- What is size exclusion chromatography?
- How does size exclusion chromatography work?
- When should I use preparative SEC for protein purification?
- When should I use analytical SEC?
- How to use SEC for desalting and buffer exchange
- Running conditions for SEC
- Cleaning and storing your SEC column
- Troubleshooting common issues when performing SEC
What is size exclusion chromatography?
Size exclusion chromatography (SEC), also known as gel filtration, is the mildest of all the chromatography techniques. SEC separates molecules by differences in size as they pass through a resin packed in a column. Unlike techniques such as ion exchange chromatography (IEX) or affinity chromatography (AC), molecules do not bind to the chromatography resin, which means that buffer composition does not directly affect resolution (the degree of separation between peaks). Consequently, a significant advantage of SEC is that conditions can be varied to suit the type of sample or the requirements for further purification, analysis, or storage without altering the separation.
Discover our free eLearning course that guides you through a range of chromatography techniques.
How does size exclusion chromatography work?
SEC resins consist of a porous matrix of spherical particles (beads) that lack reactivity and adsorptive properties. After sample has entered the column, molecules larger than the pores are unable to diffuse into the beads, so they elute first. Molecules that range in size between the very big and very small can penetrate the pores to varying degrees based on their size. If a molecule is smaller than the smallest of the pores in the resin, it will be able to enter the total pore volume. Molecules that enter the total pore volume are eluted last. Samples are eluted isocratically so there is no need to use different buffers during the separation.
Watch this animation to visualize what happens inside a chromatography column during a run
SEC resins used for purification of biomolecules have a designed fractionation range, which defines the range of molecular weights (Mr) that have partial access to the pores of the particle and can be separated. Fractionation ranges of Mr from 100 to 7000 are suited to separation of peptides and other small biomolecules while a resin with a fractionation range of 100 000 to 300 000 is excellent for purification of large proteins such as antibodies. The exclusion limit of the resin indicates the size of the molecules that are excluded from the pores and therefore elute in the void volume (the liquid volume that does not enter the pores of the resin and elutes first in the chromatography purification). The fractionation ranges and/or exclusion limits of chromatography resins are shown below.
| Superdex™ 30 Increase column | Fractionation range (globular proteins) | Resolution |
|---|---|---|
| Superdex™ 30 Increase column | 100 to 7000 | Highest resolution |
| Superdex™ 75 Increase column | 3000 to 70 000 | |
| Superdex™ 200 Increase column | 10 000 to 600 000 | |
| Superdex™ 30 prep grade column | 100 to 7000 | |
| Superdex™ 75 prep grade column | 3000 to 70 000 | |
| Superdex™ 200 prep grade column | 10 000 to 600 000 | |
| Superose™ 6 Increase column | 5000 to 5 000 000 | |
| Superose™ 6 prep grade column | 5000 to 5 000 000 | |
| Superose™ 12 prep grade column | 1000 to 300 000 | |
| Sephacryl™ S-100 HR column | 1000 to 100 000 | |
| Sephacryl™ S-200 HR column | 5000 to 250 000 | |
| Sephacryl™ S-300 HR column | 10 000 to 1 500 000 | |
| Sephacryl™ S-400 HR | 20 000 to 8 000 000 | |
| Sephacryl™ S-500 HR column | 40 000 to 20 000 000 | |
| Sephadex™ G-10 resin | 700 (exclusion limit) | |
| Sephadex™ G-25 SF resin | 5000* | |
| Sephadex™ G-25 F resin | 5000* | |
| Sephadex™ G-25 M resin | 5000* | |
| Sephadex™ G-50 F resin | 30 000* | |
| Sephadex™ LH-20 resin | 4000 to 5000† | Lowest resolution |
* Exclusion limit.
† Exclusion limit depends on the solvent used.
Particle size, column dimension, minimizing system volumes, flow rate, and the packing of the column are all important factors for you to consider to give high-resolution (narrow, well-separated) peaks. The selectivity of a resin (the distance between two peaks) together with the efficiency affect the resolution between peaks. The selectivity of a resin depends on the properties of the resin (such as pore size distribution), the interactions between sample and resin, and the conditions used. The efficiency of a resin depends mainly on particle size, the column format, and the packing of the column.
Each of these areas is covered in even greater detail in the Size Exclusion Chromatography Handbook.
When should I use preparative SEC for protein purification?
