Introduction
1.1 PROTAC background
Proteolysis targeting chimeras (PROTACs) are small molecules that can induce the proteosomal degradation of a protein target by hijacking the protein regulatory machinery of the cell. PROTACs are made up of three moieties: a target protein binder, an E3 ubiquitin ligase binder, and a linker that connects the two. Through simultaneous recruitment of the target protein and ligase in a target:PROTAC:ligase ternary complex, PROTACs expose the target protein to ubiquitination, marking it for subsequent proteosomal degradation.
In drug design, using PROTACs comes with several therapeutic advantages:
- Unlike conventional occupancy-based drug modalities, which need to disrupt the target protein’s function by binding to an active site, PROTACs can potentially use any site on the target’s surface.
- The target protein is depleted, phenocopying knockdown.
- PROTACs don’t degrade with the target protein – they remain in the cell to turn over more target molecules, and this enables sub-stoichiometric dosing.
- Cooperative PROTACs can recruit a target protein into a ternary complex that has a higher affinity than the binders the PROTACs are derived from.
- Nonspecific binders can be converted into target-specific PROTACs due to differences between ternary complex cooperativities.
1.2 Biophysical aspects of PROTAC ternary complexes
Forming a highly stable ternary complex is crucial to the mode of action of PROTACs. Longer lasting ternary complexes are associated with higher levels of ubiquitination and faster rates of protein depletion. The stability of the ternary complex derives not just from the PROTAC’s affinity to each of the protein binding partners, but also from de novo protein-protein interactions (PPIs) between the target protein and the ligase induced on forming the complex.
When the newly formed PPIs are favorable, the target protein’s affinity to the PROTAC is higher when the PROTAC has prebound the ligase than it would be to the PROTAC alone, and vice versa. The cooperativity factor (α) generally gives a good indication of how favorable or unfavorable the de novo PPIs are. To calculate the cooperativity factor, divide the binding affinity (KD) of the target to the PROTAC by the KD of the target to the PROTAC pre-complexed with the ligase (Fig 1).
Fig 1. Schematic showing two possible pathways for forming PROTAC ternary complexes. Cooperativity (α) can be calculated by dividing the binding affinity of one protein binding partner (target or E3 ligase) to the PROTAC by the affinity of that protein binding partner to the PROTAC in the presence of saturating concentrations of the other protein binding partner.
PROTACs that form highly cooperative complexes often bind target proteins in a ternary complex with a higher affinity than their constituent target binder alone. Additionally, since the PPIs induced on formation of the ternary complex can extend over a large surface area of the two proteins, cooperativity can be sensitive to small differences in protein sequence throughout the binding interface. This can result in a PROTAC displaying target-specific degradation – even if the constituent target binder did not show homolog specificity.
Table 1. Summary of biophysical parameters for PROTAC ternary complexes
| Binary affinity | Affinity to both the target protein and the ligase are important for target engagement. If the compound is cooperative, you can potentially recoup deficiencies in binary affinity. |
| Cooperativity | Measured as a ratio of binary vs ternary complex affinities. Cooperativity can be particularly important for providing isoform and variant specificity. |
| ΔGcomplex | How favorable overall it is to form the ternary complex. |
| t1/2 | The stability of the PROTAC ternary complex is a key optimization parameter during PROTAC development. Longer-lasting complexes drive greater target ubiquitination, resulting in faster target degradation. That makes it critical for scientists to have methods to reliably investigate the affinity and kinetics of ternary complex association and dissociation. |
| Positive confirmation of ternary complex formation | Positive confirmation is implied when a PROTAC displays strong cooperativity, but it is important to validate by using an assay that can directly “observe” the formation of a ternary complex. This is particularly true for noncooperative and negatively cooperative PROTACs. |
| Binding stoichiometry | The target:PROTAC:ligase ternary complex generally follows a 1:1:1 stoichiometry. |
1.3 Biophysical methods for monitoring PROTAC ternary complexes
Recent years have seen the development of more biochemical and biophysical methods for profiling the mechanism of ternary complex formation, and each method has its own advantages and limitations. Scientists today can use a variety of competition-based assays including fluorescence polarization (FP), time-resolved fluorescence energy transfer (TR-FRET), and AlphaScreen/AlphaLISA technologies.[1,2,3 ] These methods make use of an interaction between a target protein or ligase and a binding partner coupled to a probe. When in complex with the target protein, the probe produces a signal that is distinct from the signal produced when the probe is free in solution.
