FAQ
How do I store my column?
Prepacked columns
Please store the column according to instructions.
Other columns
Please follow the recommendations in the media instructions.
What chemicals are compatible with my column?
Please see the chemical stability in the instructions for prepacked columns, empty columns and media.
My column has run dry. Can I reuse the column?
If the column still is free from bacterial growth it might be possible to reuse the column. It could be worth trying.
Remove the air by running liquid slowly from bottom to top of a vertical placed column at a pressure close to maximum column pressure. The size of air bubbles decreases with increased pressure and facilitate the bubbles escape from the column.
If you don't succeed in removing air from the column, the column must be replaced (prepacked columns) or repacked.
How should I prepare my sample prior to loading the column?
Please prepare the sample according to instructions and handbooks
Prepacked column instructions
Please refer to relevant Instruction.
Other columns
Please refer to relevant Handbook.
My column is clogged and/or discoloured. How can I clean my packed column?
Prepacked columns
Please clean the column according to instructions.
Other columns
Please follow the recommendations in the media instructions.
If the issue persists after cleaning you have to replace the column with a new one (prepacked columns) or repack the column with fresh medium.
How much sample, in mg or ml of protein can I load onto my column?
Affinity Chromatography
- The binding capacity values listed in the selection guide below, are typical for the given species. However, there might be considerable deviations in binding capacity for different immunoglobulins derivated from same species, even if they are of the same subclass.
- Protein binding capacity is protein-to-protein dependent. The given capacities are to be considered as starting values.
- For optimal separation use approximately one fifth of the total binding capacity
For more information refer to:
Affinity chromatography columns and media selection guide
Glutathione Sepharose selection guide
Ni Sepharose and IMAC Sepharose selection guide
Desalting
- In group separation (desalting) the sample volumes can be up to 30% of the column volume.
- Regarding sample volumes for prepacked columns, please refer to Sample preparation for analysis of proteins, peptides and carbohydrates selection guide.
Gel filtration chromatography
- Choose a sample volume of < 0.5% of the column volume for media with average particle size in the range of 10-15 µm and < 5% of the volume for media with average particle size in the range of 30-100 µm.
- In group separation (desalting) the sample volumes can be up to 30% of the column volume.
- Regarding sample volumes for prepacked columns, please refer to selection guide below:
Sample preparation for analysis of proteins, peptides and carbohydrates selection guide.
Gel filtration columns and media selection guide and product profile selection guide.
Hydrophobic interaction chromatography
- The binding capacity values in the media and column instructions, there are examples and to be considered as starting values. Protein binding capacity is protein-to-protein dependent.
- For optimal separation use approximately one fifth of the total binding capacity.
Ion exchange chromatography
- The binding capacity values in the Ion exchange columns and media product profile, please refer to Ion exchange columns and media product profile selection guide, there are examples and to be considered as starting values. Protein binding capacity is protein-to-protein dependent.
- For optimal separation use approximately one fifth of the total binding capacity.
Reversed phase chromatography
- The binding capacity values in the media and column instructions, please refer to the related document, there are examples and to be considered as starting values. Protein binding capacity is protein-to-protein dependent.
- For optimal separation use approximately one fifth of the total binding capacity.
Can I run two columns in series to increase resolution or capacity?
Gel filtration chromatography
Running columns in series increases the resolution
Affinity chromatography, Hydrophobic interaction, Ion exchange chromatography
Reverse phase chromatography
Running columns in series increases the capacity but may have a bad impact on the resolution
due to increased dead volume.
Please note that back pressure may increase.
What is the pressure limit of my column?
Prepacked Columns
Please read
Prepacked chromatography columns for ÄKTAdesign systems
Other Columns
| Column |
Maximum pressure (bar)
|
| C 10, C 16 and C 26 |
1
|
| FineLINE Pilot 35 |
20
|
| HiScale |
20
|
| HR (High Resolution) 16 |
30
|
| SR 25 |
1
|
| Tricorn 5 |
100
|
| Tricorn 10 |
50
|
| XK 16 and XK 26 |
5
|
| XK 50 |
3
|
My column is leaking buffer, what shall I do?
Leakage around connectors
| Possible causes | Suggested Remedy |
| Connectors not compatible with each other. | Check compatibility. |
| Connectors not compatible with solvents. | Check chemical resistance with the connector supplier. |
| Connectors poorly positioned or not tightened. | Check the connectors. |
| Gaskets worn out. | Gaskets lose flexibility with time and need to be replaced regularly. Inspect and replace if necessary and at least annually. |
Leaking tubing
| Possible causes | Suggested Remedy |
| Tubing not compatible with solvents. |
Check chemical resistance with the tubing supplier. Preventive action: Always check tubing solvent compatibility prior to packing or running the column. |
Leakage around end-pieces ( Not applicable for the pre-packed columns )
| Possible causes | Suggested Remedy |
| End-piece and O-rings not properly positioned with respect to the tube. | Disassemble the column and check the position of the end-piece and O-rings. Assemble the column according to instructions and perform leakage tests. |
| O-rings worn out | O-rings loose their flexibility with time and need to be replaced regularly. Disassemble the column and inspect the O-rings. Replace if necessary and at least annually. Assemble the column according to instructions and test for leakage. Preventive action: Replace O-rings when needed or at least annually. |
How can I check the functionality of my column?
Leading and tailing peaks as defined according to the following figures:


| Leading peak | Tailing peak |
Experience shows that the best method of expressing the efficiency of a packed column is in terms of its height equivalent to a theoretical plate (HETP), reduced plate number (h) and its peak asymmetry factor (As).
Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values.
How would I get the best resolution of my column?
For stand alone columns please assemble the monitor to the column outlet.
If the column is connected to a system, connect the column as close as possible to the monitor.
My column has a gap between the packed bed and adaptor. Can I reuse the column?
| Possible causes | Suggested Remedy |
| Bed support damaged or incorrectly assembled allowing chromatography medium particles to leave the column. | Check the bed support and replace if necessary. Disassemble the column according to instructions. |
| Buffer conditions deviate with regard to temperature, conductivity, viscosity, content of organic solvent (reduces surface tension) or other factor. | Check the buffers and choose more suitable conditions. |
| Increased resistance to flow due to blocked bed support compressing the packed bed. | Clean or change the bed support. Disassemble the column and replace the support according to instructions. Preventive action: Pre-filter or centrifuge sample to avoid residues building up. |
| Poorly packed bed (not sufficiently compressed during packing). (Not applicable for prepacked columns) |
Evaluate the packing using recommended methods. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. If the results are poor, refer to the symptom Poor packing evaluation in the troubleshooting section. |
The backpressure increases during operation
| Possible causes | Suggested Remedy |
| Auxiliary equipment such as manometers and pumps not working properly. | Check the function of all auxiliary equipment. Repair/replace if necessary. |
| Column is clogged. | Clean the column according to instructions. Choose the more rigorous cleaning protocol when available. See media and column instructions. |
| Bent tubing. | Check that the flow path is not restricted. |
| Buffer viscosity too high. | Check the viscosity of all buffers. Viscosity is a function of temperature. (Lower temperature gives higher viscosity.) Let low-temperature buffer reach operating temperature before starting the run. |
| Microbial growth in buffers. The buffer normally become opalescent due to microbial growth. |
Check buffers, especially those with phosphate, for microbial growth. Replace with fresh buffer if necessary. |
| Sample and collection vessels at different levels. | Adjust the vessels to approximately the same level. |
| The prefilter might be blocked. | Check the prefilter. Preventive action: Prefilters are not meant to substitute sample treatment. |
| Valve not fully open. | Check all valves. Open any that is not fully open. |
Find more causes and remedies in the troubleshooting section.
