotor efficiency plays a role, but that RCA-derived product is suitable for direct transfection
Another method that takes advantage of this, is the synthesis of viral subunits or even entire viral particles or infectious clones which can be assembled within a suitable host following transfection.(25,26) In such cases, a TempliPhi kit is used to amplify viral sequences which are linearized and capped prior to rescue of complete viral particles.(27) In this way, study of infectious agents is possible in the lab without access to patient samples, allowing study into structure and function with capability to generate targets for vaccine production.
Mitochondrial DNA enrichment
Mitochondria have long captured our interest with their own small circular genome of approximately 16,500 bp (in humans) contributing 37 essential genes, largely involved in oxidative phosphorylation pathways. Inherited from the maternal line, mitochondrial DNA sequences are an extremely valuable resource in evolutionary and phylogenetic studies, as well as for determining relatedness of populations. Being circular, the mitochondrial genome provides an ideal template for TempliPhi rolling circle amplification, which has been used successfully as part of many sample preparation strategies in this area.(28,29,30)
Mitochondria are also recognized as playing a role in several disease states, with specific mutations in these genes giving rise to known genetic disorders such as lactic acidosis and age-related hearing loss. Researchers therefore continue to focus closely on mitochondrial genome sequence and structure. One such study by Jaberi et al identified mtDNA mutations relevant for neurodegeneration and cancer using their high resolution MitoSV-seq method which utilizes TempliPhi RCA to amplify the circular mitochondrial DNA from single cells.(4) This allows ultra-deep sequencing to investigate mutations and structural variations.
To de-branch or not to de-branch
The schematic of rolling circle amplification depicts strand displacement and multiple priming on newly synthesized strands, creating a complex structure of predominantly double stranded DNA with branches. For most applications, there is little further processing required. For example, TempliPhi amplification product is used directly in dye terminator sequencing with concatemeric copies each acting as a template. Similarly, direct use as template for mRNA production and cell-free protein expression is preferable. Even transformation using TempliPhi-amplified DNA into E.coli, yeast and mammalian cells occurs readily without de-branching, circularization or purification.
In applications considered sensitive to structure, e.g. nanopore sequencing, some researchers may carry out de-branching of the amplified product prior to sequencing. Typical treatments include incubation with T7 endonuclease or S1 nuclease. An alternative approach is to simply shear the DNA to a usable size before preparation.
In summary
TempliPhi kits provide a streamlined and user-friendly solution that is routinely used for simple high throughput preparation of circular DNA constructs for dye terminator sequencing. However, rolling circle amplification should not be considered in such a limited context, but more as a technique or tool that can be used in a range of applications to solve many challenges. The ability to amplify micrograms of DNA from nanograms of starting material at high fidelity without PCR cycling or any need for specific primers means that its application is simple, efficient and reliable. TempliPhi kits offer a ready-made solution for exploring the benefits of rolling circle amplification, exploiting the unique characteristics of phi29 DNA polymerase for amplification of circular or circularized templates wherever it may be required.
While the focus of this article is on rolling circle amplification, Cytiva also provides the GenomiPhi product range for whole genome amplification that is also based on phi29 DNA polymerase technology.
Read more about DNA amplification methods and our amplification solutions
- Enzo Z. Poirier, E., Goic, B., Tome-Poderti, L., Frangeul, L., Boussier, J., Gausson, V. et al. Dicer-2-Dependent Generation of Viral DNA from Defective Genomes of RNA Viruses Modulates Antiviral Immunity in Insects. Cell Host & Microbe 23, 353-365 (2018)
- Sophie Lanciano, S., Carpentier, M., Llauro, C., Jobet, E., Robakowska-Hyzorek, D., Lasserre, E. et al. Investigating retrotransposons in extrachromosomal DNA - Sequencing the extrachromosomal circular mobilome reveals retrotransposon activity in plants. PLoS Genet. 13, e1006630 (2017)
- K Rosario, M., Morrison, C., Mettel, K., Betancourt, W. Whole community genome amp - Novel Circular Rep-Encoding Single-Stranded DNA Viruses Detected in Treated Wastewater. Microbiol Resour Announc 8, 318-319 (2019).
- Jaberi, E., Tresse, E., Grønbæk, K., Weischenfeldt, J., Issazadeh-Navikas, S. Identification of unique and shared mitochondrial DNA mutations in neurodegeneration and cancer by single-cell mitochondrial DNA structural variation sequencing (MitoSV-seq). EBioMedcine 57, 102868 (2020).
- Blanco, L., Bernad A., Lazaro J.M., Martin G., Garmendia C. & Salas M. Highly efficient DNA synthesis by the phage phi 29 DNA polymerase. Symmetrical mode of DNA replication. J. Biol. Chem. 264, 8935- 8940 (1989).
- Blanco, L. & Salas, M. Characterization and purification of a phage Φ29-encoded DNA polymerase required for the initiation of replication. Proc. Natl. Acad. Sci. U.S.A. 81, 5325–5329 (1984).
- Blanco, L. & Salas, M. Characterization of a 3′-5′ exonuclease activity in the phage Φ29-encoded DNA polymerase. Nucleic Acids Res. 13, 1239–1249 (1985).
- Dunning, A.M., P. Talmud, & S.E. Humphries. Errors in the polymerase chain reaction. Nucleic Acids Res. 16,10393 (1988).