Preparative SEC is a high-resolution size-based separation of biomolecules with collection fractionation for further analysis or scale-up. Preparative SEC is performed to isolate one or more components of a sample. Separated components can be directly transferred to a suitable buffer for assay or storage. Sample volumes of 0.5% to 4% of the total column volume are applied at low flow rates using long columns, often 60 cm or longer. As the separation takes place in only 1 CV, it is essential to have a well packed column for good results in SEC. For convenience and optimal performance, prepacked SEC columns are recommended.
Choosing resins and prepacked columns
Efficient column packing is essential in preparative SEC, particularly for high-resolution separations. Depending on the resolution and yield needed, column bed heights of up to 100 cm are recommended for preparative SEC separation with resin bead sizes of ~ 9 to ~ 34 µm (see Table 1). The required bed volume and hence the diameter of the column is determined by the sample volume.
A well-packed column should produce narrow symmetrical peaks with minimal peak broadening. The uniformity of the packed bed and the particles influences the flow through the column and hence affects the shape and width of the peaks. High-performance SEC resins with high bed uniformity (smaller and more uniform particles) give decreased peak widths and improved resolution.
Table 1. Characteristics of resins used for preparative SEC purifications
| Product | Efficiency1 | Operational2 | Cleaning-in-place (CIP) pH | Particle size, d50v (µm)4 |
|---|---|---|---|---|
| Superdex™ 30 Increase | > 43 000 | 3 to 12 | 1 to 14 | ~ 9 |
| Superdex™ 75 Increase | > 43 000 | 3 to 12 | 1 to 14 | ~ 9 |
| Superdex™ 200 Increase | > 48 000 | 3 to 12 | 1 to 14 | ~ 8.6 |
| Superdex™ 30 prep grade | > 13 000 | 3 to 12 | 1 to 14 | ~ 34 |
| Superdex™ 75 prep grade | > 13 000 | 3 to 12 | 1 to 14 | ~ 34 |
| Superdex™ 200 prep grade | > 13 000 | 3 to 12 | 1 to 14 | ~ 34 |
1Theoretical plates per meter (prepacked columns only).
2pH range where resin can be operated without significant change in function.
3pH range where resin can be subjected to cleaning- or sanitization-in-place without significant change in function.
4Median particle size of the cumulative volume distribution.
For other general considerations regarding sample and buffer composition, see the Running conditions for SEC.
Optimizing your separation
The success of SEC depends primarily on choosing conditions that give sufficient selectivity and counteract peak broadening effects during the separation. Prepacked columns are delivered with recommended running conditions that give satisfactory results in most situations, but optimization might sometimes be necessary to reach the required resolution. As mentioned in How does size exclusion chromatography work, resolution is a function of the selectivity of the resin and the efficiency of that resin to produce narrow peaks (minimal peak broadening).
If optimization is needed, follow these steps (given in order of priority):
- Select a resin with a suitable fractionation range providing optimal resolution.
- Select a column with a bed height providing the required resolution. A bed height between 30 and 100 cm is recommended for preparative separation.
- Select a column size appropriate for the volume of sample that needs to be processed.
- Select the highest flow rate that maintains resolution and minimizes separation time.
How to scale up preparative SEC
After establishing a separation on a small column, larger columns can be packed to process larger sample volumes in a single step. When scaling up for instance in process development, or where larger sample volumes are available, the column diameter and flow rate volume need to be increased compared to high resolution preparative SEC and analytical SEC.
Note that with scaling up, small particles generate high back pressure, which can restrict separation scale-up.
When comparing results between columns of different size, the flow velocity (cm/h) is a useful measurement. Results obtained at the same flow velocity on different sized columns will be comparable as far as the effects of flow velocity are concerned.
To scale up a separation, follow this advice:
- Optimize the separation at small scale, with, for example, Superdex™ Increase 10/300 or HiLoad™ 16/600 column formats.
- Maintain the sample-to-column volume ratio.
- Maintain the bed height and increase the column volume by increasing the cross-sectional area of the column.
- Run the separation at the same flow velocity (cm/h) as used on the smaller column.
Different factors related to the equipment can affect the performance after scale-up. Peak broadening can occur on a larger system if system volumes are introduced or if the distribution system of the larger column has lower flow efficiency. This will cause extra dilution of the target molecule or even loss of resolution if the application is sensitive to variations in efficiency.