Adding PROTAC displaces the probe binding partner from the target protein, resulting in readout for the assay. To determine the cooperativity of the system, you can compare the target protein’s affinity to the PROTAC in the absence and the presence of the ligase. However, these techniques can’t measure certain important biophysical parameters, such as the kinetic rate constants for ternary complex formation and the ternary complex t1/2.
Scientists often use ITC is to measure thermodynamic properties and confirm binding stoichiometry. However, ITC is limited by very low throughput and high protein requirements, and it provides no information on binding kinetics.
1.4 Biacore™ SPR systems enable further biophysical characterization for PROTACs:
Scientists use Biacore™ SPR systems for biophysical PROTAC characterization – it’s currently the only technique capable of measuring PROTAC ternary kinetics in vitro that the field has applied. The technique enables scientists to quickly determine nearly all of the mentioned biophysical parameters, providing:
- Measurement of both binary and ternary affinity and kinetics
- Positive confirmation of ternary complex formation, calculated by comparing the experimental Rmax with the theoretical Rmax of the binary vs ternary complex
- Cooperativity of a system within a single experiment
- Reproducible results
- Label-free assay readout
- Faster throughput compared to other biophysical techniques such as ITC
- Results correlate with in-solution assays such as ITC, as well as cell-based approaches.
1.5 Case study – kinetically profiling bromodomain:MZ1:VCB ternary complexes
The PROTAC MZ1 is based on two binders; VH032 and JQ1, connected by a linker.[4] VH032 is a potent and specific ligand for the von Hippel-Lindau (VHL) E3 ligase, and JQ1 is a pan bromodomain and extra terminal (BET) inhibitor.[5,6] While JQ1 doesn’t display significant selectivity toward either bromodomain (BD) of the BET proteins Brd2, Brd3, or Brd4, MZ1 has been shown to deplete Brd2 and Brd4 in initial degradation rate experiments (ƛ = 13 × 103 min−1 and 14-18 × 103 min−1, respectively) more rapidly than Brd3 (ƛ = 4 × 103 min−1). [7]
In this note, we demonstrate the use of SPR assays to provide detailed mechanistic, kinetic characterization of PROTAC interactions that scientists can apply widely across the field. The assay was developed using a Biacore™ T200 system, but it’s transferable to other Biacore™ systems such as the Biacore™ X100, Biacore™ S200, and Biacore™ 8K series. This application example is based on work published by Roy et al. 2019 and aims to provide an overview of assay considerations used in the study.
1.6 Biacore™ assay setup considerations
To fully characterize the target:PROTAC:ligase interaction, scientists need to monitor the binary interactions (target:PROTAC or ligase:PROTAC) and ternary complex formation. Either the target or the ligase can be captured or covalently coupled to the sensor surface, but some factors can influence the choice of protein to immobilize.[8] For example, immobilizing a single ligase enables researchers to screen many PROTAC-target related protein combinations and assess target specificity within a single assay setup. Alternatively, tags on the protein target such as AviTag™ can make the capture of the protein more attractive.
We highly recommend using Sensor Chip SA to capture biotinylated ligases and targets – both for easy assay design and for maintaining a stable surface when measuring slow dissociation rate constants and half-lives. If one protein binds nonspecifically to the sensor surface and prevents its use as an analyte, you can immobilize this protein on the sensor surface, and reverse the assay. In this respect, scientists can tailor the assay setup to the specific proteins of interest.
