Mariangela Manicone, Senior Scientist
Genna Luciani, Scientist II
Traditionally, cryopreserved cells are thawed using a water bath system. We compare the performance of a water bath and VIA Freeze™ system for thawing cryopreserved T cells in vials. We demonstrate an equivalent performance between the traditional water bath and the VIA Freeze™ system in terms of post-thaw cell count and viability, opening the door to an alternative contamination-free system for thawing vials that allows for traceability.
Introduction
Thawing cryopreserved cell vials is an integral part of the cell therapy manufacturing process. The methods used can influence cell viability, function, and recovery, underscoring the need to optimize and standardize thawing procedures for consistent patient dosing. Conventionally, cell vials are thawed in a water bath at 37°C. This method poses contamination risks, requires calibration and validation, and most critically, does not provide a record of the thaw process.
In cell therapy manufacturing, traceability from collection to transfusion is key to simplifying regulatory submissions. Beyond enabling compliance, recording thawing details such as temperature monitoring, time to thaw, and operator ID also supports process reproducibility and helps researchers correlate thawing conditions with cell performance outcomes.
As the cell therapy field evolves, both cryopreserved bags and vials are increasingly used depending on the stage of development and scale. Bags are commonly used in clinical and commercial manufacturing due to their compatibility with closed-system processing and infusion workflows. In contrast, vials are often preferred in early-stage research, preclinical studies, and small-scale clinical trials, where flexibility, lower volume requirements, and easy handling are key. A transition from vials to bags would ideally be facilitated by vial thaw process data, however there are limited solutions for accurately recording this information.
The VIA Freeze™ controlled-rate freezer is designed to cryopreserve a wide range of cell types, including T cells, while enabling compliance with US Food and Drug Administration (FDA) Title 21 CFR Part 11 (electronic records). The system includes a heater assembly that performs a heating cycle at the end of each cryopreservation run to remove unwanted moisture from the system before a new protocol is initiated. This heating feature, in combination with the integrated data collection capabilities of the VIA Freeze™ system, could enable precise control of the vial thawing process in a closed system.
This study evaluates the heater assembly in the VIA Freeze™ system in combination with the corresponding Society for Biomolecular Screening (SBS) plates for thawing vials of cryopreserved CD3+ T cells and compares its performance with a water bath.
Materials and methods
Starting materials
Prior to this study, 6 × 108 to 9 × 108 T cells were isolated from the leukapheresis of five healthy donors. CD3+ T cells were magnetically separated using the EasySep human CD3 positive selection kit II and the EasySep magnet (StemCell Technologies). Immediately after isolation, cells were diluted with EasySep buffer (StemCell Technologies) and prepared for cryopreservation (Fig 1).
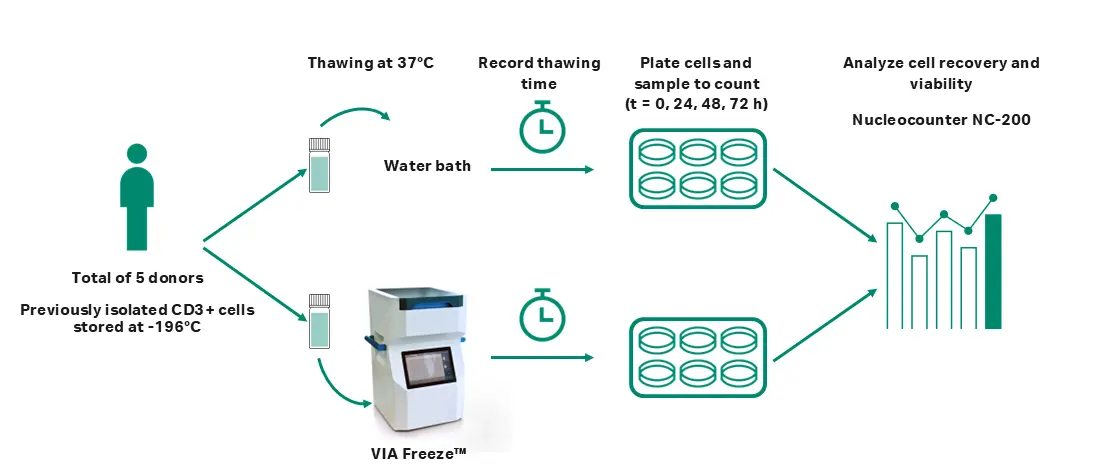
Fig 1. The experimental process used in this study.