My column contains air, what can I do?
| Possible causes | Suggested Remedy |
| Unclarified lysates may cause increased air bubble formation during purification. | An attached flow restrictor in the chromatography system can prevent this. If a flow restrictor is attached, it is important to change the pressure limit to 0.5 MPa (5 bar9 on the ÄKTAdesign system (where the column and the flow restrictor give a pressure of 0.3 MPa and 0.2 MPa respectively). |
| The column operates at room temperature after having been stored in a cold room. | Allow thermal equilibration before use. |
| - | Reverse the flow direction and pump well degassed water through the column. For recommended volumes and flow rates for your specific column, please refer to media and column instructions. |
More causes and remedies might be presented in the troubleshooting section.
Accessories
Make sure you have all connectors and tubing needed for packing and running the column.
Depending on your system configuration and column length, you may need extra long
tubing to connect your column.
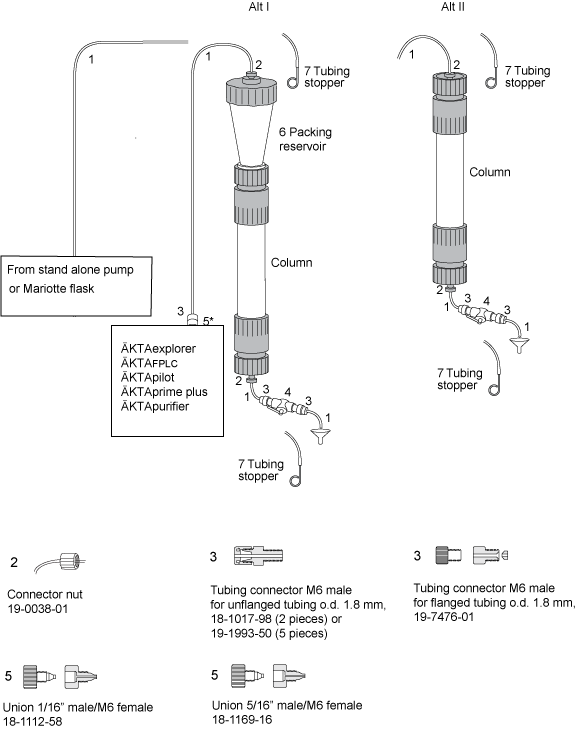
* Connect M6 male, to 5/16" (18-1169-16) for ÄKTApilot and 1/16" (18-1112-58) for ÄKTA lab systems.
| Gelfiltration chromatography movies | |
|
Exchange of buffer |
Preparing the column |
|
Packing the column |
Unpacking the columns |
| Adsorption chromatography movies | |
|
Equipment required |
Exchange of buffer |
|
Preparing the column |
Packing the column |
|
Unpacking the columns |
|
| Column evaluation movie | |
|
Column evaluation |
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 1 | Tubing cutter, for PEEK, EFTE, and FEP tubing i.d. 0.25, 0.5, 0.75, 1 and 1.6 mm | 18111246 | 99.55 USD |
Add to cart
|
| 1 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
| 3 | Tubing Connector Flangeless/M6 Male | 18101798 | 79.78 USD |
Add to cart
|
| 3 | Tubing connector flangeless-M6 male | 19199350 | 82.29 USD |
Add to cart
|
| 4 | Valve SRV-1 | 19214501 | 288.00 USD |
Add to cart
|
| 5 | Fingertight Union 1/16" Male/M6 Female | 18111258 | 135.19 USD |
Add to cart
|
| 5 | Union, M6 Female - 5/16" Male | 18116916 | 125.23 USD |
Add to cart
|
| 6 | RC 16 Packing Reservoir | 19511001 | 216.27 USD |
Add to cart
|
Make sure you have all connectors and tubing needed for running the column.
Depending on your system configuration and column length you may need extra long tubing to connect your column.
Please do not extend the tubing unnecessarily which results in a lower resolution.
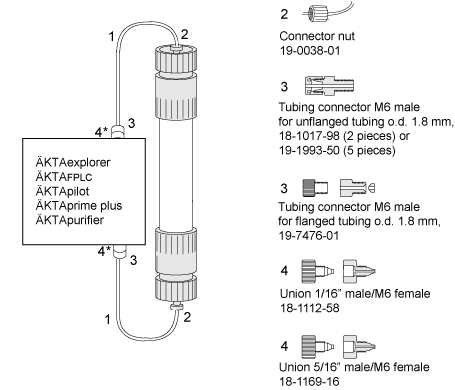
* Connect M6 male, to 5/16" (18-1169-16) for ÄKTApilot and 1/16" (18-1112-58) for ÄKTA lab systems.
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 1 | Tubing cutter, for PEEK, EFTE, and FEP tubing i.d. 0.25, 0.5, 0.75, 1 and 1.6 mm | 18111246 | 99.55 USD |
Add to cart
|
| 1 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
| 3 | Tubing Connector Flangeless/M6 Male | 18101798 | 79.78 USD |
Add to cart
|
| 3 | Tubing connector flangeless-M6 male | 19199350 | 82.29 USD |
Add to cart
|
| 4 | Fingertight Union 1/16" Male/M6 Female | 18111258 | 135.19 USD |
Add to cart
|
| 4 | Union, M6 Female - 5/16" Male | 18116916 | 125.23 USD |
Add to cart
|
Running C columns with a stand alone pump, for instance using HiLoad pump P-50 for step elution.
The figure below shows a complete set up which facilitates bypassing the column, running cleaning-in-place etc.
Make sure you have all connectors and tubing needed for running the column.
Depending on your system configuration and column length you may need extra long tubing to connect your column.
Please do not extend the tubing unnecessarily which results in a lower resolution.