Preparative SEC application example
Using preparative SEC to purify IGF-1
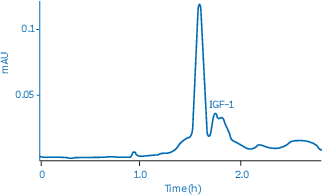
Figure 1 shows the separation of IGF-1 (Mr 7600) from its ZZ fusion protein partner (Mr 14 500) and uncleaved material on HiLoad™ 16/600 Superdex™ 75 pg. This column is an excellent choice since the resin separates proteins and peptides in the molecular weight range Mr 3000 to 70 000 and performs best between Mr 8000 and 50 000.
| Column: | HiLoad 16/600 Superdex™ 75 pg |
| Sample: | IGF-1, ZZ fusion protein, and uncleaved material |
| Buffer: | 0.15 ammonium acetate, pH 6.0 |
| Flow rates | 0.75 mL/min (22.5 cm/h) |
Fig 1. Separation of recombinant IGF-1 (Mr 7600) from its ZZ fusion protein partner (Mr 14 500) and uncleaved material.
When should I use analytical SEC?
Analytical SEC is a very effective method for protein analysis as it allows size profiling of protein samples due to the mild separation conditions that can be used to obtain high-resolution separations. Analytical SEC can be used to:
- Monitor protein preparation quality
- Evaluate protein stability
- Study complex formation
- Evaluate tendency to aggregate and quantity of aggregates
- Evaluate tendency to degrade and quantity of degraded forms
- Identify protein interaction partners and interaction conditions
Analytical SEC is performed to check the quality of the sample or to study the properties of a biomolecule. Small sample volumes, often 0.3% to 0.5% of the column volume are applied at low flow rates using long columns, typically 30 cm. Prepacked columns are an excellent choice for ensuring reliable results and for convenience.
Analytical SEC can also be used for rapid purity checks and screening. For this, shorter columns of typically 15 cm, which provide adequate resolution are used giving short cycle times together with small sample volume and low buffer consumption. Note that shorter columns typically used for purity check/screening give lower resolution than longer columns.
The types of prepacked columns used in analytical SEC are shown in Figure 2.
Fig 2. Three different SEC column sizes of for various application needs.
Selecting a resin for performing analytical SEC
For highest resolution, select a resin with suitable fractionation range and a small particle size. The size range of 4 to 12 µm is traditionally the standard for analytical SEC resins. However, the trend is towards smaller particles of < 2 µm, with the use of ultra high-performance liquid chromatography (UHPLC) systems for even faster separations in high-throughput mode.
Superdex™ Increase resins provide high resolution and shorter run times than predecessor Superdex™ resins. This is achieved by using a high-flow agarose base matrix with higher pressure-flow tolerance, smaller particle size (~ 9 μm), and narrower particle size distribution. The design enables high-resolution separations in short run times, resulting in excellent analytical results and high final purity. Superdex™ Increase resins cover a molecular weight (Mr) range and are suitable for:
- separation of peptides and other small biomolecules with Mr from 100 to 7000
- separation of proteins ranging from 3000 to 70 000
- separation of proteins from 10 000 to 600 000 with an optimized resolution for the antibody molecular weight range of 100 000 to 300 000
Should I choose silica or agarose-based resins?
The most commonly used resins for analytical purposes are based on either silica or agarose. Agarose is obtained from natural sources and contains a very low amount of ionic and hydrophobic groups that could interact with the compound of interest. In contrast, silica-based resins have silanol groups that require coating before use. Properties of silica-based and agarose resins are given in Table 2.
Table 2. Properties of silica-based and agarose-based resins1
| Property | Silica particles | Agarose particles |
|---|---|---|
| Chemical composition | SiO2 | Polysaccharide (D-galactose-3,6-Anhydro-L-galactose) |
| Surface coating | Silanols are blocked to minimize absorption | Not required |
| pH stability | Typically, pH 2 to 8 (operational)2 | Typically, pH 3 to 11, operational2 pH 1 to 12, cleaning-in-place (CIP)3 |
| Mechanical | Rigid, high pressure stability | Rigidity achieved by chemical cross-linking |
| Porosity | Can be controlled to achieve the desired separation range | Can be controlled to achieve the desired separation range |
1 This information is taken from product information on vendor’s web pages.
2 pH range where resin can be operated without significant change in function.
3 pH range where resin can be subjected to cleaning- or sanitization-in-place without significant change in function.
