FAQ
Sephadex G-10, G-15, G-25, G-50, G-75, and G-100 are based on dextran of different molecular weight ranges. The lower the G-value, the higher the degree of cross-linking of the dextran polymers, and the lower the molecular weight fractionation range. Additionally, with increasing degree of cross-linking, the swelling of the medium decreases.
Coarse and Medium grades are preferred for group separations at process scale where high flow rates and low operating pressures are required. The Fine and Superfine grades are for routine laboratory work including small-scale preparative separations (e.g. HiPrep 26/10 Desalting [Fine], HiTrap Desalting [Superfine]). The smaller particle sizes of the Fine and Superfine grades give shorter diffusion distances and allow for highly efficient separations at high flow rates. DNA grade is tested and certified to ensure reproducibility and high recovery with DNA to ensure no nonspecific binding.
| Medium | Grade | Fractionation range, peptides and globular proteins (Mr) |
|---|---|---|
| Sephadex G-10 | - 700 | |
| Sephadex G-15 | - 1 500 | |
| Sephadex G-25 | Superfine Fine Medium Coarse |
1 000 - 5 000 1 000 - 5 000 1 000 - 5 000 1 000 - 5 000 |
| Sephadex G-50 | Superfine Fine Medium Coarse |
1 000 - 30 000 1 000 - 30 000 1 000 - 30 000 1 000 - 30 000 |
| Sephadex G-75 |
Superfine |
3 000 - 80 000 3 000 - 70 000 |
| Sephadex G-100 |
Superfine |
4 000 - 150 000 4 000 - 100 000 |
* in distilled water
Packing By Gravity Flow
- Pour the entire slurry into the column in one portion; be careful not to trap air bubbles.
- Start the gravity flow to initiate packing.
Packing Using a Flow Adapter
Good packing is essential to obtain good resolution. Ideally, the column should be packed at the highest pressure possible without deforming the beads.
For softer resins, like Sephadex, care must be taken to avoid compressing the resin. Do not increase the pressure beyond the values given in the table below.
With wider columns, slightly reduced maximum operating pressures must be used. Flow rate is inversely proportional to bed height: increasing the bed height will decrease the flow rate, but does not affect the maximum operating pressure.
- Using a reservoir if necessary, pour the entire slurry into the column in one portion.
- Start the pump to initiate packing.
- Once all the resin has settled into the column, remove the reservoir.
- Insert an adapter and pack at the maximum operating pressure of the resin.
- Once the resin is thoroughly packed into the column, adjust the flow adapter to the surface of the resin.
- Wash the column with an additional 2 to 3 column volumes of the buffer to be used in the separation. This stabilizes and equilibrates the bed.
- Readjust the flow adapter to the surface of the resin bed.
| Chromatography medium | Grade | Approx. maximum operating pressure (bar) |
|---|---|---|
| Sephadex G-10 | All | 0.6 |
| Sephadex G-15 | All | 0.6 |
| Sephadex G-25 | All | 0.8 |
| Sephadex G-50 | All | 0.1 |
| Sephadex G-75 | All | 0.16 |
| Sephadex G-100 | All | 0.096 |
| Product | PSDdry (Within specification)1 | PSDwet2 |
|---|---|---|
| µm | µm | |
| Sephadex G-10 | 40-1203 | 52-173 (20-180) |
| Sephadex G-15 | 40-1203 | 58-184 (20-160) |
| Sephadex G-25 SF | 20-504 | 33-89 (10-110) |
| Sephadex G-25 F | 20-804 | 33-140 (10-180) |
| Sephadex G-25 M | 50-1505 | 84-262 (20-280) |
| Sephadex G-25 C | 100-3005 | 166-531 (90-550) |
| Sephadex G-50 SF | 25-504 | 51-106 (20-130) |
| Sephadex G-50 F | 20-804 | 41-171 (20-240) |
| Sephadex G-50 M | 50-1505 | 103-320 (40-370) |
| Sephadex G-75 | 40-1204 | 92-293 (40-320) |
| Sephadex G-75 SF | 20-504 | 46-122 (20-150) |
| Sephadex G-100 | 40-1204 | 94-329 (40-360) |
| Sephadex G-100 SF | 20-504 | 49-134 (20-170) |
1 Within sieving limits, guide for which Size Exclusion resin to choose depending on application
2 Calculated wet PSD range based on water regain. Range within which particles can be found is presented within parentheses and should be used as guide for choosing filter.
3 ≥ 85% of the volume of particles are found within the range shown
4 ≥ 80% of the volume of particles are found within the range shown
5 ≥ 90% of the volume of particles are found within the range shown
Sephadex is compatible with commonly used aqueous solutions in the pH range 2 to 13. It is stable to all commonly used buffers, solutions of urea (8 M), guanidine hydrochloride (6 M), and all non-ionic and
ionic detergents. Lower alcohols (methanol, ethanol, propanol) may be used in the buffer or the sample, but we recommend that the concentration be kept below 25 v/v%. Prolonged exposure (hours) to pH values below 2 or above 13, or to oxidizing agents should be avoided.