Cryopreservation with VIA Freeze™ system
A minimum of two vials per donor were prepared for cryopreservation with CryoStor CS10 (Biolife Solutions) at a density of 1 × 107 to 2.5 × 107 cells/mL in 2 mL Corning cryogenic vials and frozen at a rate of -1°C/min to -100°C using a VIA Freeze™ system. Vials were then stored at -196°C for a minimum of two years.
Cell thawing with water bath and the VIA Freeze™ system
The 48 × 2 mL standard size vial SBS plates (ASY 30075, Cytiva) were used in combination with the VIA Freeze™ system to allocate the vials (1 mL sample volume) and load them into the system. The VIA Freeze™ system, together with the SBS plates, was warmed to a setpoint temperature of 37°C with a 1.5°C/min warming rate prior to introducing samples.
For each donor, two vials were thawed: one using the VIA Freeze™ Duo system and the other using a water bath (Fig 1). Once the setpoint temperature was reached, vials were transferred from the -196°C storage to each thawing system. During the thawing run, vials were inspected approximately every 30 s to qualitatively assess thawing. Vials were removed from the system when most of the ice was melted, with the remaining ice dissolving upon gentle agitation of the vials being an indication of successful thawing. Thawing time was monitored and recorded for both procedures.
Cell culture
After thawing, cells were promptly washed using Xuri™ T cell expansion medium (Cytiva) supplemented with 5% GemCell US-origin human AB serum (GeminiBio) and Xuri™ interleukin-2 (IL-2) growth factor (350 UI/mL) (Cytiva) to remove residual cryoprotectant. Cells were then seeded at a concentration of 1 × 106 cells/mL with the same media in 6-well clear flat bottom ultra-low attachment plates (Corning Costar) for up to 72 h.
Cell analysis
Cell number and membrane integrity were analyzed immediately post-thaw (T0) and after 24, 48, and 72 h in culture (T24, T48, and T72). The analyses were conducted using the NucleoCounter NC-200 with Via1-cassette containing acridine orange and 4′,6-diamidino-2-phenylindole (DAPI) stains (ChemoMetec) following the manufacturer’s procedure.
For cell analysis, an appropriate volume of cell culture was sampled (approx. 0.5 mL), diluted 1:1 in the same culture media [Xuri™ T cell expansion medium supplemented with 5% GemCell US-origin human AB serum (GeminiBio) and Xuri™ interleukin-2 (IL-2) growth factor (350 UI/mL) (Cytiva)], and counted in triplicate. An average of the three counts were reported for each parameter analyzed: cell viability (%), cell concentration (viable cells per mL), and total live cell count post-thaw.
Cell recovery was calculated as the percentage of total live cell number at T0 (immediately post-thaw) relative to the estimated live cell number in the cryopreserved sample.
Results
The average cell viability observed immediately post-thaw (T0) was 97.49% ± 1.85% with the VIA Freeze™ system and 97.72% ± 1.75% for the water bath method. The average cell recovery was 94.7% ± 4.5% with the VIA Freeze™ system and 96.6% ± 4.6% for the water bath. No statistically significant difference in cell viability or recovery immediately post-thaw (T0) was observed between the two methods (n = 5; viability P value = 0.842; recovery P value = 0.531; unpaired, two-tailed T test, GraphPad Prism 10 software) (Fig 2).