An advantage with agarose-based resins is that the same SEC column can be used for different samples. This is not the case with silica-based resins as they do not tolerate cleaning in NaOH, since silica-based resins are stable below a pH of about 7 but do not tolerate pH above 8. Efficient cleaning prolongs the column lifetime and minimizes the risk of carry-over between runs allowing the same SEC column to be used for different samples. For analytical agarose-based SEC columns, it may be sufficient to clean the column by regular injections of a small volume of NaOH.
Analytical SEC application example
Analysis of monoclonal antibody homogeneity
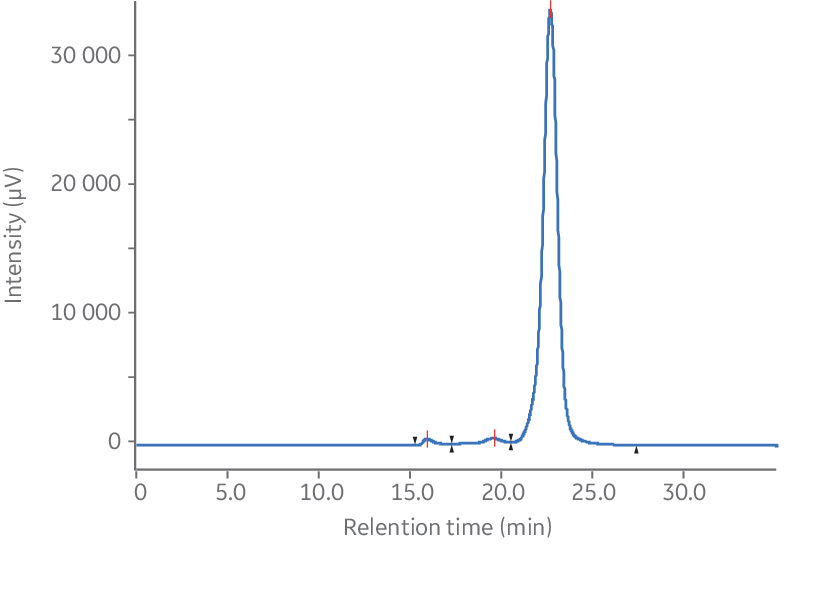
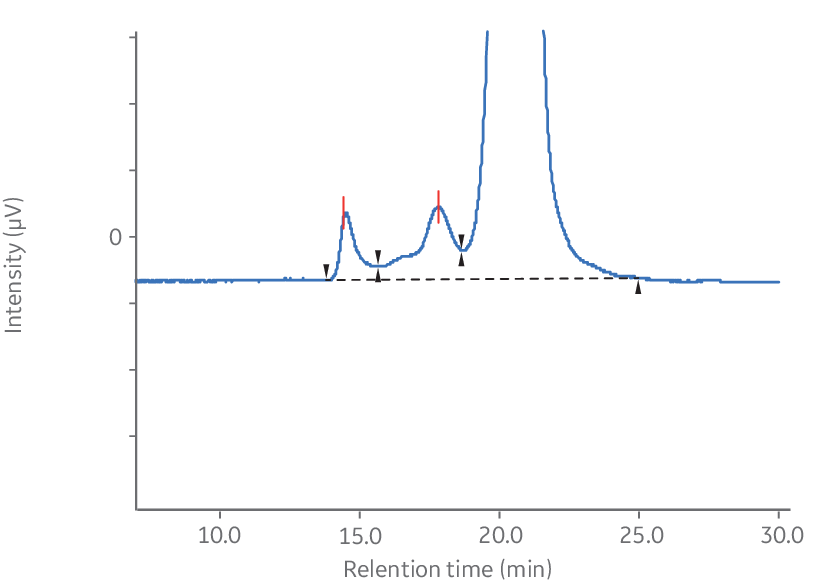
A critical step in monoclonal antibody (mAb) production and characterization is the analysis of aggregates and determination of purity of the monomeric fraction. Figures 3 A and B illustrate the separation of a monoclonal mouse IgG antibody from its aggregates using Superdex™ 200 Increase 10/300 GL. As shown, the aggregates and dimers were very well separated from the monomeric peak.
| Column: | Superdex™ 200 Increase 10/300 GL |
| Sample: | mAb |
| Sample volume: | 5 µL |
| Flow rate: | 0.75 mL/min |
| System: | HPLC system |
Fig 3. Analysis of monoclonal mouse IgG1 antibody aggregates using Superdex™ 200 Increase 10/300 GL. (A) Chromatogram without magnification. (B) Magnified chromatogram showing details of aggregates, dimer, and monomer peaks.