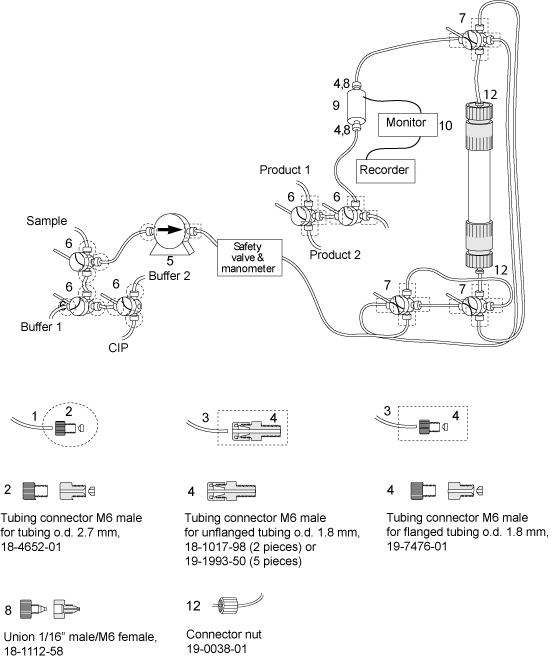
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 1 | Tubing cutter, for PEEK, EFTE, and FEP tubing i.d. 0.25, 0.5, 0.75, 1 and 1.6 mm | 18111246 | 99.55 USD |
Add to cart
|
| 1 | Unflanged tubing i.d. 1.9 mm, o.d. 2.7 mm | 18820702 | 338.00 USD |
Add to cart
|
| 2 | Tubing connectors o.d. 2.7 mm, M6 male | 18465201 | 129.93 USD |
Add to cart
|
| 3 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
| 4 | Tubing Connector Flangeless/M6 Male | 18101798 | 79.78 USD |
Add to cart
|
| 4 | Tubing connector flangeless-M6 male | 19199350 | 82.29 USD |
Add to cart
|
| 5 | Pre-filter 6000 | 18458201 | 1,367.00 USD |
Add to cart
|
| 5 | Filter kit | 18458401 | 156.83 USD |
Add to cart
|
| 5 | Holder Valve PSV-50 | 19199255 | 153.64 USD |
Add to cart
|
| 5 | Solenoid Valve PSV-50 | 19199401 | 819.00 USD |
Add to cart
|
| 8 | Fingertight Union 1/16" Male/M6 Female | 18111258 | 135.19 USD |
Add to cart
|
| 9 | UV Flow Cell, 2 mm | 18111110 | 2,857.00 USD |
Add to cart
|
| 10 | Filter 215 nm | 11000733 | 808.00 USD |
Add to cart
|
| 10 | Filter 260 nm | 11000734 | 781.00 USD |
Add to cart
|
| 10 | Filter 280 nm | 11000735 | 628.00 USD |
Add to cart
|
| 10 | Filter 405 nm | 11000736 | 760.00 USD |
Add to cart
|
| 10 | Empty Filter Holder | 11000738 | 248.00 USD |
Add to cart
|
| 10 | Monitor UVis-920 Excluding Filter and Flow Cell | 11000754 | 10,200.00 USD |
Add to cart
|
| 10 | Short optical fibre kit, 200 mm | 18113485 | 970.00 USD |
Add to cart
|
| 10 | Long optical fibre kit, 500 mm | 18113486 | 716.00 USD |
Add to cart
|
| 10 | UV flow cell, i.d. 8 mm | 28966687 | 3,723.00 USD |
Add to cart
|
| 10 | O-ring kit | 28969705 | 162.00 USD |
Add to cart
|
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| AC 16 Flow Adapter | 19510901 | 257.27 USD |
Add to cart
|
Spare parts
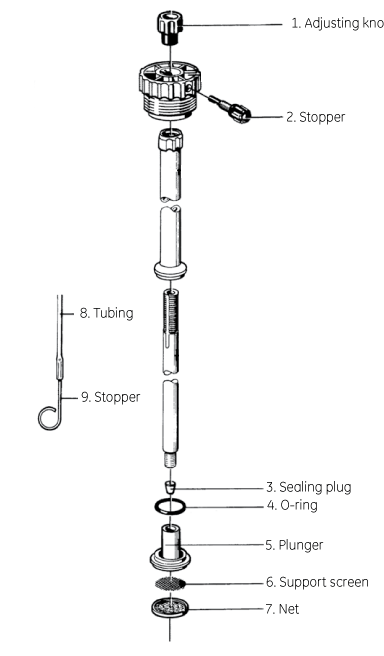
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 1 | Adjusting knob | 19522701 | 77.07 USD |
Add to cart
|
| 3 | Sealing Plug | 19003901 | 77.62 USD |
Add to cart
|
| 4 | O-Ring, 10.3 × 2.4 mm | 19016301 | 60.47 USD |
Add to cart
|
| 5 | Plunger for XK 16 and AK 16 | 18103180 | 55.35 USD |
Add to cart
|
| 6 | Support Screen for Column, i.d. 16 mm | 19065101 | 52.28 USD |
Add to cart
|
| 7 | Filter Tool | 18115320 | 166.05 USD |
Add to cart
|
| 7 | Net for Columns, i.d 16 mm, 10 µm | 18876101 | 76.88 USD |
Add to cart
|
| 8 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 1 | Net for Columns, i.d 16 mm, 10 µm | 18876101 | 76.88 USD |
Add to cart
|
| 2 | Support Screen for Column, i.d. 16 mm | 19065101 | 52.28 USD |
Add to cart
|
| 3 | O-ring 6.1 x 1.6 mm | 19068001 | 25.18 USD |
Add to cart
|
| 5 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
C 16 Column
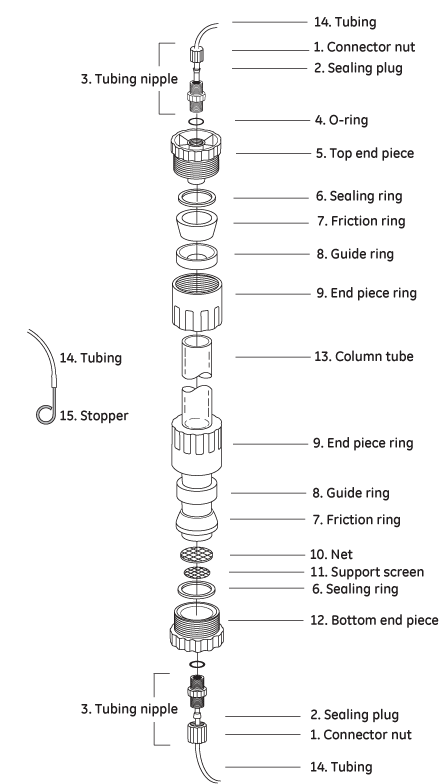
| # | Product Name | Product Code | Price | |
|---|---|---|---|---|
| 2 | Sealing Plug | 19003901 | 77.62 USD |
Add to cart
|
| 3 | Tubing nipple complete | 19003501 | 94.04 USD |
Add to cart
|
| 4 | O-ring 6.1 x 1.6 mm | 19068001 | 25.18 USD |
Add to cart
|
| 10 | Filter Tool | 18115320 | 166.05 USD |
Add to cart
|
| 10 | Net for Columns, i.d 16 mm, 10 µm | 18876101 | 76.88 USD |
Add to cart
|
| 11 | Support Screen for Column, i.d. 16 mm | 19065101 | 52.28 USD |
Add to cart
|
| 14 | Tubing, 10 m, i.d. 1.0 mm, o.d. 1.8 mm | 19004001 | 84.87 USD |
Add to cart
|
Troubleshooting
Find solutions to product related issues. For unlisted issues please contact local Cytiva service representation.