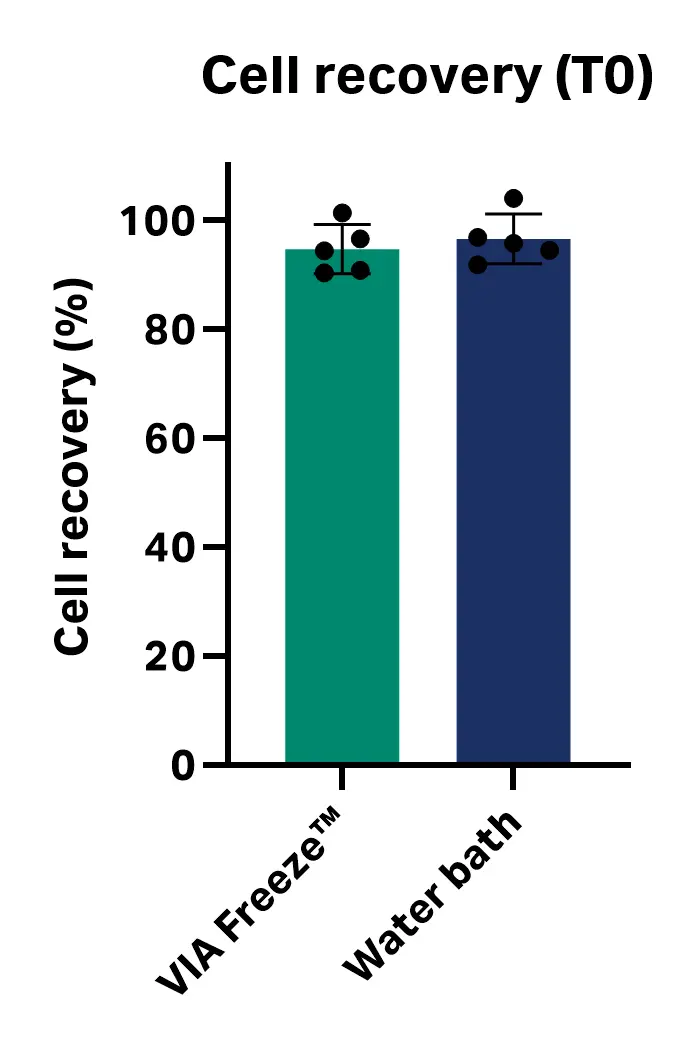
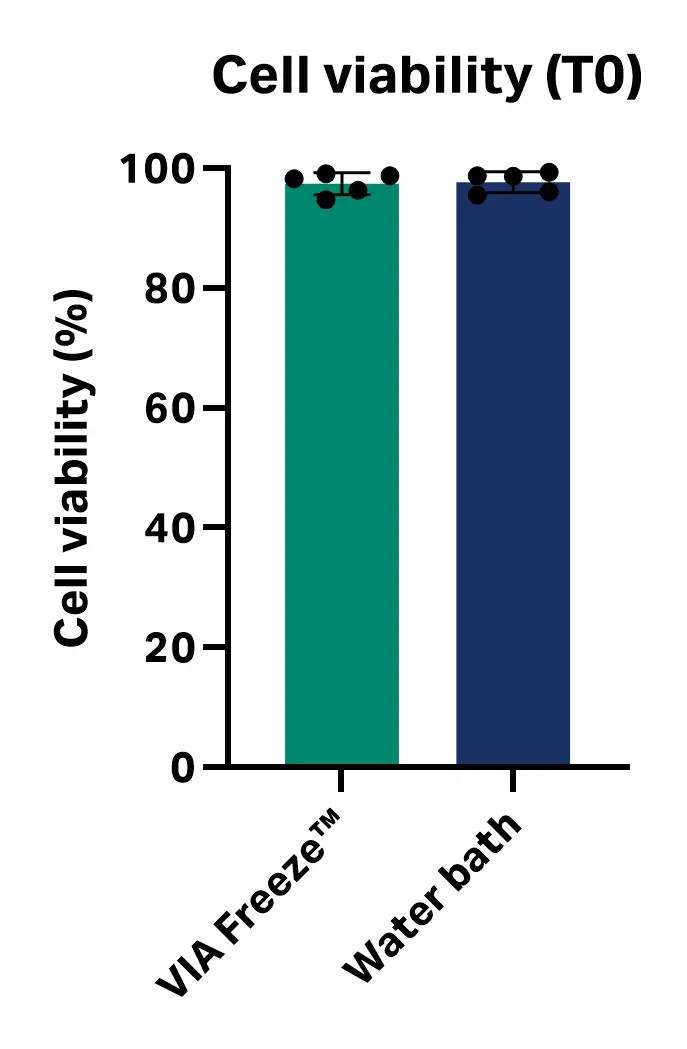
Fig 2. Cell viability and recovery immediately post-thaw (T0) with VIA Freeze™ system and water bath.
Over 72 h of culture, no significant differences in cell viability or total viable cell number were observed between the thawing methods at any time point. These results suggest that thawing with the VIA Freeze™ system performs comparably to a water bath for cryopreserved CD3+ T cells under the tested conditions [n = 5; viability P values = 0.730 (0 h), 0.301 (24 h), 0.232 (48 h), and 0.436 (72 h); total viable cell number P values = 0.044 (0 h), 0.315 (24 h), 0.141 (48 h) and 0.185 (72 h), multiple unpaired T test, GraphPad Prism 10 software] (Fig 3). Table 1 shows average viability and total viable cell number at each time point.