You can set up Biacore™ binding experiments between the target protein and PROTAC in a similar way to other small molecule SPR experiments.[9,10] However, some additional factors need to be considered when designing ternary SPR binding experiments:
- Analyte protein concentration: Ternary complex kinetic experiments are set up with an excess of target protein relative to the PROTAC in the binding buffer. We recommend near-saturating concentrations – ideally 20-50 times greater than binary KD. This setup helps to overcome the well-recognized “hook-effect”, where binary PROTAC interactions out-compete ternary complex formation at high concentrations of PROTAC.[11]
- Single-cycle kinetics (SCK) vs multi-cycle kinetics: Late-stage PROTAC projects can involve ternary complexes with extremely long half lives in the hundreds, or thousands of seconds.[12] SCK can substantially reduce the experimental runtime and increase throughput, since only one complete dissociation phase is measured.
- Data analysis: Rmax indicates whether a binary or ternary complex is formed (Rmax for ternary complex formed is likely to be >10x the Rmax for binary complex, depending on the size of the PROTAC and target). Sensorgrams yield kinetic data, ka and kd, enabling scientists to calculate of KD, ΔG, and t1/2. The cooperativity factor (α) is defined as the ratio of binary and ternary dissociation constants (α = KDbinary/KDternary).
Materials and methods
1.7 Protein constructs
Wild-type and mutant BET BDs Brd2BD1, Brd2BD2, Brd3BD1, Brd3BD2, Brd4BD1 and Brd4BD2, as well as a VHL-elonginB/C-Avi tag E3 ligase construct referred to as VCB, were expressed and purified as described by Roy et al. 2019. The PROTAC MZ1 was provided by the Boehringer Ingelheim opnMe portal.
1.8 Surface preparation
A Series S Sensor Chip SA, was equilibrated in VHL running buffer containing 20 mM HEPES pH 7.0, 150 mM NaCl, 1 mM TCEP, 0.005 % TWEEN 20, and 1 % DMSO. Using the automated wizard in Biacore™ T200 Control Software, biotinylated VCB (100 nM) was captured to 900 RU (flow rate 10 μL/min).
1.9 Ternary complex direct binding assay
Initially, PROTACs (10 mM in 100% DMSO) were prepared at 1 μM or 200 nM in VHL running buffer with a concentration of 2% DMSO. This solution was mixed 1:1 with a solution of 50 μM of the corresponding bromodomain target protein in VHL running buffer without DMSO, to prepare a final solution (300 μL) of 0.5 μM PROTAC and 25 μM bromodomain in VHL running buffer containing 1% DMSO. This complex was serially diluted in VHL running buffer containing 2 μM bromodomain and 1% DMSO (five-point five-fold serial dilution, 500 nM - 0.8 nM final concentration of PROTAC, 25 μM - 2 μM final concentration of bromodomain). To ensure nearly all PROTAC molecules (95%-98%) remained in a bromodomain-bound state throughout the ternary complex binding experiments, a minimum target bromodomain protein concentration of 2 μM was maintained throughout the experiments.
For ternary experiments, solutions were injected from low to high concentration in single-cycle kinetic format without regeneration (four replicate series per experimental repeat, contact time 180 sec, flow rate 100 μL/min, dissociation time 1000 sec) using a stabilization period of 30 sec and extra wash (50% DMSO) between injections. For use in blank subtractions, at least two series of blank injections (VHL running buffer containing 2 μM bromodomain and 1% DMSO) were collected for all single-cycle experiments. All SPR measurements were performed at 12 °C.
1.10 Data analysis
The scientists analyzed the data using Biacore™ Insight Evaluation Software. Sensorgrams were reference and blank-subtracted and a solvent-correction was applied prior to analysis. To calculate the association rate (kon), dissociation rate (koff), and equilibrium dissociation constant (KD), the scientists fitted a 1:1 Langmuir interaction model to the data from all binary (multi-cycle) and ternary (single-cycle) experiments, with a term for mass-transport included. The software automatically calculates t1/2 (ln(2)/koff).