Poor product recovery
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Activity of protein is not compatible with solution A and has been irreversibly denatured. |
Choose another solution system after testing the salt and pH stability of the protein. |
Conditions that are sufficiently effective to elute molecules along the column may irreversibly denature both desorbed substances and the ligand. |
Elution should always be carried out in the reverse direction to sample application. Reverse the flow direction through the column by installing it upside down during elution. |
Hydrophobic interaction chromatography - Protein is probably denatured by high salt and cannot refold properly upon elution with low salt solution to give active protein. |
Try the protein at different salt concentrations and see when activity is lost. Then enter the salt stability limit in Case Data and start a New Case. If this is not successful choose another technique. |
Protein probably can not withstand the low pH of the elution solution. |
Adjust eluted fractions immediately to a higher pH. If this does not help choose another technique. |
Protein is sensitive to the speed of neutralization and is partially denatured. |
Use a weaker neutralizing solution of lower pH. |
Unstable protein at the pH used for chromatography. |
Determine the pH stability profile and start a New Case, or choose another technique. |
| Possible cause | Suggested remedy |
|---|---|
Lipoprotein may be unstable under the conditions used. |
Do not use chaotropic salts if the native structure is to be preserved. Ordinary salts like sodium chloride, sodium sulfate, etc., should be employed. |
| Possible cause | Suggested remedy |
|---|---|
At sufficiently high ionic strength some proteins may be precipitated from solution. One factor is that the high concentration of salt may remove water of hydration from proteins. |
Your protein may be applied at a lower ionic strength than that at which the column is equilibrated provided that precipitation is reversible and that the sample volume is kept to a minimum, 5% to 20% Vt.. |
| Possible cause | Suggested remedy |
|---|---|
Unstable protein at the chosen ionic strength and/or pH resulting in inactivated. |
Try changing the ionic strength and/or pH up or down. |
| Possible cause | Suggested remedy |
|---|---|
Different assay conditions used before and after chromatography. |
Use the same conditions for all assays. |
Inhibiting component removed during purification. |
Happens some times. Take into consideration and try to avoid. |
Target molecules transferred from previous run due to poor cleaning between runs. |
Evaluate your cleaning procedure |
| Possible cause | Suggested remedy |
|---|---|
Hydrophobicity of your protein is higher than your current estimate and the lower solubility of hydrophobic proteins can cause decreased recovery. |
Use chaotropic salts for elution to increase the recovery of hydrophobic proteins (their lower salting out effect increases sample solubility). However, they might break down protein structure. |
Hydrophobicity of your protein is higher than your current estimate and binds more strongly to the chromatography medium than you have expected. |
Remedy 1. Add detergents to solution B. Detergents work as displacers of the proteins. |
Ionic strength of solution B may have caused the protein to precipitate onto the column. Some proteins tend to precipitate at high salt concentration due to increased hydrophobic interactions. |
Use chaotropic salts in the eluting solution. |
Lower solubility of hydrophobic proteins can cause decreased recovery. |
Remedy 1. Add detergents to solution B. Detergents work as displacers of the proteins. |
Precipitation of protein on the column may have occurred. Some proteins tend to precipitate at high salt concentration due to increased hydrophobic interactions. |
Use chaotropic salts in the eluting solution. However, they may break down protein structure. |
Smaller proteins and polypeptides sometimes show lower solubility in salt. |
Chaotropic salts may be chosen for elution to give higher recoveries of smaller proteins. These salts allow maximum sample solubility. |
Solution A may have caused the protein to precipitate onto the column. |
Change the running conditions to be compatible with your sample (e.g. salt concentration, pH). |
Unstable protein at salt concentration used for elution. |
Change the pH of solution B away from your protein’s pI keeping the salt concentration constant. You are increasing the polarity of the protein which decreases the protein’s interaction with the chromatography medium. If this is not successful choose another technique. |
| Possible cause | Suggested remedy |
|---|---|
Protein is probably sensitive to low pH. |
Neutralize the eluted fractions. Let the protein elute into tubes containing high pH buffer |
| Possible cause | Suggested remedy |
|---|---|
Protein is too sensitive to the salt concentration used for elution and can not be recovered by increasing the ionic strength of the eluted fractions. |
Choose another technique. |
| Possible cause | Suggested remedy |
|---|---|
Binding between the protein and the medium is stronger than expected. |
Try using urea in solution B. |
| Possible cause | Suggested remedy |
|---|---|
A co-factor used to define recovery has been removed, thus causing lower activity. |
Check the integrity of the enzyme or enzyme/co-factor complex. |
Precipitation of protein on the column may have occur due to the high ionic strength of solution B. |
Try to elute the protein with an increasing pH keeping the salt concentration constant. Determine the salt stability profile and start a New Case, or choose another technique. |
Precipitation of protein on the column may have occur due to the low ionic strength or dilution. |
The decreased solubility is caused by electrostatic interactions that result in formation of aggregates. To minimize these interactions, use higher concentration of salt in solution A and sample solution. |
Precipitation of protein on the column and not dissolved by decreasing the ionic strength. |
Combine the decreasing of ionic strength with an increasing ethylene glycol gradient. If this is not successful choose another technique. |
Protein precipiated on the column. |
If the protein is initially solubilized with special additives, ensure that these are present during chromatography. Also check pH and ionic strength. |
Protein may exhibit some hydrophobic interactions with the matrix. |
If possible decrease the ionic strength, increase pH or introduce an organic (e.g. 24% propanol or 10% ethylene glycol) in solution A in order to minimize hydrophobic interactions. |
Too low salt concentration in the elution buffer. |
Check and adjust elution buffer composition. |
Column not equilibrated properly. |
Check the pH and the conductivity of the effluent before applying the sample to make sure the column is properly equilibrated. Continue to equilibrate with start buffer if necessary. |
Column over-loaded. |
Decrease the amount of sample or increase the amount of chromatography medium. I.e. increase the margin of safety. We recommend a margin of safety of approximately 10 %. |
For ion exchange chromatography - Ionic strength of the sample too high or pH incorrect. |
Check and adjust the ionic strength and/or pH. Calibrate your conductivity and pH meters. |