Sephadex is supplied as a dry powder and must be allowed to swell in excess buffer before use (see steps below). After swelling, adjust with buffer to form a thick slurry from which air bubbles are removed under vacuum. Approximately 75% settled medium is suitable. Fine particles can be decanted. Filter all buffers through a 0.22 µm Whatman filter to help prevent microbial growth.
- Weigh out the appropriate amount of dry Sephadex for the required bed volume of your column (see table). For group separations, use up to a 10 cm bed height. For example, if using a 26 mm i.d. column, a 10 cm bed height equals 53 ml of medium. Sample volumes can be up to 30% of the column volume. Pack a quantity of medium up to five-fold the volume of the sample. If packing at the maximum pressure for the medium, choose the low end of the bed range from the table for calculating the amount of dry Sephadex.
- Calculate the total volume of the column. Add 1.3 times this volume of your swelling buffer to the dry beads. Swelling times for the different types of Sephadex are given in the table. The process is accelerated by using a boiling water bath, which also serves to deaerate the buffer.
- Stir - excessive stirring should be avoided as it may break the beads. DO NOT USE MAGNETIC STIRRERS!
- After swelling is complete, decant the supernatant.
- Add buffer to make a 75% suspension (i.e., a ratio of 75% settled gel to 25% buffer, as measured by height in a beaker or other container).
- Degas the suspension before packing.
| Resin | Grade | Approx. mL of bed volume/1 g resin* |
Swelling time, 20° C (h) |
Swelling time, 90° C (h) |
|---|---|---|---|---|
| Sephadex G-10 | All | 2-3 | 3 | 1 |
| Sephadex G-15 | All | 3-4 | 3 | 1 |
| Sephadex G-25 | All | 5-6 | 3 | 1 |
| Sephadex G-50 | Superfine | 121 | 3 | 1 |
| Sephadex G-50 | Fine | 13-16 | 3 | 1 |
| Sephadex G-50 | Medium | 14-15 | 3 | 1 |
| Sephadex G-75 | All | 191 | 24 | 3 |
| Sephadex G-100 | All | 211 | 72 | 5 |
* Swelling in swollen bed in 0,02M NaCl, should be used as guide to column/vessel size.
1 Representative swelling presented, statistical foundation not available
Packing procedures
One step compression packing.
| Column | Maximum operation pressure (bar) |
|---|---|
| HiScale | 20 |
| XK 16 | 5 |
| XK 26 | 5 |
| XK 50 | 3 |
| BPG 100 | 8 |
| BPG 140 | 6 |
| BPG 200 | 6 |
| BPG 300 | 4 |
| BPG 450 | 2.5 |
| BioProcess LPLC | 6 |
| BioProcess MPLC | 20 |
| Chromaflow 400 | 3 |
| Chromaflow 600 | 3 |
| Chromaflow 800 | 3 |
| Chromeflow 1000 | 3 |
| FineLINE Pilot 35 | 20 |
| FineLINE 70 | 20 |
| FineLINE 100 | 20 |
| FineLINE 200 | 20 |
| FineLINE 350 | 20 |
Constant flow packing.
Refer to the document Constant flow packing in BPG columns, method description and practical example, which you will find under the Related Documents tab. The same method is valid for Blue Sepharose 6 Fast Flow, Butyl Sepharose 4 Fast Flow, Butyl-S Sepharose 6 Fast Flow, Chelating Sepharose Fast Flow, CM Sepharose Fast Flow, DEAE Sepharose Fast Flow, Heparin Sepharose 6 Fast Flow, IMAC Sepharose 6 Fast Flow, Ni Sepharose 6 Fast Flow, Octyl Sepharose 4 Fast Flow, Phenyl Sepharose 6 Fast Flow (low sub), Phenyl Sepharose 6 Fast Flow (high sub), PlasmidSelect, Q Sepharose Big Beads, Q Sepharose Fast Flow, Q Sepharose XL, Sephadex G-25, Sepharose 4 Fast Flow, Sepharose 6 Fast Flow, SP Sepharose Big Beads, SP Sepharose Fast Flow and SP Sepharose XL
in BPG columns.
Packing buffer: Water for Blue Sepharose 6 Fast Flow, Chelating Sepharose Fast Flow, CM Sepharose Fast Flow, DEAE Sepharose Fast Flow, Heparin Sepharose 6 Fast Flow, IMAC Sepharose 6 Fast Flow, Ni Sepharose 6 Fast Flow, PlasmidSelect, Q Sepharose Big Beads, Q Sepharose Fast Flow, Q Sepharose XL, Sepharose 4 Fast Flow, Sepharose 6 Fast Flow, SP Sepharose Big Beads, SP Sepharose Fast Flow and SP Sepharose XL.