Results
SPR experiments were performed in both binary and ternary formats for MZ1 in the absence and presence of each individual BET protein BD (Fig 2) – and the resulting KD values were in agreement with the data previously obtained with ITC.[7] This demonstrates that the affinity data generated using Biacore™ systems agrees with in-solution methods – however, the assay provides significant throughput advantages and enables kinetic characterization of ternary complex formation, which is not possible using ITC.
Fig 2. Schematic showing example assay set-up for binary vs ternary complex direct binding assays for MZ1 Firstly, VHL-EloBC (VCB) is captured on the sensor chip. The left-hand side shows a direct binding assay format for PROTAC alone, with representative MCK data (concentration series of MZ1 from 1.6 – 1000 nM in duplicate. The right hand side shows a schematic of PROTAC binding VHL-EloBC in the presence of the target protein, with a representative SCK sensorgram of 0.8 nM – 500 nM MZ1 in the presence of Brd4BD2
MZ1 degrades Brd4 more than Brd2 and Brd3, despite highly comparable binary binding affinity to all BET bromodomains – suggesting PROTAC-induced cooperativity might play a role. Based on this, the scientists used a ternary complex Biacore™ assay to compare the cooperativity and binding kinetics of MZ1 to VCB in the presence and absence of bromodomains BD1 and BD2 for each BET protein Brd2, Brd3, and Brd4, as shown in Figure 3.
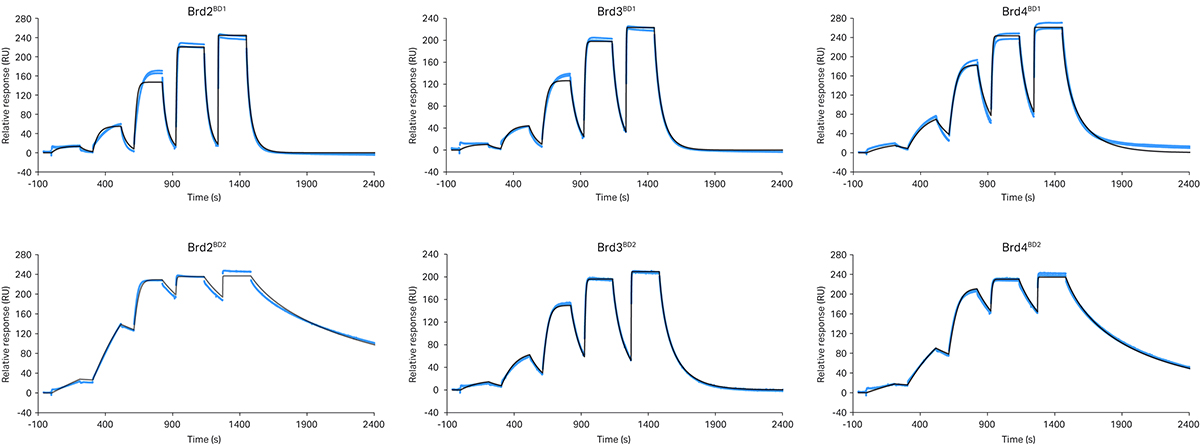
Fig 3. SPR sensorgrams for different MZ1/BD complexes reveal marked differences in binding kinetics. VCB is captured on a sensor surface and MZ1 injected from 0.8 – 500 nM using SCK. A 1:1 binding model is fit to the data with kinetics rate constants presented in table 2. In particular VHL/MZ1/Brd2BD2 and VHL/MZ1/Brd4BD2 ternary complexes dissociated relatively slowly (as a result of the high positive cooperativity, α, and greater complex stability).
