We sought to evaluate the reproducibility and tech transfer of a fed-batch process for recombinant monoclonal antibody (mAb)-producing Chinese hamster ovary (CHO) cell in Xcellerex™ X-50 bioreactors. We transferred the production process from a facility in Uppsala, Sweden, to one in Portsmouth, UK, using a ReadyToProcess WAVE™ 25 rocking bioreactor to generate the seed culture. Nutrient/metabolite, cell growth, and titer data were consistent across runs, indicating a reproducible process and successful tech transfer.
Fig 1. Xcellerex™ X-platform bioreactors including the Xcellerex™ X-50 bioreactor (left)
Introduction
Reproducibility of cell culture runs from one facility to another is crucial in bioprocess development, scale-up, and scale-out. It’s also a key process performance benchmark in cell culture processes and bioreactor systems. Here we used common cell culture performance data to test the reproducibility of a fed-batch CHO cell process in a 50 L Xcellerex™ X-platform bioreactor (X-50) after tech transfer from one Cytiva facility to another. In this application note, we’ll describe the cell culture process including materials used, our approach to tech transfer of the process, and the cell culture performance results.
Tech transfer
To ensure the successful transfer of the process, we employed a multi-step approach involving three key categories: documentation and material transfer, risk assessment and gap analysis, and the study plan and batch records.
The first step is the documentation and material transfer, beginning with a comprehensive review of the current documentation. This includes cell culture protocols, allowing us to understand the process in detail and identify any necessary adaptations to our own protocols. A comprehensive risk assessment is performed to identify potential risks, including equipment, site, and process differences, as well as process variability and supply chain disturbances. Each identified risk is then evaluated and prioritized, allowing us to develop mitigation strategies to reduce the likelihood and risk impact. Finally, a study plan is developed to ensure process alignment. This document details the process description and set points and serves as the reference for the study.
As part of this project, we made the move from paper to electronic batch records using Cytiva Chronicle™ automation software. We used the software to create electronic standard operating procedures (eSOPs) that formed the basis of the batch records that were used as the step-by-step guide to run the batch. This meant scientists not involved in the document transfer and study plan writing could confidently operate the process.
Fig 2. Chronicle automation software
Moving from paper to electronic batch manufacturing records (eBMRs) has substantially enhanced our tracking and audit capabilities through electronic signatures and real-time data recording. Crucially, storing data at the point of entry strengthens the data integrity and makes it easier to keep our process steps well-organized. The system flags any deviations at the point of data entry, helping us avoid mistakes and making the subsequent review easier. We've also implemented barcode and QR code scanning. This means that for critical equipment, such as the bioreactor, we can scan directly within the eBMR to link the equipment to the relevant batch data and process steps. All process data is then uploaded to Chronicle™ software, ensuring a centralized and easily accessible record.
The fed-batch cell culture process
CHO cells are a bioprocessing industry standard for producing mAbs, owing to their well-established regulatory history and ability to generate complex glycoproteins. Fed-batch stirred-tank processes have been widely used to generate culture with high cell densities, resulting in high protein titers. Here we wanted to assess the reproducibility of cell culture performance in the Xcellerex™ X-platform stirred-tank bioreactor, which was developed to support high-density cell cultures. For the seed culture, we used a ReadyToProcess WAVE™ 25 bioreactor, a single-use 25 L rocking bioreactor designed for fast installation and convenient handling. This makes it well suited for use as a N-1 bioreactor to deliver reliable and predictable performance suitable for research, process development, and manufacturing operations.
We ran three replicate cultures (R1, R2, and R3) at our Portsmouth, UK, facility in the X-50 bioreactor based on an established historical process carried out in the Xcellerex™ X-50 in Uppsala, Sweden. The criteria for success were comparable cell growth, metabolite profiles, and mAb titer.