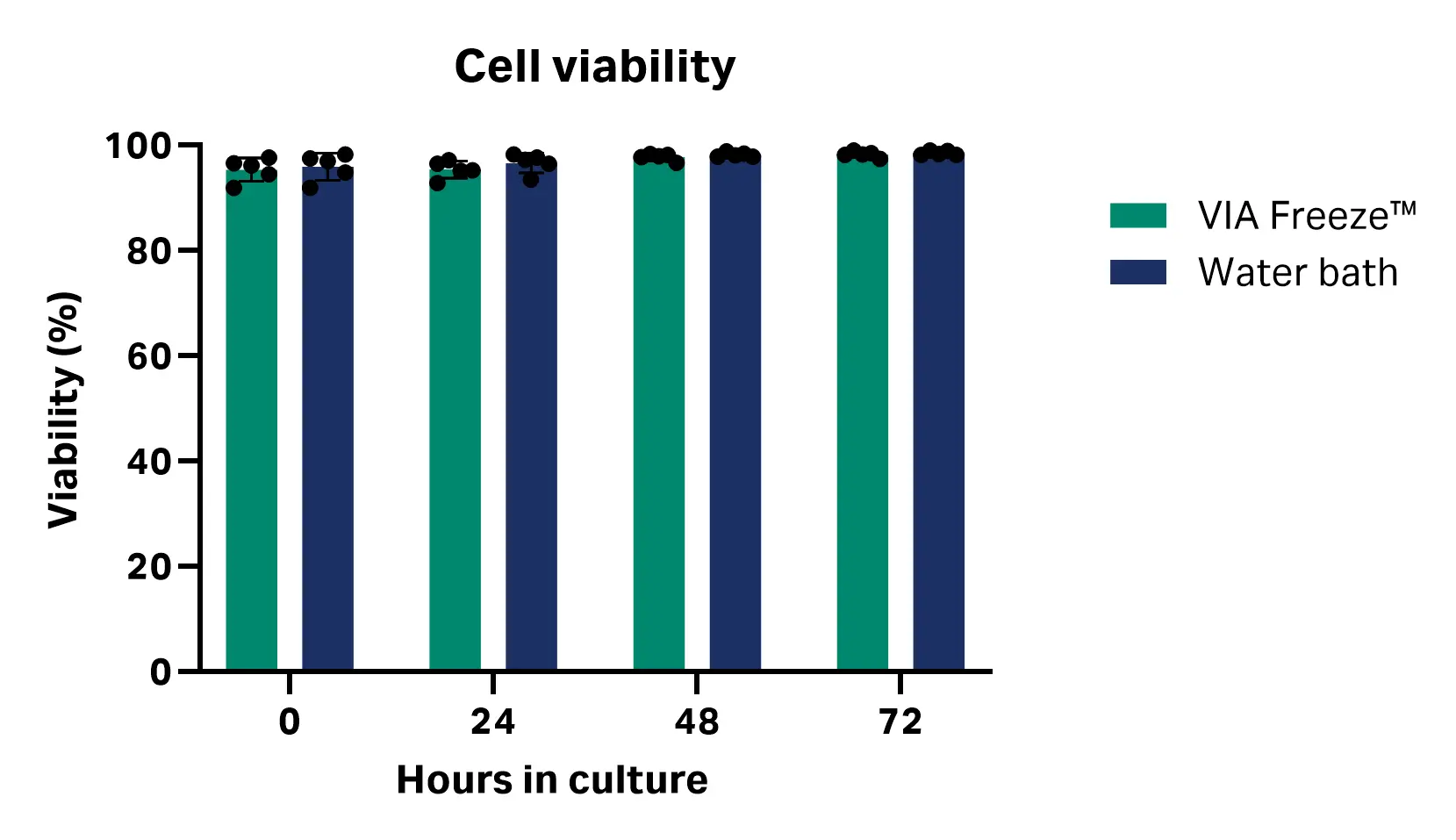
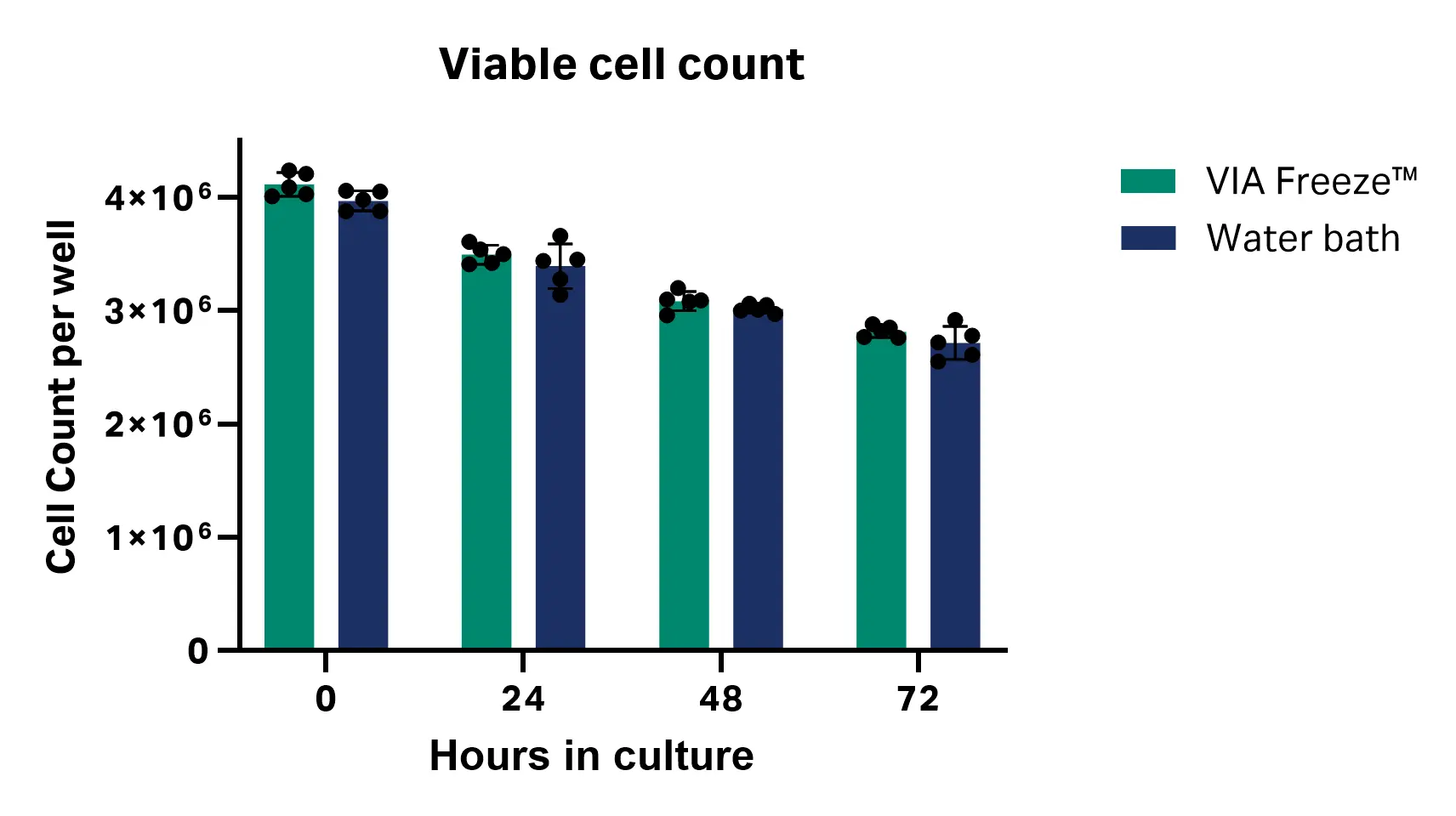
Fig 3. Cell viability and total viable cell count at 24, 48, and 72 h post-thaw with VIA Freeze™ system and water bath.
Table 1. Cell viability and total viable cell count results (n = 5)
| VIA Freeze™ system | Water bath | |||
|---|---|---|---|---|
| Time post-thaw (h) | Viability (%) | Viable cell count (cells/sample) | Viability (%) | Viable cell count (cells/sample) |
| 0 | 95.41 ± 2.23 | 4.12 × 106 ± 1.04 × 105 | 95.95 ± 2.56 | 3.97 × 106 ± 8.78 × 104 |
| 24 | 95.42 ± 1.66 | 3.50 × 106 ± 8.39 × 104 | 96.65 ± 1.86 | 3.39 × 106 ± 1.96 × 105 |
| 48 | 97.79 ± 0.66 | 3.09 × 106 ± 8.53 × 104 | 98.24 ± 0.41 | 3.02 × 106 ± 3.70 × 104 |
| 72 | 98.30 ± 0.57 | 2.82 × 106 ± 5.13 × 104 | 98.55 ± 0.40 | 2.72 × 106 ± 1.45 × 105 |
The only statistically significant difference in thawing performance between the VIA Freeze™ system and the water bath methods was time: cells thawed with the VIA Freeze™ system took 95 s longer to thaw on average than the vials thawed in the water bath (average thawing times with the VIA Freeze™ system and water bath were 221 ± 8 s and 127 ± 3 s, respectively; P value ≤ 0.0001; unpaired, two-tailed T test, GraphPad Prism 10 software) (Fig 4). This difference had no impact on cell viability or recovery at any of the analyzed time intervals.