The data revealed fast dissociation of MZ1 from VCB in the presence of all BD1 domains, with low positive cooperativity (α = 2.0 - 3.6). In contrast, all BD2 domains exhibit slower dissociation (up to 800 times slower), higher cooperativity (α = 3.9 - 48) and high complex stability in the ternary complex binding experiments. In particular, the t1/2 of the VCB/MZ1/BD complex was significantly longer for Brd2BD2 and Brd4BD2 (t1/2 = 400 and 220 s respectively) relative to Brd3BD2 (t1/2 = 40 s). The correlation between MZ1 forming longer lived ternary complexes with Brd2BD2 and Brd4BD2 relative to Brd3BD2, and the faster MZ1-induced initial degradation rate of Brd2BD2 and Brd4BD2 support the notion that ternary complex stability might be a key driver of PROTAC activity. [7]
By overlaying a ternary complex crystal structure of VCB/MZ1/Brd4BD2 with Brd3BD2, Roy et al identified a single amino acid residue within the ZA loop of Brd3BD2 (Glu344, which corresponds to the positions Gly382 in Brd2BD2 and Gly386 in Brd4BD2) that appeared to be responsible for the differences in cooperativities observed for the different bromodomains.[7] The substitution of Glu344 was proposed to induce a severe steric clash with the VHL/MZ1 portion of the complex, leading to destabilization.
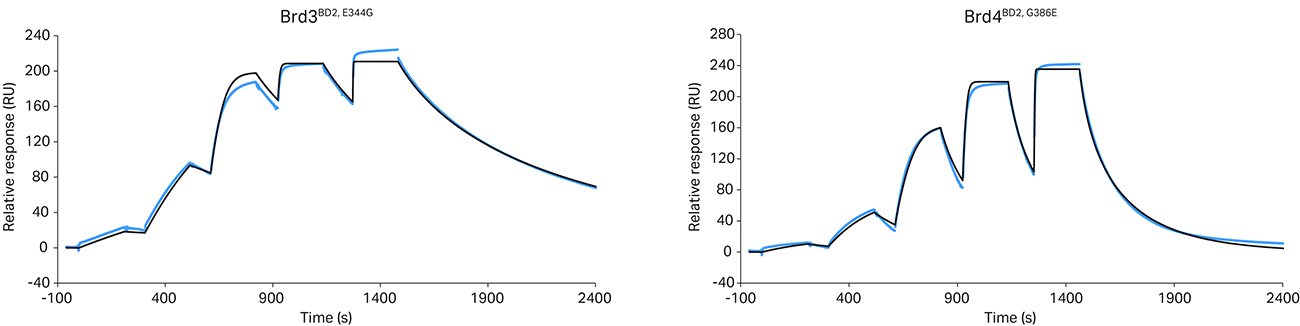
For this reason, using the previously described Biacore™ assay, ternary complex kinetics for Brd4BD2 and Brd3BD2 with G-to-E and E-to-G target mutations (Brd4BD2,G386E and Brd3BD2,E344G) (Fig 4) were measured.
Fig 4. Ternary complex formation is influenced by a single amino acid substitution. The crystal structure of the VCB/MZ1/Brd4BD2 ternary complex (PDB: 5T35) suggested that a VHL/MZ1/Brd3BD2 ternary complex adopting the equivalent close-packing interaction would likely be less stable due to steric clash with the VHL/MZ1 of a single amino acid within the ZA loop of the bromodomain (Glu344 of Brd3BD2, which corresponds to Gly386 in Brd4BD2). Reciprocal exchange of this single Gly/Glu residue in Brd3BD2 (a) or Brd4BD2 (b) yields a corresponding swap of the kinetic profile in the resulting VHL/MZ1/BD ternary complex SPR sensorgram. Kinetic parameters are reported in table 2.