How to use SEC for desalting and buffer exchange
Desalting and other types of buffer exchange are examples of group separations on size exclusion chromatography (SEC) resins. The purpose is to separate small molecules, such as salts, from large biomolecules such as proteins. Samples can be prepared for storage or for other chromatography techniques and assays. Large sample volumes—up to 30% of the total column volume—can be applied at high flow rates using broad, short columns. Examples of group separations include:
- Removal of phenol red from culture fluids prior to anion exchange chromatography or nucleic acid preparations
- Removal of unincorporated nucleotides during DNA sequencing
- Removal of free low-molecular weight labels
- Termination of reactions between macromolecules and low molecular weight reactants
- Removal of products, cofactors, or inhibitors from enzymes
- Removal of unreacted radiolabels such as [α-32P] adenosine triphosphate (ATP) from nucleic acid labeling reactions
Desalting and buffer exchange can be used before purification, between purification steps, and/or after purification. It can typically be performed in a range of prepacked columns, spin columns, or 96-well plates depending on sample volume and on the method of operation (centrifugation or vacuum extraction). For advice on choosing different formats for your desalting/buffer exchange see our blog on optimizing protein desalting for more information.
For most separations, the instructions supplied with each product ensure satisfactory results and very little optimization should be necessary. Ensure that buffer conditions are optimal for the separation and that the maximum sample volume for the column is not exceeded.
Advantages of desalting using SEC
Desalting using size exclusion chromatography provides several advantages over dialysis, which is generally a slow technique that requires large volumes of buffer and carries the risk of losing material and activity of the target molecule during handling.
When desalting, sample volumes of up to 30% of the total column volume can be processed. SEC resins with large particles packed in broad, short columns can be used, which enables fast separations using high flow rates. A sample can be processed in less than 5 min with a high recovery. The recovery depends on the biomolecule, the column format, and the purification protocol but is often > 95%. Low target molecule concentrations often result in lower recovery.
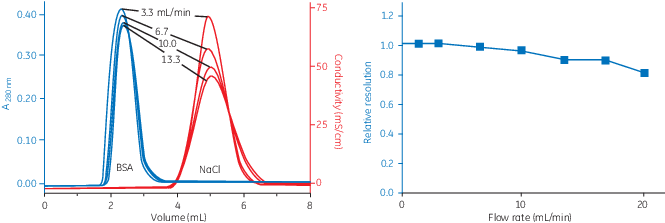
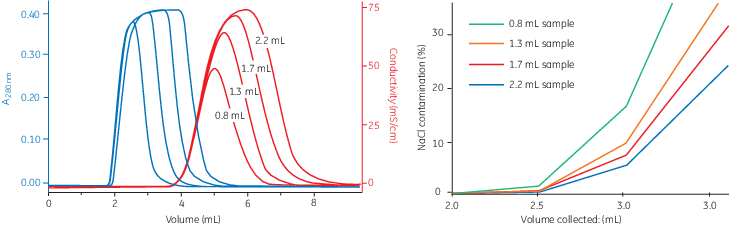
The high speed and capacity of the separation allows even relatively large sample volumes to be processed rapidly and efficiently, as illustrated in Figure 4 and 5. Figure 4 shows that the resolution between the protein peak and the salt peak remains high when increasing the flow rate. The position, shape, and width of the peaks are similar in all runs irrespective of flow rate.
| Column: | HiTrap™ Desalting |
| Sample: | Bovine serum albumin, 2 mg/mL in 500 mM NaCl, 50 mM sodium phosphate, pH 7 |
| Buffer: | 50 mM sodium phosphate, 150 mM sodium chloride, pH 7.0 |
| Sample volume: | 0.8 mL |
| Flow rates: | 1.7, 3.3, 6.7, 10.0, 13.3, 16.7, 20.0 mL/min |
Fig 4. Influence of flow rate on resolution.
Figure 5 shows the effect of increasing sample volumes on resolution. The peaks become wider when the sample volume increases, which leads to a larger overlap of the protein peak and the NaCl peak. The effect on resolution is illustrated by the NaCl concentration in the collected protein fraction. The size of the collected volume will affect the NaCl concentration in the protein fraction and also the target protein yield.
| Column: | HiTrap™ Desalting |
| Sample: | Bovine serum albumin, 2 mg/mL in 50 mM sodium phosphate, 500 mM sodium chloride, pH 7 |
| Buffer: | 50 mM sodium phosphate, 150 mM sodium chloride, pH 7.0 |
| Sample volume: | 0.8, 1.3, 1.7, 2.2 mL |
| Flow rate: | 5 mL/min |
Fig 5. Influence of sample volume on resolution.