- John R. Nelson, j., Cai, Y., Giesler, T., Farchaus, J., Sundaram, S., Ortiz-Rivera, M. et al. TempliPhi, φ29 DNA Polymerase Based Rolling Circle Amplification of Templates for DNA Sequencing. BioTechniques 32, S44-S47 (2002).
- Dean, F.B., Nelson, J.R., Giesler, T.L. & Lasken, R.S. Rapid Amplification of plasmid and phage DNA using phi29 DNA polymerase and multiply-primed rolling circle amplification. Genome Research 11, 1095-1099 (2001).
- Dang, J., Bruce, G., Zhang, Q., Kiviat, N. Identification and characterization of novel human papillomaviruses in oral rinse samples from oral cavity and oropharyngeal cancer patients. Journal of oral biosciences 61, 190-194 (2019).
- Gheit, T., Dutta, S., Oliver, J., Robitaille, A., Hampras, S., Combes, et al. Isolation and characterization of a novel putative human polyomavirus. Virology 506, p45-54 (2017).
- Gräfe, D., Ehlers, B., Mäde, D., Ellerbroek, L., Seidler, T., Johne, R. Detection and genome characterization of bovine polyomaviruses in beef muscle and ground beef samples from Germany. International Journal of Food Microbiology 241, 168-172 (2017).
- Kraberger, S., Cook, C., Schmidlin, K., Fontenele, R., Bautista, J., Smith, B. et al. Diverse single-stranded DNA viruses associated with honey bees (Apis mellifera). Infection, Genetics and Evolution 71, 179-188 (2019).
- Jarullah, B., Sohrab, S., Jarullah, J. First Report of Tomato leaf curl Sudan virus Infecting Tomato Plants in Gujarat State, India. Plant Disease 101, 1685-1685 (2017).
- Fiallo-Olivé, E., Chirinos, D., Castro, R., Navas-Castillo, J. A Novel Strain of Pepper Leafroll Virus Infecting Common Bean and Soybean in Ecuador. Plant Disease 103, 167 (2019).
- Qadir, R., Khan, Z., Monga, D., Khan, J. - Diversity and recombination analysis of Cotton leaf curl Multan virus: a highly emerging begomovirus in northern India. BMC Genomics 20, 274 (2019).
- Van Nostrand, J., Yin, H., Wu, L., Yuan, T., Zhou, J. Hybridization of Environmental Microbial Community Nucleic Acids by GeoChip. Microbial Environmental Genomics 1399, 183-196 (2016)
- Mehta, D., Hirsch-Hoffmann, M., Were, M.,Patrignani, A., Shan-E-Ali Zaidi, S. et al. A new full-length circular DNA sequencing method for viral-sized genomes reveals that RNAi transgenic plants provoke a shift in geminivirus populations in the field. Nucleic Acids Research 47, 9 (2019).
- Carlson, E., Gan, R., Hodgman, C. & Jewett, M. Cell-Free Protein Synthesis: Applications Come of Age. Biotechnol.Adv. 30, 1185–1194 (2012).
- Dopp, J.L., Rothstein, S.M., Mansell, T.J., Reuel, N.F. Rapid prototyping of proteins: Mail order gene fragments to assayable proteins within 24 hours. Biotechnology and Bioengineering 116, 667-676 (2019).
- Dopp, J.L. & Reuel, F. One-Pot E. coli Cell-Free Extract for in vitro Expression of Disulfide Bonded Proteins bioRxiv doi: https://doi.org/10.1101/2019.12.19.883413 (2019)
- Hadi, T., Nozzi, N., Melby, J., Gao, W., Fuerst, D., Kvam, E. Rolling circle amplification of synthetic DNA accelerates biocatalytic determination of enzyme activity relative to conventional methods. Nature Scientific Reports 10, 10279 (2020).
- Duthie, R., Gao, W., Lowery, L., Davis, B., Nelson, J. Synthetic DNA for Transient Engineering of Cell Culture, Poster AGBT (2005).
- Schnepp, B., Jensen, R., Clark K., Johnson, P. Infectious Molecular Clones of Adeno-Associated Virus Isolated Directly from Human Tissues. Journal of Virology 83, 1456–1464 (2009).
- Lucarelli, J., Duggal, N., Brault, A., Geiss, B., Ebel, G. Rescue and Characterization of Recombinant Virus from a New World Zika Virus Infectious Clone. J. Vis. Exp. 124, 55857 (2017)>
- Grubaugh, N., Fauver, J., Ruckert, C.,Weger-Lucarelli, J.,Garcia-Luna, S., Murrieta, R., et al. Mosquitos Transmit Unique West Nile Virus Populations during Each Feeding Episode. Cell Reports 19, 709-718 (2017).
- Zardoya, R., Suárez, M. Sequencing and Phylogenomic Analysis of Whole Mitochondrial Genomes of Animals. Phyologenomics 442, 185-200 (2008)
- Tang, S. & Hyman, B. Rolling Circle Amplification of Complete Nematode Mitochondrial Genomes. Journal of Nematology 37, 236–241 (2005)
- Simison, W., Lindberg, D., Boore, J. Rolling circle amplification of metazoan mitochondrial genomes. Molecular Phylogenetics and Evolution 39, 562-567 (2006)