The thawing time difference is likely a reflection of the thermal properties of the two systems. The water bath conducts heat to the vials through direct contact with water (at 37°C), whereas the VIA Freeze™ system transfers heat from the heating plate to the SBS plates, and then to the vials. However, other factors such as opening and closing the VIA Freeze™ system lid to inspect the vials at frequent time intervals could have impacted thawing time.
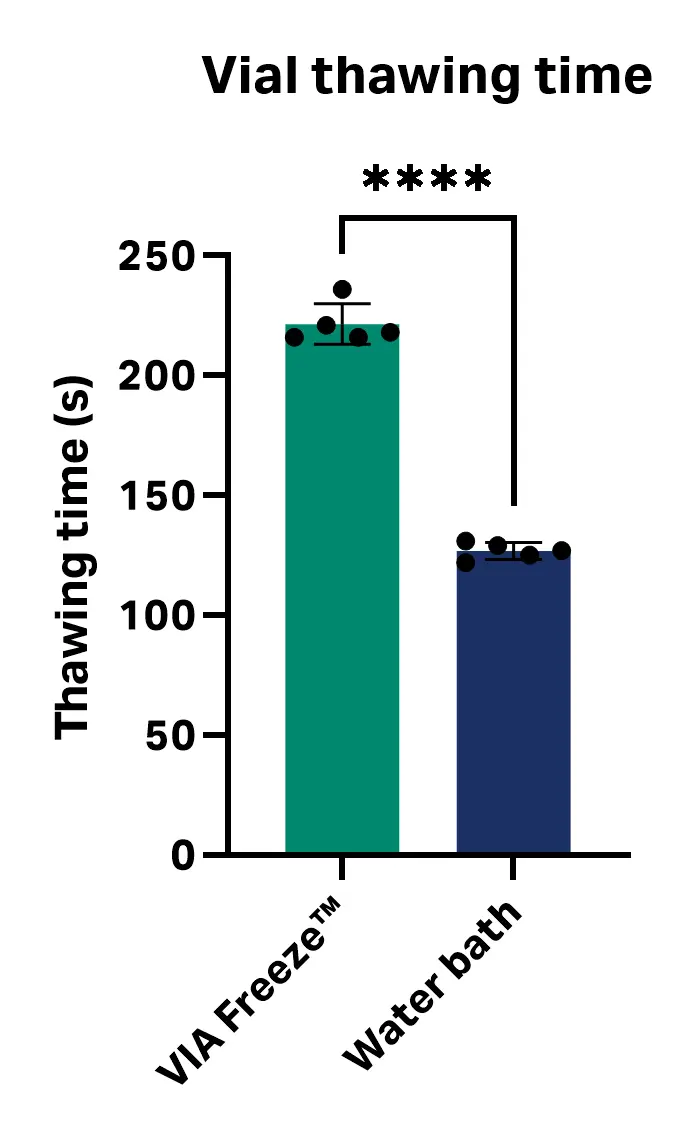
Fig 4. Thawing time with the VIA Freeze™ system and water bath.
Conclusion
This study demonstrates that the VIA Freeze™ system provides equivalent performance to conventional water bath thawing in terms of CD3+ T cell viability and density. Importantly, it enables GMP compliance by eliminating open contamination risks inherent to water baths and by automatically logging thawing data. Each thawing run on the VIA Freeze™ system can capture critical parameters such as protocol details, temperature profiles, user ID, batch ID, and timestamps—supporting traceability, reproducibility, and process optimization. These features make the VIA Freeze™ system a robust solution for closed, standardized vial thawing in early-stage cell therapy workflows.
CY55754
Cytiva and the Drop logo are trademarks of Life Sciences IP Holdings Corporation or an affiliate doing business as Cytiva.
VIA Freeze™ and Xuri™ are trademarks of Global Life Sciences Solutions USA LLC or an affiliate doing business as Cytiva. Any other trademarks are the property of their respective owners.
© 2025 Cytiva
For local office contact information, visit cytiva.com/contact