Resin selection for desalting and buffer exchange
SEC based on Sephadex™ chromatography resins enables desalting and buffer exchange of biomolecules. There are many different types of Sephadex™ resins available, which have been a mainstay in desalting and buffer exchange since the 1950´s. Sephadex™ is prepared by cross-linking dextran with epichlorohydrin. The exclusion limit and the fractionation range are controlled by varying the degree of cross-linking of the resin.
For most applications we recommend Sephadex™ G-25 or G-10. Use G-25 if your protein of interest has a molecular weight > 5000 and G-10 for smaller molecules with a molecular weight > 700. Loose resins for column packing are available, but we suggest prepacked formats for time savings, ease of use, and reproducibility. Biomolecules with sizes above the exclusion limit of the resin can be separated from contaminants such as salts, dyes, and radioactive labels that have access to the pores. The characteristics of Sephadex™ resins are summarized in Table 3.
Table 3. Sephadex™ characteristics and typical applications
| Resin | Particle size (µm) | Swelling factor§ | Separation range (Mr) globular proteins | Typical applications |
|---|---|---|---|---|
| Sephadex™ G-10 | 40 to 120* | 1.4 | < 7 × 102 | Separation of small biomolecules such as peptides (Mr > 700) from smaller molecules (Mr < 100). |
| Sephadex™ G-25 Superfine Sephadex™ G-25 Fine Sephadex™ G-25 Medium Sephadex™ G-25 Coarse |
20 to 50† 20 to 80† 50 to 150‡ 100 to 300‡ |
1.7 1.7 1.7 1.7 |
1 × 103 to 5 × 103 1 × 103 to 5 × 103 1 × 103 to 5 × 103 1 × 103 to 5 × 103 |
Recommended for most group separations involving globular proteins, excellent for removing salts and other small contaminants from molecules with Mr > 5000. |
| Sephadex™ G-50 Fine | 20 to 80† | 2.1 | 1 × 103 to 3 × 104 | Suitable for separation of molecules with Mr > 30 000 from molecules Mr < 1500 such as labeled protein or DNA from unconjugated dyes. Often used for removal of small nucleotides from longer chain nucleic acids. |
* ≥ 85% of the volume of particles are found within the range shown.
† ≥ 80% of the volume of particles are found within the range shown.
ೠ90% of the volume of the particles are found within the range shown.
§ Suitable swelling solutions are water, buffers, or salt solutions. Note that Sephadex particles shrink in alcohol solutions.
Use this flow chart to help select the correct prepacked columns for desalting and buffer exchange.
Sample and buffer considerations
When performing size exclusion chromatography, the sample should be fully dissolved. Centrifuge or filter the sample to remove particulate material and use degassed buffers to avoid introducing air into the column.
Use 10 to 50 mM sodium phosphate buffer, with at least 25 mM NaCl, pH 7.0 to 7.4, or select an appropriate buffer depending on the purpose of the separation. To prevent potential ionic interactions the presence of a low salt concentration (25 mM sodium chloride) is recommended in the buffer during desalting. Avoid salt concentration >1.0 M. Volatile buffers such as 100 mM ammonium acetate or 100 mM ammonium hydrogen carbonate can be used if it is necessary to avoid the presence of sodium chloride.
- Re-equilibration is not necessary between runs with the same buffer. Extended equilibration might be needed with buffers containing detergents. Equilibrate with at least 5 column volumes (CV).
Generally, sample concentration does not significantly influence the separation providing viscosity does not become too high in relation to the viscosity of the eluent. Also, the target molecule needs to be stable and soluble at the concentration used.
Sample volume
Desalting generally causes dilution of the sample (except for spin protocols). Minimal dilution is obtained when the maximum desalting capacity of the column is utilized. When possible, select a prepacked column that is suited to the volume of sample that needs to be desalted. As the sample volume increases the dilution factor decreases.
For most applications, up to 30% of the total bed volume may be used. The final dilution factor also depends on the collected elution volume. Maximizing the recovery of the target molecule by collecting a large volume will increase the dilution of the sample. Note that large sample volumes and collection of large elution volumes can increase the amount of salt remaining in the sample after elution.