Materials and methods
Table 1. Consumables and reagents used during this study.
| Type | Name |
| Cell line | Chinese hamster ovary (CHO) cell line expressing the immunotherapeutic mAb, Herceptin™ (Genentech Inc.) |
| Seed train | 125 mL shake flask |
| Seed train | 250 mL shake flask |
| Seed train | 1000 mL shake flask |
| N-1 bioreactor | Cellbag™ with Fortem film, pHOPT, DOOPT II |
| Bioreactor | X-50 bag, fed-batch II |
| Manifold | Probe sheath |
| Reagent | ActiPro™ medium |
| Reagent | HyClone Cell Boost™ 7a supplement |
| Reagent | HyClone Cell Boost™ 7b supplement |
| Reagent | HyClone™ ADCF™ antifoam |
| Reagent | 45% glucose |
| Reagent | Sodium bicarbonate |
| Reagent | L-Methionine sulfoximine (MSX) |
| Reagent | Sodium hydroxide |
CHO cells were expanded in shake flasks in ActiPro™ medium supplemented with 37.5 µM L-methionine sulfoximine. The cells were sub-cultured until a sufficient number of cells to inoculate the ReadyToProcess WAVE™ 25 bioreactor at 10 L was achieved. During the seed train, the cells were kept in the early exponential phase (0.3-5.0 × 106 cells/mL). The cells were cultured in ActiPro™ basal medium and fed with HyClone Cell Boost™ 7a and 7b supplements. ActiPro™ medium in combination with HyClone Cell Boost™ 7a and HyClone Cell Boost™ 7b supplements, is a chemically defined, animal-derived component-free (ADCF) system specifically designed to maximize protein production from CHO cells in fed-batch suspension cultures. Manufactured under cGMP conditions, this system is free of peptides, hydrolysates, hypoxanthine, and thymidine.
ReadyToProcess WAVE™ bioreactor culture
We set up the ReadyToProcess WAVE™ 25 rocker using standard procedures as described in ReadyToProcess WAVE™ 25 User Manual. To summarize, we unpacked a 20 L Cellbag™ bioreactor bag and connected the associated manifolds aseptically. We then transferred the bioreactor bag to the rocking platform and connected the dissolved oxygen (DO) and pH probe adapters to the respective sensors on the underside of the bag. We attached the gas line and filter heater to the bag and then inflated the bag with air. The day before inoculation, we added 7 L medium to the bag and initiated mixing, gas flow, and temperature at the set points listed in Table 2. The media was allowed to equilibrate overnight at 37°C and 7.5% CO2. A sample was taken for analysis on a Nova BioProfile Flex 2 cell culture analyzer, and the bioreactor pH probe was offset by the difference between the reading on the controller and the analyzer. Once the DO reading was stable, the DO was re-calibrated to 92.5% air saturation.
We calculated the volume of cells required to inoculate the ReadyToProcess WAVE™ 25 rocker according to Table 2, topped up the calculated volume to 3 L with pre-warmed medium, and added it to the bioreactor. The operating conditions for the ReadyToProcess WAVE™ 25 rocker are described in Table 2. We sampled the culture daily for analysis on the cell culture analyzer and a Vi-CELL XR cell counter (Beckman). Cells were cultured until reaching the target inoculation concentration for the Xcellerex™ X-50 bioreactor (Table 2).
Table 2. ReadyToProcess WAVE™ 25 rocker set points.
| Parameter | Set point |
| Temperature | 37°C |
| pH control | N/A |
| DO set point | 40% |
| DO control | Air/oxygen |
| Rocking speed | 22 rpm |
| Rocking angle | 6° |
| Rocking motion | 50% |
| Air flow | 0.25 L/min |
| CO2 concentration | 7.5% |
| Starting volume (after inoculation) | 10 L |
| Target inoculation concentration | 0.8 ± 0.1 × 106 cells/mL |
X-50 culture
We set up the X-50 as described in Xcellerex™ X-50 and X-200 bioreactors Figurate™ powered by PlantPAx® Operating Instructions. To summarize, we unpacked, installed, and inflated the X-50 fed-batch perfusion II bag. The probes were calibrated, autoclaved, and installed in the X-50 bioreactor. One day before inoculation, we added 25 L medium to the X-50 bag and initiated agitation, pH, gas flow and temperature at the set points listed in Table 3. The media was allowed to equilibrate at 37 °C overnight. A sample was taken for analysis on the BioProfile Flex 2 cell culture analyzer, and the bioreactor pH probes were offset as described above. Once the DO was stable, the DO was re-calibrated to 100% air saturation and control started.