Incorrect buffer pH. |
Check and adjust the pH. Calibrate your pH meter. |
Ionic interactions between the protein and the matrix may cause this behavior. |
These effects can be eliminated by the use of solutions with an ionic strength exceeding 0.15 to 0.3 M. |
Low ionic strength of start or eluting buffer may have caused precipitation of the protein on the column. |
The decreased solubility is caused by electrostatic interactions that result in formation of aggregates. To minimize these interactions, use 10 to 50 mM of salt in start of eluting buffer and sample solution. |
Oppositely-charged detergents or other additivies adsorbed to the column. |
Clean the column and chromatography medium according to instructions |
Precipitation of protein on the column may have occurred. |
Change pH and/or add urea. |
| Possible cause | Suggested remedy |
|---|---|
Irreversible adsorption to matrix |
First try to modify the elution conditions. If this doesn’t help, clean and re-generate the column and chromatography medium according to instructions. |
| Possible cause | Suggested remedy |
|---|---|
Tertiary structure disrupted |
Shorten the residence time on the column. Use higher flow velocities to minimize exposure. |
Worrying peak shape
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing. |
Protein precipitated on the bed supports and/or in the bed. |
Clean and regenerate the column and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Column over-loaded |
Decrease the amount of sample or increase the amount of chromatography medium. I.e. increase the margin of safety. |
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. If the results are poor, refer to the symptom Poor packing evaluation. |
Protein precipitated on the bed supports and/or in the bed. |
Clean and regenerate the column and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. If the results are poor, refer to the symptom Poor packing evaluation. |
For reversed phase chromatography - Organic solvents have denatured the protein. |
Use a higher flow velocity and/or an adapted gradient to shorten residence time on the column and thus minimize exposure. |
Gradient slope or isocratic step too shallow. |
Increase steepness of gradient or isocratic step. |
Protein precipitated on the bed supports and/or in the bed. |
Clean and regenerate the column and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. If the results are poor, refer to the symptom Poor packing evaluation. |
Protein precipitated on the bed supports and/or in the bed. |
Clean and regenerate the column and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. If the results are poor, refer to the symptom Poor packing evaluation. |
Protein precipitated on the bed supports and/or in the bed. |
Clean and regenerate the column and chromatography medium according to instructions |
Drifting baseline
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
At a certain salt concentration, the detergent forms micelles, resulting in a sharp increase in UV absorption. |
Work above the critical micelle concentration (CMC) or change the gradient so that the increase in UV absorption does not occur while the samples are eluting. |
| Possible cause | Suggested remedy |
|---|---|
Column bleeding from previous run. |
Check and adjust your cleaning procedure. Clean the equipment and chromatography medium according to instructions. |
Contaminated salts and solutions. |
Remedy 1 |
Difference in UV-absorbance between solutions A and B. The difference in ionic strength produces a different ratio between the acid and base form of the buffer. The slope of the baseline is very sensitive to small pH differences. |
Gradient baseline drift can be adjusted by small pH changes in solution B. |
For ion exchange chromatography - Buffering capacity of buffer is too low. Dissociated ions bind to the charged groups of the chromtography medium. |
Make sure that the buffering capacity is sufficient. |
Solution not in equilibrium with column. |
Continue to flush the system until equilibrium is established. |
Unwashed buffer vessels were refilled with fresh solutions. |
Prepare new buffers. Make sure the glassware that is used to prepare and store buffers is clean. If plastic bottles are used check that plastic additives are not dissolved in the solution. |
Poor reproducibility
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Bound substances not removed during cleaning. |
Clean the chromatography medium according to instructions. |
Ligand partially degraded. |
Replace the chromatography medium. |
| Possible cause | Suggested remedy |
|---|---|
Affinity, hydrophobic interaction and ion exchange chromatography - Continuous build-up of contaminants has altered the selectivity of the chromatography medium. |
Clean the chromatography medium according to the Column and media instructions. |
Reversed phase chromatography - Strongly retained contaminants accumulate on the column and saturate active sites on the stationary phase. |
Clean and regenerate the column and chromatography medium according to instructions. |
| Possible cause | Suggested remedy |
|---|---|
Column not properly equilibrated |
Check the pH and the conductivity of th effluent before applying the sample. Continue to equilibrate with start buffer if necessary |
Insufficient column regeneration. |
Prolong the regeneration. |
| Possible cause | Suggested remedy |
|---|---|
Protein properties change with concentrations |
Dilute or concentrate the sample to minimize effects. |
Proteins precipitate at high concentration. |
Reduce sample concentration and/or binding capacity. |
| Possible cause | Suggested remedy |
|---|---|
Column bleeding from previous run. |
Check and adjust your cleaning procedure. |
Column clogged with denatured proteins and/or lipids. |
Clean and regenerate the column and chromatography medium according to the Column and media instructions. |
Incomplete equilibration of the column. |
Check pH and conductivity of the effluent before applying the sample. Continue to equilibrate if necessary. |
Incorrect pH and/or ionic strength of the solutions. |
Check pH and conductivity and adjust if necessary. Calibrate your conductivity and pH meters. |
Larger sample mass load applied compared with earlier runs. |
Keep mass of sample constant when repeating runs. (High proteins concentration can cause protein interaction, resulting in change of elution profile.) |
Reversed phase and hydrophobic interaction chromatography - Changes in ambient temperature affect retention times. |
Keep the temperature constant. |
Sample volume is different from earlier runs. |
Resolution is dependent on the sample volume. Keep sample volume constant when repeating runs. |
| Possible cause | Suggested remedy |
|---|---|
Altered stationary phase properties |
Confirm by running a known sample. If the change is persistant, replace the chromatography medium. |
Poor packing evaluation result
On this troubleshooting site, we refer to leading and tailing peaks as defined according to the following figures.
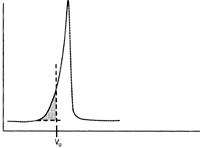

| Leading peak | Tailing peak |
Experience shows that the best method of expressing the efficiency of a packed column is in terms of its height equivalent to a theoretical plate (HETP), reduced plate number (h) and its peak asymmetry factor (As).
Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values.