- Use a large sample volume (≤ 30% of the total bed volume) for minimal sample dilution. Application of sample volumes larger than 30% of the bed volume will give less efficient desalting. Reduce sample volume (< 30% of the column volume) when highest resolution is required for the separation.
Optimizing your chromatography system
Using a chromatography system equipped with monitors facilitates the possibility to follow the separation and simplifies optimization of the separation. Elution of biomolecules can be monitored by UV absorption (280 nm for proteins) and the appearance of salt can be followed exactly by a conductivity monitor.
- If conductivity cannot be monitored and recovery of a completely desalted sample is the major requirement, apply sample volumes of between 15% and 20% of the total bed volume.
Running conditions for SEC
Buffer composition
Buffer and sample composition are important parameters to achieve high resolution. Properties of proteins such as number of charge groups, hydrophobicity, and size influence separation by SEC. Resins must therefore be chemically stable in a wide range of buffers to minimize interaction from the resin and changes in retention time, peak area, and resolution. Buffers with pH between 6.0 and 8.0 are normally used in SEC applications since many proteins are stable in that pH range.
- A running buffer of 50 mM sodium phosphate, 150 mM sodium chloride, pH 7.0 works well for many SEC separations.
Further, it is important to select an ionic strength that is beneficial for the protein of interest. Higher ionic strength decreases the retention time for positively charged proteins and increases it for negatively charged proteins. An ionic strength that is too high might also increase hydrophobic interactions between resin and proteins, which affects retention time as well as peak area and resolution. Some general tips to consider when making buffers are:
- Use high-quality water and chemicals. Solutions should be filtered through 0.45 μm or 0.22 μm filters before use. Degas buffers before SEC separation since air bubbles can reduce performance. Buffers are automatically degassed if filtered under vacuum.
- Buffers and columns must have the same temperature before use. Rapid changes in temperature, for example removing packed columns from a cold room and applying buffer at room temperature, can introduce air bubbles in the packed bed, resulting in poorer separation.
- Samples must be clear and free from particulate matter, especially when working with particle sizes of 34 μm or smaller. For small sample volumes, a syringe-tip filter of cellulose acetate or polyvinylidene difluoride (PVDF) might be sufficient.
Sample viscosity
Increased sample viscosity does not affect sample resolution but may cause an increase in back pressure on the column. Use lower flow rates for high-viscosity solutions and/or low temperature (Table 4). When running viscous samples such as samples containing glycerol, dilution of the sample or lowering the flow rate might be needed if the back pressure is increased during sample load.
- Dilute viscous samples, but no more than necessary to keep the sample volume low. For example, for proteins such as human serum albumin, the concentration should generally not exceed 70 mg/mL. Remember that viscosity varies with temperature.
- Sample concentration does not generally influence the separation if the viscosity does not differ by more than a factor of 1.5 from that of the buffer used. If the viscosity of the sample is very high, this may be compensated for by increasing the viscosity of the eluent, for example by addition of sucrose or dextran.
High protein concentration or additives in the sample can cause instability of the separation, with a resulting increase in back pressure on the column.
Note also that the viscosity of liquids increases with high salt concentrations. Mixing organic and aqueous solution will also affect the viscosity.
Table 4. Example of maximum flow rate limits at different viscosity and temperature, Superdex™ 30 Increase 10/300 GL
| Temperature | Buffer | Flow rate (mL/min) |
|---|---|---|
| 20⁰C to 25⁰C | Water 30% acetonitrile 20% ethanol 10% glycerol 40% methanol |
1.20 1.20 0.60 0.60 0.60 |
| 4⁰C to 8⁰C | Water 30% acetonitrile 20% ethanol 10% glycerol 40% methanol |
0.60 0.60 0.30 0.30 0.30 |
Sample volume
In analytical SEC, the sample volume should be approximately 0.3% of the bed volume to achieve optimal results. For most applications, the sample volume should not exceed 2% to achieve high resolution. Depending on the nature of the specific sample, it might be possible to load larger sample volumes, particularly if the peaks of interest are well separated. This can only be determined experimentally.
It is also relevant to consider how the sample loop (capillary) is filled and emptied, a factor that often is overlooked. The sample assumes the shape of an arrow with a broad head and long shaft in its passage through the capillary, rather than the shape of a compact plug. The sample therefore occupies a longer section of the capillary than expected, which can lead to a peak broadening effect.