Table 3. Xcellerex™ X-50 bioreactor set points.
| Parameter | Bioreactor set point |
| pH ± deadband | 6.95 ± 0.15 |
| Temperature | 37°C |
| DO | 40% |
| pH control | CO2 (sparger 1)/NaHCO3 (7.5%) |
| DO control | O2 (sparger 1) |
| Sparger 1 air flow rate (initial) | 0.3 L/min |
| Superficial gas velocity | 0.052 mm/s |
| VVM | 0.006 |
| Agitation speed for equilibration* | 148 rpm |
| Power input per volume for equilibration* | 45 W/m3 |
| Agitation speed after inoculation* | 166 rpm |
| Power input per volume after inoculation* | 63 W/m3 |
| Agitation direction | Upflow |
| Volume (after inoculation) | 35.6 L |
| Inoculum cell concentration | 0.9 ± 0.1 × 106 cells/mL |
*Determined from Bioreactor Scaler Pro v3.0.674.
We calculated the number of cells required to inoculate the Xcellerex™ X-50 to a final concentration of 0.9 ± 0.1 × 106 cells/mL at a post-inoculation volume of 35.6 L, and added the inoculum to the bioreactor, along with enough media to achieve a final volume of 35.6 L. The bioreactor operating conditions are described in Table 3. The cell culture was sampled daily, and the bioreactor was inspected for foam buildup, with antifoam added dropwise as required.
Three days after inoculation, we initiated the fed-batch phase utilizing HyClone Cell Boost™ 7a and 7b supplements and 45% glucose solution. HyClone Cell Boost™ supplement 7a was added at 2.5% of the bioreactor starting volume and HyClone Cell Boost™ supplement 7b at 0.25% of the bioreactor starting volume (35.6 L) on a daily basis. HyClone Cell Boost™ supplement 7b was added first, followed by HyClone Cell Boost™ supplement 7a, both over a period of 15 min. After each feed, the agitation speed was adjusted (if required) to maintain the power to volume ratio (P/V) between 61 W/m3 and 63 W/m3, calculated according to the Bioreactor Scaler. The bioreactor culture was fed daily until day 14, at which point the cells were harvested.
We monitored the glucose concentration daily and calculated the theoretical post-feed concentration, taking into account the concentration of glucose from the HyClone Cell Boost™ supplement 7a (77 g/L). A 45% glucose feed was added according to Table 4.
Table 4. Glucose feed criteria.