Packing buffer: Water or 0.15 M NaCl for Sephadex G-25
Packing buffer: 20 % Ethanol for Butyl Sepharose 4 Fast Flow, Butyl-S Sepharose 6 Fast Flow, Octyl Sepharose 4 Fast Flow, Phenyl Sepharose 6 Fast Flow (low sub) and Phenyl Sepharose 6 Fast Flow (high sub).
Recommended flow velocity intervals in the table below are for packing the column. The intervals are given in reverse order to indicate that the high flow velocity can be used for shorter beds whereas higher beds only allow lower flow velocity, due to the higher pressure drop across the bed.
| Column | Compression factor | Slurry concentration (%) | Bed height (cm) | Flow velocity (cm/h) |
|---|---|---|---|---|
| BPG 100 | 1.2 | 50 - 70 | 10 - 20 | 320 - 250 |
| BPG 100 | 1.2 | 50 - 70 | 20 - 30 | 250 - 150 |
| BPG 140 | 1.2 | 50 - 70 | 10 - 20 | 320 - 250 |
| BPG 140 | 1.2 | 50 - 70 | 20 - 30 | 250 - 150 |
| BPG 200 | 1.2 | 50 - 70 | 10 - 20 | 320 - 250 |
| BPG 200 | 1.2 | 50 - 70 | 20 - 30 | 250 - 150 |
| BPG 300 | 1.2 | 50 - 70 | 10 - 20 | 320 - 250 |
| BPG 300 | 1.2 | 50 - 70 | 20 - 30 | 250 - 150 |
10 µm net stainless steel
Two step packing using constant flow in both steps.
Packing buffer: water or 0.15 M NaCl
| BPG 100 |
|---|
| 10 or 12 µm nets |
| Adaptor net 10 µm Code no: 18-1103-05 |
| Adaptor net 12 µm Code no: 18-1103-06 |
| End piece net 10 µm Code no: 18-0251-77 |
| End piece net 12 µm Code no: 18-1104-41 |
| BPG 140 |
| 10 or 12 µm nets |
| Adaptor net 10 µm Code no: 18-1113-03 |
| Adaptor net 12 µm Code no: 18-1113-05 |
| End piece net 10 µm Code no: 18-1113-02 |
| End piece net 12 µm Code no: 18-1113-04 |
| BPG 200 |
| 10 or 12 µm nets |
| Adaptor net 10 µm Code no: 18-0252-76 |
| Adaptor net 12 µm Code no: 18-1104-42 |
| End piece net 10 µm Code no: 18-0252-77 |
| End piece net 12 µm Code no: 18-1104-43 |
| BPG 300 |
| 10 or 12 µm nets |
| Adaptor net 10 µm Code no: 18-1012-55 |
| Adaptor net 12 µm Code no: 18-1104-44 |
| End piece net 10 µm Code no: 18-1012-35 |
| End piece net 12 µm Code no: 18-1104-45 |
| Column | Compression factor | Slurry concentration (%) | Bed height (cm) |
|---|---|---|---|
| XK 16/40 | 1.2 | 50 - 70 | 10 - 30 |
| XK 26/40 | 1.2 | 50 - 70 | 10 - 30 |
| XK 50/30 | 1.2 | 50 - 70 | 10 - 29 |
Column evaluation
The efficiency of a column depends on how well it is packed. A poorly packed column gives rise to uneven flow, resulting in zone broadening and reduced resolution. It is thus important to have a method by which the column can be tested before it is put into operation. Such a method should be simple, quantitative and should not introduce contaminating materials. It is also an advantage if the same method can be used to monitor column performance over its working life, so that it is easy to determine when the medium should be re-packed or replaced.
Avoid methods that use colored compounds such as Blue Dextran. They do not meet the above criteria and cannot be used with ion exchange and affinity chromatography media.
Experience has shown that the best method of expressing the efficiency of a column is in terms of the height equivalent to a theoretical plate, HETP, reduced plate number, h, and the peak asymmetry factor, As. These values can be determined easily by applying a NaCl or acetone solution, to the column (see below).
It is important that the column is properly equilibrated ( >2 column volumes) before evaluating the packing. Ideally, run three test runs to see whether the values are stable. If an initially poor result improves during a later test, the reason can be that the column was not properly equilibrated. To check that the bed is stable, run the column at 70% packing pressure for 20 hours and test it again.
Note that pressure spikes may cause poor packing (cracking). If this happens, fit an air trap and a pressure relief valve between the pump and column. Locate the pressure relief valve between the air trap and the column.