Issues related to Poor packing evaluation result
| Possible cause | Suggested remedy |
|---|---|
Blocked bed support creates zones of different flow velocities. |
To remove protein: |
Cracks in the bed or a gap between the chromatography medium and inlet. The column may have been packed to loosely. |
It may be possible to only adjust the adaptor. Otherwise, repack the column with increased packing flow rate or pressure. After re-packing the column, evaluate the packing, using the recommended method. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
A crack due to a torn bed support resulting in a jet stream. |
Replace the bed support according to instructions. |
Cracks in the bed due to pump pulsation or a too densely packed bed. The cracks are mainly vertical. |
Repack the column with a pulse-free pump. After repacking the column evaluate the packing Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Test molecule is repelled by the chromatography medium under the conditions used, e.g. salt for ion exchange chromatography, or chromatography medium in water as running buffer. |
Use appropriate test molecule and conditions, e.g. 800 mM salt as sample in 400 mM salt buffer for ion exchange media, or use acetone. |
| Possible cause | Suggested remedy |
|---|---|
Test molecule interacts with the chromatography medium under the conditions used, e.g. acetone interacts with SOURCE media. |
Use appropriate test molecule and conditions, e.g. salt for SOURCE media. |
| Possible cause | Suggested remedy |
|---|---|
Flow velocity too high or too low. |
Flow velocity should be in the range 15-60 cm/h depending on particle size. The smaller the particle, the higher the velocity. |
Large liquid volume in tubing, valves, connectors, etc. |
Minimize dilution by reducing the liquid volume. For example, if the inner diameters of the tubing, valves, connectors, etc. are wider than the column inlet and outlet, change them to the same diameter. |
Too great a distance between the sample injection and column inlet, or between the column outlet and analyzing instrument |
Shorten these distances (minimizes dilution). |
Too large sample volume (gives a broad peak). |
Use a sample volume that is maximum 2% of the column volume. |
| Possible cause | Suggested remedy |
|---|---|
Bed packed too densely. |
Repack the column with decreased packing flow rate or pressure (prepare a pressure/flow curve and use the data to define the correct packing flow rate, i.e. approx. 70% of maximum flow (plateau)). After re-packing the column evaluate the packing. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Bed settled too hard during the first step. |
Repack the column with decreased packing flow rate for the sedimentation step. After re-packing the column evaluate the packing. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Extra volume in the system, e.g. new tubing (longer, shorter or different diameter) affects peak position and HETP. |
Ensure the equipment is the same or identical to your test routine or SOP. |
| Possible cause | Suggested remedy |
|---|---|
Column not properly equilibrated before packing evaluation |
Re-equilibrate the column. |
| Possible cause | Suggested remedy |
|---|---|
Column not properly equilibrated before packing evaluation |
Re-equilibrate the column. |
| Possible cause | Suggested remedy |
|---|---|
Bed packed too densely or too loosely. |
Repack the column. Evaluate the packing again, using recommended methods. For a leading peak and one-step packing: |
| Possible cause | Suggested remedy |
|---|---|
Bed packed too densely. Cracks present at the beginning get worse. |
Repack the column. Evaluate the packing again, using recommended methods. One-step packing: Two-step packing: Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Bed too loosely packed causing space between chromatography medium and inlet. |
Repack the column. One-step packing: Two-step packing: Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Bed not packed densely enough. |
Repack the column with increased packing flow rate or pressure (prepare a pressure/flow curve and use the data to define the correct packing flow rate, i.e. approx. 70% of maximum flow (plateau)). After re-packing the column, evaluate the packing. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Bed settled too loosely during the first step. |
Repack the column with increased packing flow rate for the sedimentation step. After re-packing the column, evaluate the packing. Preventive action: |
General advice to achieve good performance
Before using the system make sure that:
- The correct system has been selected in UNICORN System Control
- The correct wavelength has been set for UV/UPC monitor
- All tubing has been properly connected
- All connectors are free from leakage
- No tubing is folded or twisted
- Online filter, if used, is changed on a regular basis
- Correct buffers are used for the chosen columns and proteins
- All inlet tubing has been immersed in correct buffer solutions
- Enough buffer has been prepared
- Buffers have been equilibrated to the environment temperature
- Buffers/eluents have been degassed if necessary (e.g., in RPC runs)
- Suitable columns have been selected for the target proteins
- Columns have been cleaned and prepared according to column instructions
- Samples have been clarified by centrifugation and/or filtration prior to sample loading
- Samples have been adjusted to binding buffer conditions
- Auto sampler (if used) has been prepared according to user manual
- The fraction collector has been filled with appropriate number of microtiter plates or tubes
- Appropriate arrangement for waste handling has been prepared
Column leakage
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Connectors not compatible with each other. |
Check compatibility. |
Connectors not compatible with solvents. |
Check chemical resistance with the connector supplier. |
Connectors poorly positioned or not tightened. |
Check the connectors. |
Gaskets worn out. |
Gaskets lose flexibility with time and need to be replaced regularly. Inspect and replace if necessary and at least annually. |
| Possible cause | Suggested remedy |
|---|---|
End-piece and O-rings not properly positioned with respect to the tube. |
Disassemble the column and check the position of the end-piece and O-ring. Assemble the column according to instructions and perform leakage tests. |
O-rings worn out |
O-rings loose their flexibility with time and need to be replaced regularly. Disassemble the column and inspect the O-rings. Replace if necessary and at least annually. Assemble the column according to instructions and test for leakage. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Tubing not compatible with solvents. |
Check chemical resistance with the tubing supplier. |
Unsatisfactory elution
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Column is not properly equilibrated. |
Check pH and conductivity of the effluent before applying the sample to make sure the column is properly equilibrated. Continue to equilibrate with start buffer if necessary. |
Components in the sample displace the target molecule before elution starts. |
Reduce the amount of sample or find more suitable binding conditions. |
Flat titration curve of target protein. |
Decrease start and elution pH by 0.5 units next time. |
| Possible cause | Suggested remedy |
|---|---|
Highly charged protein at the chosen pH and the amount of salt in solution B is not enough to elute the protein. Your protein is more basic than most other proteins. |
Increase the salt concentration in solution B to release the protein from the column. |
pI of the protein is higher than your current estimate. |
Increase start and elution pH by 0.5 units next time |
| Possible cause | Suggested remedy |
|---|---|
This behavior may be due to ionic interaction between the protein and the chromatography medium. |
Keep the ionic strength between 0.15 to 0.3 M to minimize ionic interactions. |
| Possible cause | Suggested remedy |
|---|---|
Retardation may be due to aromatic and hydrophobic interactions between protein and the matrix. |
If possible decrease the ionic strength, increase pH or introduce an organic solvent (e.g. 24% propanol or 10% ethylene glycol) in solution A in order to minimize hydrophobic interactions. |
| Possible cause | Suggested remedy |
|---|---|
Eluent contains carbon dioxide. |
Degas all buffers prior to use. |
Eluent composition is incorrect. |
Check eluent pH. Make up fresh eluent if necessary. |
| Possible cause | Suggested remedy |
|---|---|
Column not properly equilibrated. |
Re-equilibrate the column. |
Sample not properly equilibrated. |
Equilibrate sample to correct operating conditions (pH, ionic strength, etc.) |
| Possible cause | Suggested remedy |
|---|---|