The data revealed that the G-to-E mutation in Brd4BD2 shortened the ternary complex t1/2, decreasing cooperativity and complex stability. However, the reverse mutation in Brd3BD2 extended the ternary complex t1/2 to more closely resemble the kinetic profile for Brd4BD2. The data was also cross validated using a competitive fluorescence polarization assay, confirming good correlation between KD and KI values and cooperativity.
Table 2. Kinetic parameters of the ternary complex formation study
| Bromodomain | ka(×106 M-1s-1) | kd (×10-2 s-1) | KD(nM) | t1/2 (s) |
| BRD2-BD1 | 5.0 (± 0.07) | 7.1 (± 0.01) | 14 | 9.7 |
| BRD3-BD1 | 1.6 (± 0.004) | 2.6 (± 0.006) | 17 | 27 |
| BRD4-BD1 | 2.8 (± 0.03) | 2.6 (± 0.02) | 9.2 | 27 |
| BRD2-BD2 | 2.5 (± 0.01) | 0.17 (± 0.0004) | 0.69 | 410 |
| BRD3-BD2 | 2.0 (± 0.01) | 1.7 (± 0.007) | 8.5 | 41 |
| BRD4-BD2 | 1.4 (± 0.002) | 0.32 (± 0.0003) | 2.2 | 220 |
| BRD3-2_E344G | 1.6 (± 0.002) | 0.20 (± 0.0001)) | 1.3 | 340 |
| BRD4-2_G386E | 1.3 (± 0.01) | 1.2 (± 0.004) | 9.4 | 56 |
The size of the analyte (PROTAC + protein) is much greater in ternary SPR binding experiments than in binary SPR binding experiments (PROTAC alone). The data presented here provides positive confirmation of ternary complex formation, given the high Rmax relative to the binary experiments (Fig 2). While both binary and ternary complex data can be measured using the same protein immobilization, a separate lower density surface for ternary complex experiments is recommended to overcome mass transport limitation and reduce other experimental artefacts that can be problematic with a higher density surface.
Discussion
We describe a simple and robust method to characterize the kinetics of target protein:PROTAC:ligase ternary complex formation. Attachment of an E3 ligase to the sensor surface enables scientists to rapidly measure multiple PROTAC:target combinations, agnostic of the target protein. In theory, researchers can apply this assay format to any PROTAC:target system and directly transfer it to other SPR systems such as Biacore™ X100. However, the format can also be reversed in cases where target protein is in short supply, or to capture a tagged protein directly from cell lysates and minimize assay development. The KD values obtained from this study compare well to those obtained orthogonally by techniques such as ITC and indirect KI measurements from FP assays. Critically, this Biacore™ assay enables scientists to determine kinetic rate constants and t1/2, with higher throughput compared to other direct binding assays. The data reveals that a single amino acid substitution can significantly affect the kinetics, cooperativity, and t1/2 of ternary complex formation. Together, the data highlights the importance of understanding the lifetime of the ternary complex to predict efficient target degradation, analogous to understanding drug-target residence time for traditional small molecule inhibitor drug discovery.
A clear future progression of the work is to incorporate the assay earlier in the PROTAC drug discovery process. An early understanding of ternary KD, t1/2 and cooperativity in the PROTAC hit identification and optimization stages can allow selection of those that induce desirable stabilizations of the ternary complex, potentially leading to more efficient target degradation, and ultimately more efficacious drugs. In late-stage projects, ternary complexes can display incredibly high t1/2s. Therefore, it can be helpful to have the option to run several single-cycle kinetic experiments in parallel on a multineedle instrument such as a Biacore™ 8K or Biacore™ 8K+ system. In addition to PROTACs, the field can more widely employ the assay format to characterize other three-partner binding equilibria induced by other hetero-bivalent molecules, such as lysosome target chimeras (LYTACS) or bispecific antibodies.
Cytiva would like to thank Professor Alessio Ciulli and Dr David Zollman, University of Dundee for sharing their data for the publication of this article.