To ensure reproducible analysis, always ensure that the syringe is completely filled for manual injection. For autosampler injection, it is important to check the settings on how the injections are performed to avoid inconsistent results.
Flow rate
One of the most important parameters that affects resolution is the flow rate (especially for large proteins). The goal for most separations is to achieve the required resolution in the shortest possible time.
If the analysis shows low resolution between protein peaks, the first action should be to set a lower flow rate for the run. Generally, a lower flow rate will allow time for molecules to diffuse in and out of the chromatography matrix and improve resolution. The effect is most pronounced for large molecules.
Decreasing the flow rate can, on the other hand, negatively impact on resolution for very small molecules.
Cleaning your SEC column
Cleaning columns routinely—for example after each 10–20 runs—will prolong the column life and contribute to the column’s optimal performance. Cleaning removes any precipitated proteins or other contaminants that might have built up on the column. In addition to routine cleaning, you should also clean the column if you see colored bands in the top of the column or if the backpressure has increased substantially.
SEC columns tolerate high pH well and can be cleaned many times with NaOH. The concentration of NaOH depends on the resin type: use 0.5 M NaOH for Superose™ and Superdex™ columns but only 0.2 M NaOH for Sephacryl™ columns. Cleaning of single-use products used for desalting/buffer exchange and containing Sephadex™ is not recommended.
Never store columns in NaOH. Instead, equilibrate the column immediately after cleaning with two column volumes (CV) of water followed by two CV of running buffer.
Detailed cleaning protocols are found in the instructions for each size exclusion chromatography column.
Store your column in 20% ethanol
Whenever you are not using your column for more than two days store it in 20% ethanol. Also, to prevent air from entering the column, connect the transport tool (provided with all new columns) to the column outlet. Then, fill it with storage solution up to about 50% of its total volume.If you follow these suggestions, your column should perform well for a long time.
Troubleshooting common issues when performing SEC
This section focuses on practical problems that can occur during size exclusion chromatography.
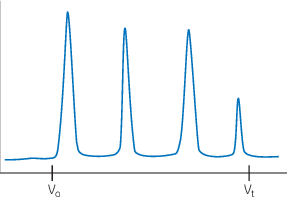
Satisfactory separation
Well-resolved, symmetrical peaks
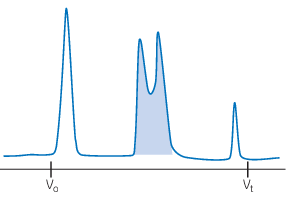
Poor resolution
Review factors affecting resolution, including the selectivity of the resin, particle size, sample-to-column volume ratio, flow rate, and the equipment used.
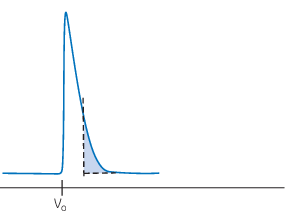
Asymmetric peaks: Tailing
Tailing peaks can be caused by an underpacked column, that is, a column packed at a very low pressure or flow rate. Ensure the resin is evenly packed. Tailing peaks can also be explained by uneven sample application or interactions with the column.
Asymmetric peaks: Fronting
Fronting peaks can be caused by an overpacked column, that is, a column packed at excessive pressure or flow rate. Peaks appearing before the void volume indicate channeling in the packed bed.
Late elution
Peaks eluting after 1 CV of buffer can be caused by nonspecific interactions with the resin. Include a wash step or extend the elution to ensure elution of late eluting molecules. Changing the buffer composition might also be beneficial.
If your sample is eluting at the wrong time, see Quick fixes for retention time issues
For more in-depth information, download our Protein purification troubleshooting guide
CY32845-12Oct22-AN
Other chromatography techniques
- Ion exchange chromatography
- Affinity chromatography (antibodies)
- Affinity chromatography (tagged proteins)
- Hydrophobic interaction chromatography
- Multimodal chromatography
Other resources
- Chromatography glossary
- Tips for maximizing your SEC column life
- Inspect your column to ensure you get it right
- How to optimize protein desalting
- How to avoid trapping air in the column
- Prevent columns from drying out using an ÄKTA™ system
- Prevent SEC columns from drying out using a syringe
- How to best replace Superose™ 12 with better-performing resins
- How to determine column-specific pressure limits for Superdex™ Increase and Superose™ Increase columns
- How to select a SEC column
SEC presentations