At the running pH, the overall charge of the protein is high and it binds very tight to the column. |
The pH stability profile of the protein limits the possibility to decrease the pH. Try to increase the ionic strength of your solution A to obtain an earlier elution, or choose another technique. |
pI of the protein is lower than most other proteins. |
Decrease start and elution pH by 0.5 units next time. |
pI of the protein is higher than most other proteins. |
Increase start and elution pH by 0.5 units next time. |
| Possible cause | Suggested remedy |
|---|---|
Column poorly packed. |
Evaluate the packing using recommended methods. |
Elution strength too low. |
Check and adjust the elution strength. Also check gradient, gradient shape or isocratic step. |
| Possible cause | Suggested remedy |
|---|---|
Carbon dioxide is present. |
Degas all buffers prior to use. In alkaline regions, degas water before making up eluent. |
Counterions are incorrect. |
Use recommended counterions. |
| Possible cause | Suggested remedy |
|---|---|
Larger sample load applied compare with earlier runs. |
Keep mass of sample constant when repeating runs. (High protein concentration can cause protein interaction, resulting in change of elution profile.) |
Sample volume is different from earlier runs. |
Resolution is dependent on the sample volume. Keep sample volume constant when repeating runs. |
| Possible cause | Suggested remedy |
|---|---|
Hydrophobicity of your protein is much higher than your current estimate and binds more strongly to the chromatography medium than you expected. |
Decreasing the ionic strength of solution A will save time. Another advantage is that many of the contaminants that would normally bind to the chromatography medium may elute in the void without binding. |
| Possible cause | Suggested remedy |
|---|---|
Target protein not stable under the chosen conditions and partly degrades. |
Find better ways to stabilize the protein, e.g. shorten the process time. |
The detergent has formed micelles with the protein, thereby increasing its size and changing its elution position. This effect is critical especially in gel filtration. |
Reduce the concentration of detergent to below it´s critical micelle concentration (CMC ) value. |
Column is bleeding from the previous run. |
Check and adjust your cleaning procedure. Clean the equipment and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Column not properly equilibrated. |
Check the pH and conductivity of the effluent before applying the sample. Continue to equilibrate with start buffer if necessary |
Components in the sample displace the target molecule before elution starts. |
Reduce the amount of sample or find more suitable binding conditions. |
| Possible cause | Suggested remedy |
|---|---|
Aggregate formation of protein sample. |
Use of zwitterions in the solutions, betaine up to 10%, pH 2 to 12, or taurine up to 4%, pH 4 to 6, to inhibit the formation of charge aggregates. |
If you protein is a lipoprotein, aggregate formation may have occurred. |
Use of zwitterions in the solutions, betaine up to 10%, or taurine up to 4%, may increase resolution and stabilize these complexes during chromatography |
Low molecular weight compounds can taken up the charge capacity of the column very quickly. |
The resolution may be improved if organic solvents or zwitterions are used in the buffers. However, the solubility of the zwitterions is lower in organic solvents than in water. |
Microbial growth in the column. |
Microbial growth rarely occurs in a column during use. However, always take steps to prevent infection of packed columns and solutions. Store the column according to instructions |
pH might not be optimal for your separation. |
Try running at another pH. It may affect the selectivity. |
pI of the protein is close to the pI of some major impurities. |
Try to obtain higher resolution by increasing the pH by 0.5 units and/or use a more shallow gradient. |
Too much sample has been loaded onto the column. |
Decreasing the sample load may improve the resolution or increase the amount of chromatography medium. I.e. increase the margin of safety significantly. |
Dead volume in chromatography systems is high |
Minimize dead volume in the chromatography system by decreasing capillary length and dimensions between injector and detector. Bypass unused system components e.g. column valves from the flow path. |
Different counter ions have different elution strengths. |
Switching to another counter ion may improve the selectivity. |
Flow velocity too high. |
Run the separation at a lower flow velocity. This is especially important for adsorbents that bind several substances and where selectivity is low. |
Gel filtration chromatography - Gel filtration column will resolve only molecules with approximately a two-fold difference in molecular weight. |
Use lower flow rate and/or try another technique |
Gel filtration chromatography- Sample is too viscous |
Dilute the sample with solution A. Reduce the protein concentration two-fold. |
Gel filtration chromatography- Sample volume is too large or the sample has been improperly applied. |
Decrease sample volume and apply the sample carefully. |
Gradient is too steep. |
Use a more shallow gradient. A plateau in the gradient where your protein elutes may improve the resolution. |
Hydrophobicity of your protein is much lower than your current estimate. The interaction between the protein and the chromatography medium is much weaker than expected. |
Apply the sample under conditions than enhance hydrophobic interaction, increase salt concentration and/or change pH towards the pI of your protein. |
| Possible cause | Suggested remedy |
|---|---|
Strength of the binding between the ligand and the protein varies with the protein type. |
Remedy 1 |
Buffers have wrong pH. |
Calibrate your pH meter, prepare new buffers and try again. |
pH of eluting buffer is too high. |
Try a lower pH range. |
pI of the protein is lower than your current estimate. |
Decrease start and elution pH by 0.5 units next time. |
pI of the protein is higher than your current estimate. |
Increase start and elution pH by 0.5 units next time. |
Protein is more acidic than estimated and is therefore highly charged at the suggested pH and is still bound to the column. |
If the protein does not elute in the end of the gradient, increase the salt concentration in eluting buffer to release the protein from the column. |
At the chosen pH the protein is highly charged and the amount of salt in solution B is not enough to elute the protein. Your protein is more basic than your first estimate. |
Increase the salt concentration in solution B to release the protein from the column. |
| Possible cause | Suggested remedy |
|---|---|
Composition of eluting buffer is incorrect. |
Check pH of eluting buffer. |
Highly charged protein at the chosen pH and the amount of salt in solution B is not enough to elute the protein. Your protein is more basic than most other proteins. |
Increase start and elution pH by 0.5 units next time. |
Hydrophobic proteins interact very strongly with the chromatography medium. |
Use an additive that decreases hydrophobic interactions. Mild organic solvents such as ethylene glycol, or detergents, or urea will elute tightly bound proteins but may lead to denaturation. |
Target protein is more acidic than most other proteins. It's therefore highly charged at the suggested pH. |
Decrease start and elution pH by 0.5 units next time. |
| Possible cause | Suggested remedy |
|---|---|
Column overloaded |
Decrease the amount of sample or increase the amount of chromatography medium. I.e. increase the margin of safety. |
pI of the protein is lower than most other proteins. |
Decrease start and elution pH by 0.5 units next time. |
pI of the protein is lower than your current estimate. |
Decrease start and elution pH by 0.5 units next time, or enter a new pI estimate for the protein and start a New Case. |
pI of the protein is higher than your current estimate. |
Increase start and elution pH by 0.5 units next time, or enter a new pI estimate for the protein and start a New Case. |
Protein is associated with another component that changes its true charge character. |
Use a preliminary clean up to remove these components or use zwitterions in the solutions. |
Protein can not bind to the chromatography medium, and may have been precipitated owing the low ionic strength of solution A. |
Choose another technique. |
Sample dissolved in wrong solution. |
Prepare a new solution A, check the pH and conductivity; dissolve your sample in solution A and repeat the run. |
Solution A and B have been mixed up. |
Check pH and conductivity of the solutions, dissolve the sample in solution A and repeat the run. |