| Glucose condition | Lactate condition | Target glucose concentration |
| < 2.0 g/L | > 2.0 g/L | 4 g/L |
| = 0 g/L | < 2.0 g/L | 5 g/L |
| = 0 g/L | < 1 g/L | 5-6 g/L |
Results and discussion
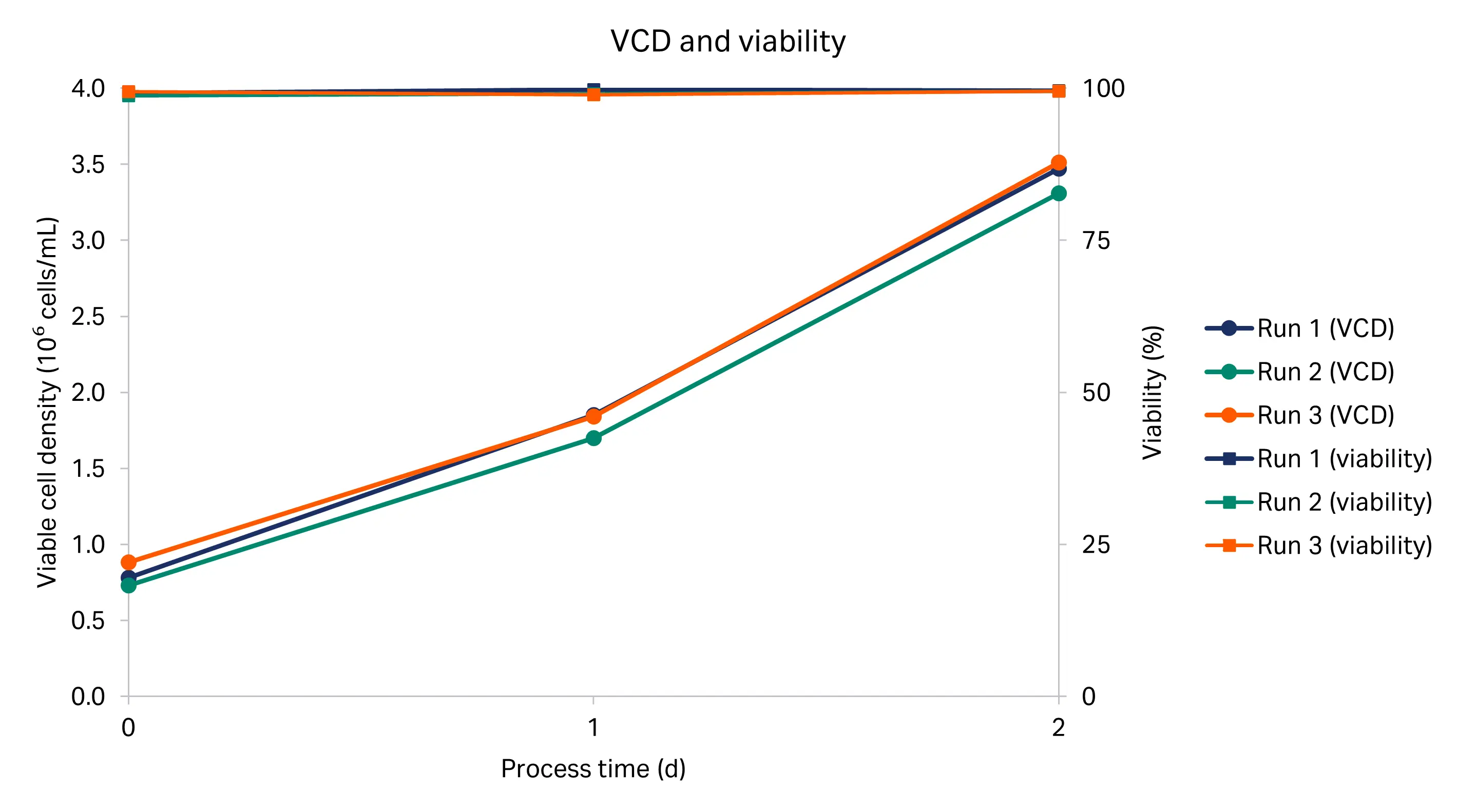
The viable cell density (VCD) and viability for the three ReadyToProcess WAVE™ 25 rocker cultures are shown in Figure 3. These cultures were used to inoculate the Xcellerex™ X-50 bioreactor cultures after 3 d of growth, when the cell density reached 3.4 ± 0.1 × 106 cells/mL.
Fig 3. VCD (solid line) and viability (dashed line) profiles for three runs in the ReadyToProcess WAVE™ 25 rocker (R1, R2, R3).