References
- W. Farnaby, M. Koegl, M. J. Roy, C. Whitworth, E. Diers, N. Trainor, D. Zollman, S. Steurer, J. Karolyi-Oezguer, C. Riedmueller, T. Gmaschitz, J. Wachter, C. Dank, M. Galant, B. Sharps, K. Rumpel, E. Traxler, T. Gerstberger, R. Schnitzer, O. Petermann, P. Greb, H. Weinstabl, G. Bader, A. Zoephel, A. Weiss-Puxbaum, K. Ehrenhöfer-Wölfer, S. Wöhrle, G. Boehmelt, J. Rinnenthal, H. Arnhof, N. Wiechens, M.-Y. Wu, T. Owen-Hughes, P. Ettmayer, M. Pearson, D. B. McConnell and A. Ciulli, Nat Chem Biol, 2019, 15, 672–680.
- Q. L. Sievers, G. Petzold, R. D. Bunker, A. Renneville, M. Słabicki, B. J. Liddicoat, W. Abdulrahman, T. Mikkelsen, B. L. Ebert and N. H. Thomä, Science, 2018, 362, eaat0572.
- M. E. Matyskiela, W. Zhang, H. W. Man, G. Muller, G. Khambatta, F. Baculi, M. Hickman, L. LeBrun, B. Pagarigan, G. Carmel, C.-C. Lu, G. Lu, M. Riley, Y. Satoh, P. Schafer, T. O. Daniel, J. Carmichael, B. E. Cathers and P. P. Chamberlain, J. Med. Chem., 2018, 61, 535–542.
- M. S. Gadd, A. Testa, X. Lucas, K.-H. Chan, W. Chen, D. J. Lamont, M. Zengerle and A. Ciulli, Nat Chem Biol, 2017, 13, 514–521.
- J. Frost, C. Galdeano, P. Soares, M. S. Gadd, K. M. Grzes, L. Ellis, O. Epemolu, S. Shimamura, M. Bantscheff, P. Grandi, K. D. Read, D. A. Cantrell, S. Rocha and A. Ciulli “Potent and selective chemical probe of hypoxic signalling downstream of HIF-α hydroxylation via VHL inhibition. Nat Commun 7, 13312 (2016)
- P. Filippakopoulos, J. Qi, S. Picaud, Y. Shen, W. B. Smith, O. Fedorov, E. M. Morse, T. Keates, T. T. Hickman, I. Felletar, M. Philpott, S. Munro, M. R. McKeown, Y. Wang, A. L. Christie, N. West, M. J. Cameron, B. Schwartz, T. D. Heightman, N. La Thangue, C. A. French, O. Wiest, A. L. Kung, S. Knapp and J. E. Bradner, (December 2010). "Selective inhibition of BET bromodomains". Nature. 468 (7327): 1067–73.
- M. J. Roy, S. Winkler, S. J. Hughes, C. Whitworth, M. Galant, W. Farnaby, K. Rumpel and A. Ciulli, ACS Chem Biol, 2019, 14, 361–368.
- Biacore™ Sensor Surface Handbook
- Fragment and small molecule screening
- Kinetics and affinity measurements
- E. T. Mack, R. Perez- Castillejos, Z. Suo and G. M. Whitesides, 2008, 80,5550– 5555.
- T. H. Pillow, P. Adhikari, R. A. Blake, J. Chen, G. Del Rosario, G. Deshmukh, I. Figueroa, K. E. Gascoigne, A. V. Kamath, S. Kaufman, T. Kleinheinz, K. R. Kozak, B. Latifi, D. D. Leipold, C. Sing Li, R. Li, M. M. Mulvihill, A. O'Donohue, R. K. Rowntree, J. D. Sadowsky, J. Wai, X. Wang, C. Wu, Z. Xu, H. Yao, S.-F. Yu, D. Zhang, R. Zang, H. Zhang, H. Zhou, X. Zhu and P. S. Dragovich, ChemMedChem, 2020, 15, 17–25.