Choice of test sample for columns
The most appropriate material for column testing is, of course, the sample that is to be run in the application, but this is not always practical or economical. As an alternative, a solution of either NaCl or acetone will give a good indication of the column packing quality. The eluate is monitored by measuring conductivity or UV absorption, and the resulting elution profile is used to calculate the HETP value.
The advantages of using NaCl are that it is readily available and can be used safely to test all columns. One disadvantage is that NaCl may interact with the medium matrix, especially ion exchanger matrices, and thus give erroneous results.
Acetone, in contrast, does not interact with the matrix and is detected by UV absorption at 280 nm. Alternatively, you can increase the running buffer concentration 10-fold and use it as test solution.
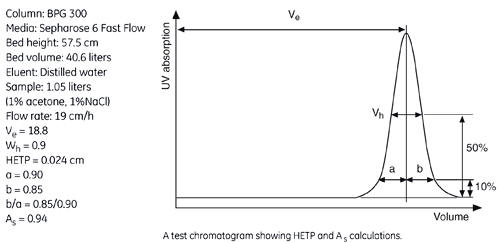
The figure below shows a UV trace for acetone in a typical BPG column application and gives calculated HETP and As values.
HETP calculation
The sample volume should be approximately 1% of the total bed volume and the concentration 1.0% v/v NaCl, or equivalent when using stronger buffer. Alternatively, use 1.0% v/v acetone. The flow velocity should be between 10 and 30 cm/h depending on the bead size of the chromatography medium. The high flow velocity could be used for small beads whereas large beads only allow low flow velocity. To avoid diluting the sample, apply it as close to the column inlet as possible. If an airtrap is included in the system, by-pass it during sample application to avoid back-mixing. Calculate the HETP value from the conductivity (or UV) curve as follows:
HETP, in its simplest terms, is expressed as:
HETP = L/N
where,
L = Bed height (cm)
N = Number of theoretical plates.
N is defined by the equation:
N = 5.54 (Ve /Wh)2
where,
Ve = Elution volume (ml)
Wh = Peak width at half height (ml)
Ve is measured as the volume passed through the column to the peak maximum.
Wh is measured as the peak width at half-peak height.
From the example in the figure, the HETP value can be calculated from the chromatogram as follows:
| Ve (ml) | Wh (ml) | N | N/m | HETP cm | |
| Acetone | 18800 | 900 | 2417 | 4203 | 0.024 |
Well-packed columns have low HETP values. However, it is only possible to compare columns that have been packed with the same type of media and that have been tested under identical conditions.
As a general rule-of-thumb, a good HETP value is approximately two to four times the mean bead diameter of the medium in question, provided that the sample does not interact with the medium.
In practice, the correlation between HETP and column performance can only be assessed by the column operator. Once this has been established, a standard can be set to judge the acceptability of a column packing.
For example, the column operator may know from experience that a column packed with Sephadex G-25 gel filtration medium with HETP values above 0.05 cm does not give the required separation. Consequently, the operator will set this value as the maximum permissible i.e. the minimum acceptable quality.
Reduced plate number
Definition of reduced plate number: h = HETP/dp
h = reduced plate number.
HETP = above described height equivalent to a theoretical plate.
dp = mean particle diameter of the chromatography medium beads.
The reduced plate number should be in the range of 2-4 times the mean particle diameter of the chromatography medium beads.
Peak asymmetry factor calculation
The peak asymmetry factor should be as close as possible to 1, and the shape of the peak should be as symmetrical as possible. This is usually the case for gel filtration media, but for certain ion exchange and affinity media, the shape may be asymmetrical due to interaction with the media. A change in peak shape is usually the first indication of column deterioration.
The peak asymmetry factor, As, is calculated from the graph above:
As = b/a
where,
a = distance from peak apex to 10% of the peak height on the ascending side of the peak
b = distance from peak apex to 10% of the peak height on the descending part of the peak
Note: Measuring HETP, h and As values is the best way to judge the condition of the packed column. A packed column can look good, but still need repacking for optimal performance. Always check the column after packing and regularly between runs to ensure best performance.
| XK 16/40 |
|---|
| 10 µm nets (5 pieces/pack) Code no: 18-8761-01 |
| XK 26/40 |
| 10 µm nets (5 pieces/pack) Code no: 18-8760-01 |
| XK 50/30 |
| 10 µm nets (5 pieces/pack) Code no: 18-8759-01 |
| Column | Compression factor | Slurry concentration (%) | Bed height (cm) |
|---|---|---|---|
| BioProcess LPLC 400-800 | 1.2 | 50 - 70 | 10 - 30 |
| BioProcess LPLC 900-1200 | 1.2 | 50 - 70 | 15 - 30 |