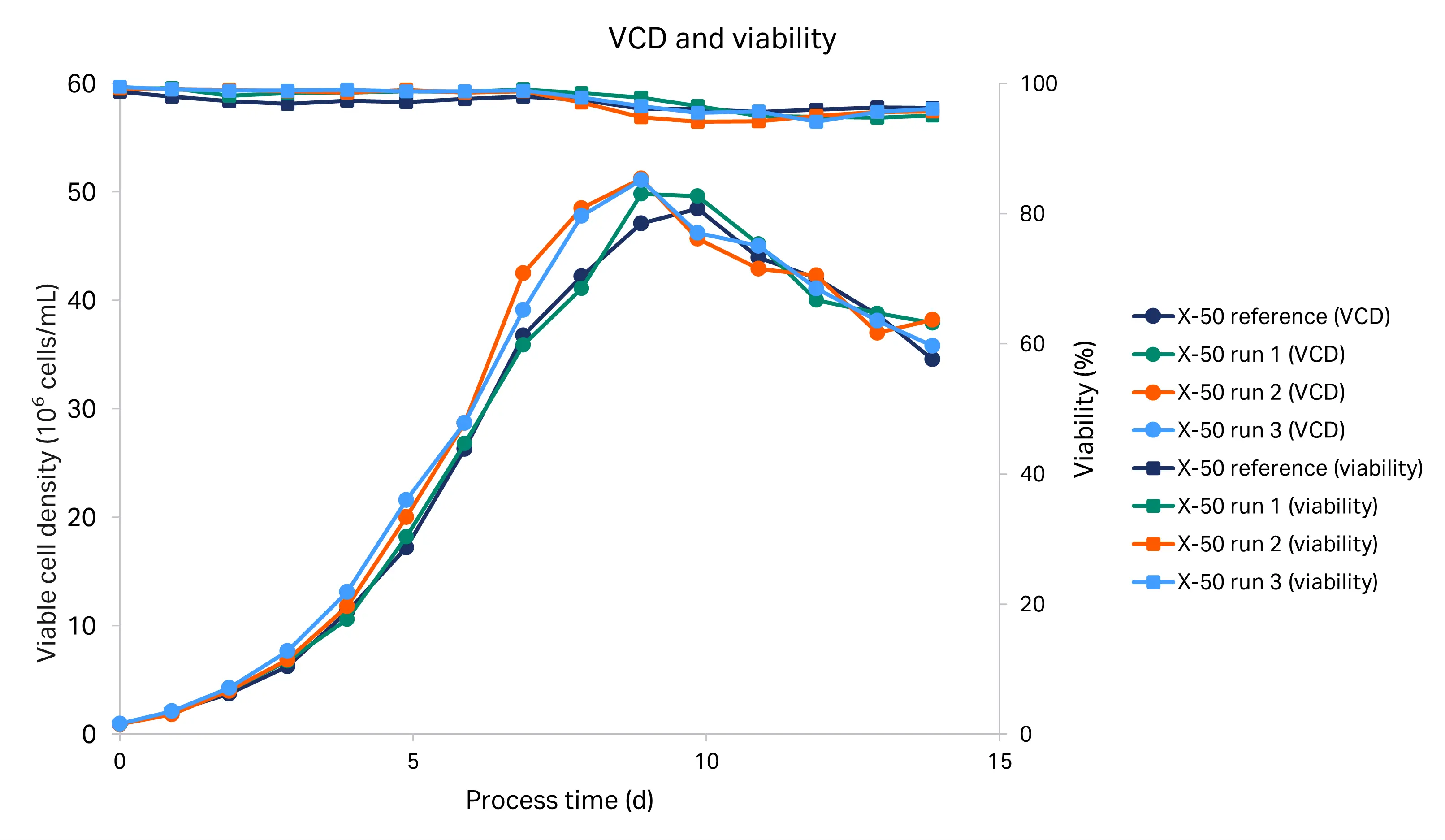
The cell growth profiles for the historical run (X-50 reference) and tech transfer runs (X-50 R1, X-50 R2, and X-50 R3) in the Xcellerex™ X-50 bioreactor are shown in Figure 4. For all runs there was close overlap during growth stage, with minor variability at peak cell density within the analytics error and consistent with the large dilutions required for cell counts (1:6). The viable cell density peaked at 48-51 × 106 cells/mL between day 8 and 10. The viability measured on the Vi-CELL XR cell counter remained high (95-100%) from inoculation to harvest for all runs.