For ion exchange chromatography - Ionic strength of the sample too high or pH incorrect. |
Check and adjust the ionic strength and/or pH. Calibrate your conductivity and pH meters. |
For ion exchange chromatography - Reduced net charge due to complex between the component in the sample and the target molecule. |
Check the composition of the sample. Remove any interfering compounds such as nucleic acids. |
Oppositely - charged detergents or other additives adsorbed on the column. |
Clean the column according to the Media and column instructions. |
pH of the sample incorrect. |
Check and adjust the pH. Calibrate your pH meter. |
The column not properly equilibrated. |
Check the pH and conductivity of the effluent before applying the sample. Continue to equilibrate with start buffer if necessary. |
Highly charged protein at the chosen pH and the amount of salt in solution B is not enough to elute the protein. Your protein is more basic than your first estimate. |
Increase the salt concentration in solution B to release the protein from the column. |
Hydrophobic interaction chromatography- Hydrophobicity of your protein is much lower than your current estimate. The interaction between the protein and the chromatography medium is much weaker than expected. |
Apply the sample under conditions that enhance hydrophobic interactions; increase salt concentration and/or change pH towards the pI of your protein. |
Ionic strength of solution A is too high for binding of the protein. |
Since some salt is needed in solution A, try decreasing the pH of solution A to obtain binding of the protein. |
| Possible cause | Suggested remedy |
|---|---|
Anionic detergents or other additives are adsorbed to the column. |
Clean the column according to the Media and column instructions. |
Ionic strength of the sample is too high. |
Remove salt in the sample by gel filtration in Polybuffer. |
pI of the protein is higher than most other proteins. |
Increase start and elution pH by 0.5 units next time. |
Protein is associated with another component which changes its true charge character. |
Use a preliminary cleanup to remove these components or use zwitterions in the buffers. |
Sample is dissolved in wrong buffer. |
Prepare new buffers, check the pH and conductivity, dissolve your sample in start or eluting buffer and repeat the run. |
Start pH is too low. |
Choose a start buffer with a higher pH. |
| Possible cause | Suggested remedy |
|---|---|
Buffers have wrong pH, ionic strength or solvent concentration. |
Check and adjust the buffer conditions. Calibrate your instruments. |
Back pressure increases during operation
| Possible cause | Suggested remedy |
|---|---|
Air trapped in the bed supports. |
Get rid of the air by wetting and washing the bed supports with 20% ethanol. Fill a syringe with 20% ethanol and inject the liquid into the bottom of the column. Trapped air in the top net should be avoided by inject 20 % ethanol to the top adaptor before assembling the adaptor. Make sure that the ethanol passes the nets. |
Microbial growth in buffers. |
Check buffers, especially those with phosphate, for microbial growth. Replace with fresh buffer if necessary. |
Precipitation of protein on the column. |
Remedy 1 |
Precipitation of proteins on the column at high salt concentration resulting in increased hydrophobic interaction. |
Use chaotropic salts in solution B. However, if the protein structure is destroyed choose another technique. |
Sample and collection vessels at different levels. |
Adjust the vessels to approximately the same level. |
The prefilter might be blocked. |
Check the prefilter. |
Valve not fully open. |
Check all valves. Open any that is not fully open. |
Auxiliary equipment such as manometers and pumps not working properly. |
Check the function of all auxiliary equipment. Repair/replace if necessary. |
Bed packed too densely. |
Evaluate the packing. Please be aware of that for small columns, less than 10 ml bed volume, the system dead volume has an impact on the column evaluation values. |
Bent tubing. |
Check that the flow path is not restricted. |
Buffer viscosity too high. |
Check the viscosity of all buffers. Viscosity is a function of temperature. (Lower temperature gives higher viscosity.) Let low-temperature buffer reach operating temperature before starting the run. |
Column bed supports blocked by protein or medium. |
To remove protein: |
For hydrophobic interaction chromatography - Protein precipitated on the column due to incorrect ionic strength. |
Check and adjust conductivity to the correct value. Clean and regenerate the column and chromatography medium according to instructions. |
Ionic strength of solution may have caused the protein to precipitate in the column. |
Remedy 1 |
Lipoproteins are present and may clog the column. |
Lipoproteins may be precipitated prior to chromatography by the addition of 10% dextran sulfate and 1 M calcium chloride to final concentrations of 0.2 % and 0.5 M respectively. Remove precipitate by centrifugation. |
Unusual column appearance
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Buffers and column at different temperatures. |
The column must generally be repacked. However, you can remove a small amount of air trapped at the column top by pumping a solution with a temperature a few degrees centrigrade higher than the chromatography through the column. Use upward flow. If the flow has been reversed, re-evaluate the packing prior the use. Evaluate the packing using recommended methods. |
Connectors not correctly tightened or not compatible with each other. |
The column must generally be repacked. However, you can remove a small amount of air trapped at the column top by pumping a solution with a temperature a few degrees centrigrade higher than the chromatography through the column. Use upward flow. If the flow has been reversed, re-evaluate the packing prior the use. Evaluate the packing using recommended methods. Preventive action: |
The column operates at room temperature after having been stored in a cold room. |
Allow thermal equilibration before use. |
Valves that should be closed are not shut tightly. |
The column must generally be repacked. However, you can remove a small amount of air trapped at the column top by pumping a solution with a temperature a few degrees centrigrade higher than the chromatography through the column. Use upward flow. If the flow has been reversed, re-evaluate the packing prior the use. Evaluate the packing. Preventive action: |
| Possible cause | Suggested remedy |
|---|---|
Bed support damaged or incorrectly assembled allowing chromatography medium particles to leave the column. |
Check the bed support and replace if necessary. Disassemble the column according to instructions. |
Buffer conditions deviate with regard to temperature, conductivity, viscosity, content of organic solvent (reduces surface tension) or other factor. |
Check the buffers and choose more suitable conditions. |
Increased resistance to flow due to blocked bed support compressing the packed bed. |
Clean or change the bed support. Disassemble the column and replace the support according to instructions. Preventive action: |
Poorly packed bed (not sufficiently compressed during packing). |
Evaluate the packing. |
| Possible cause | Suggested remedy |
|---|---|
Some carbon dioxide gas bubbles might develop with a carbonate buffer during ligand coupling. |
It does not affect the coupling result. |
| Possible cause | Suggested remedy |
|---|---|
Column not stored under bacteriostatic conditions. |
Include a bacteriostatic agent in the storage solution, e.g. 20% ethanol or 10 mM NaOH. |
Connectors not correctly tightened. |
Check and tighten if necessary. |
Column and chromatography medium not correctly cleaned. |
Clean the column and chromatography medium according to instructions. Replace the bed supports with new ones if necessary |
Strange peaks
Select the description that fits with your symptom
| Possible cause | Suggested remedy |
|---|---|
Unwashed buffer tanks. |
Clean the tanks and prepare fresh solutions. |
Leakage of non-eluted protein due to poor cleaning-in-place. |
Clean and regenerate the column and chromatography medium according to instructions |
| Possible cause | Suggested remedy |
|---|---|
Air inadvertently injected with the sample. |
Try to remove air bubbles by washing extensively with solution at high flow velocity. Run in upward flow direction. |
Column and/or solution subjected to sudden temperature changes. |
Equilibrate the column and solutions to working temperature before use. |
Sample not completely dissolved in buffer. |
Ensure that the sample is completely dissolved before applying it to the column. |
System effects, valve-switching, dead volumes, etc. cause uneven buffer velocity. |
Minimize if possible. Accept the remaining effects. |