Fig 4. VCD and viability for the historical and tech transfer X-50 runs.
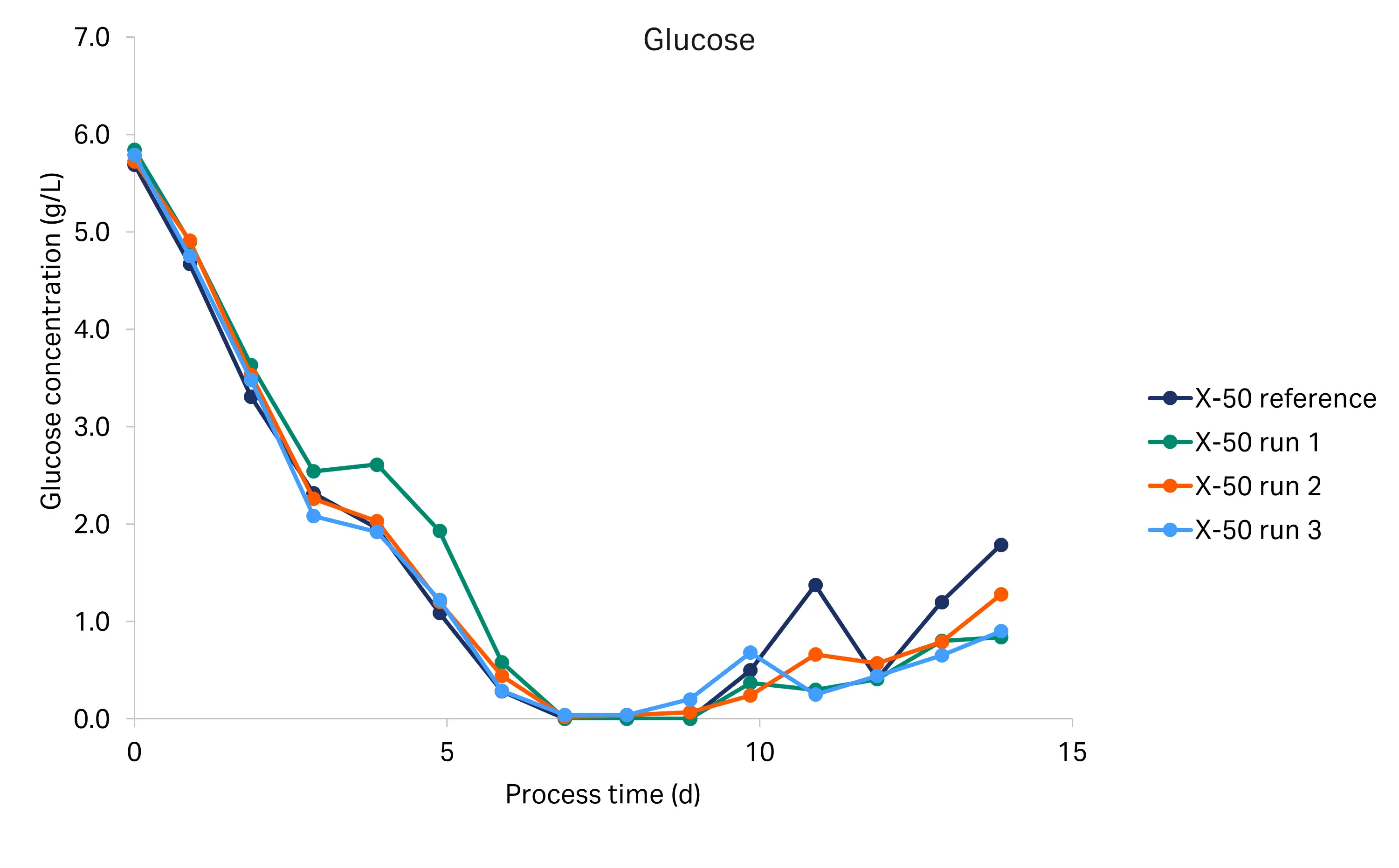
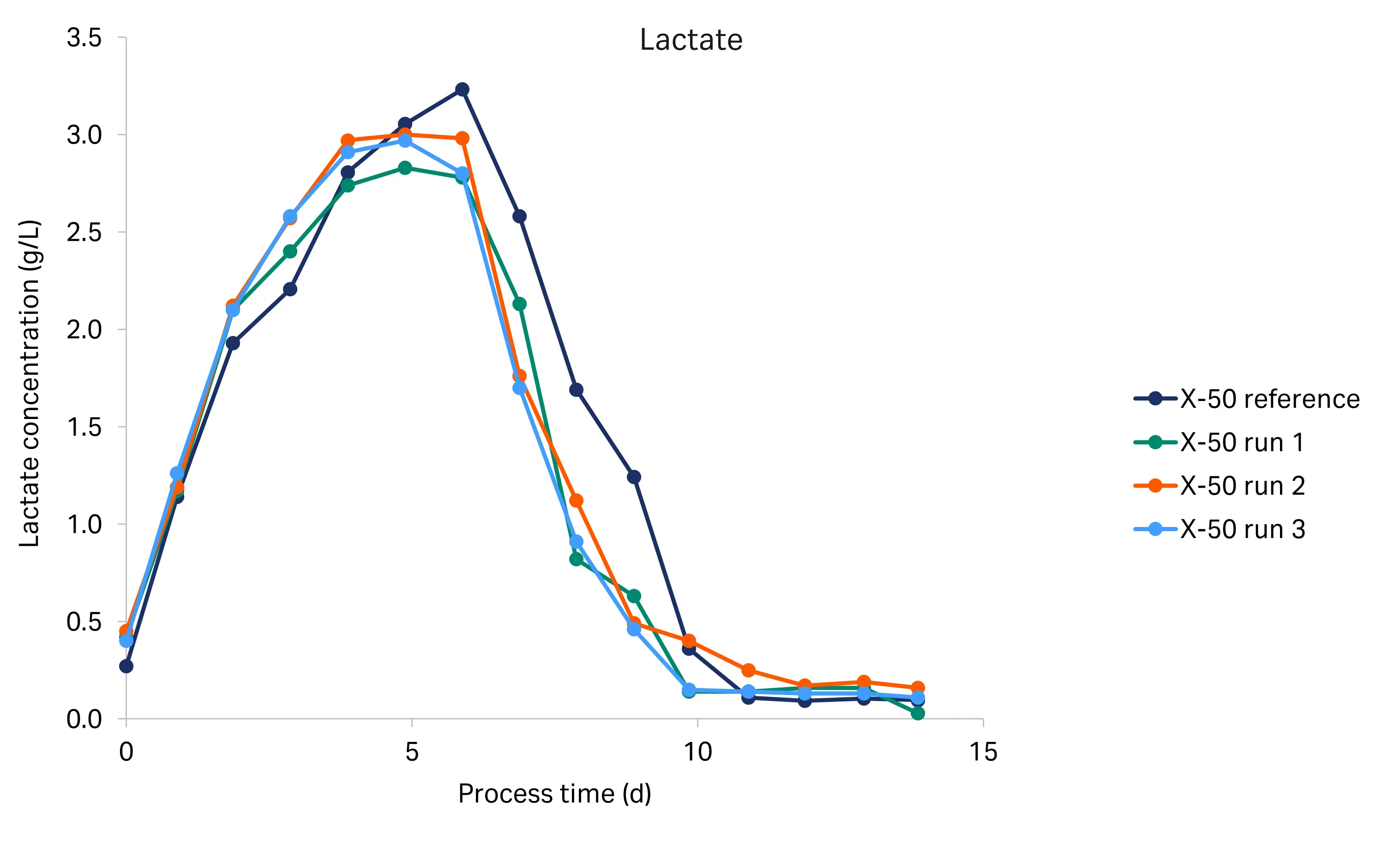
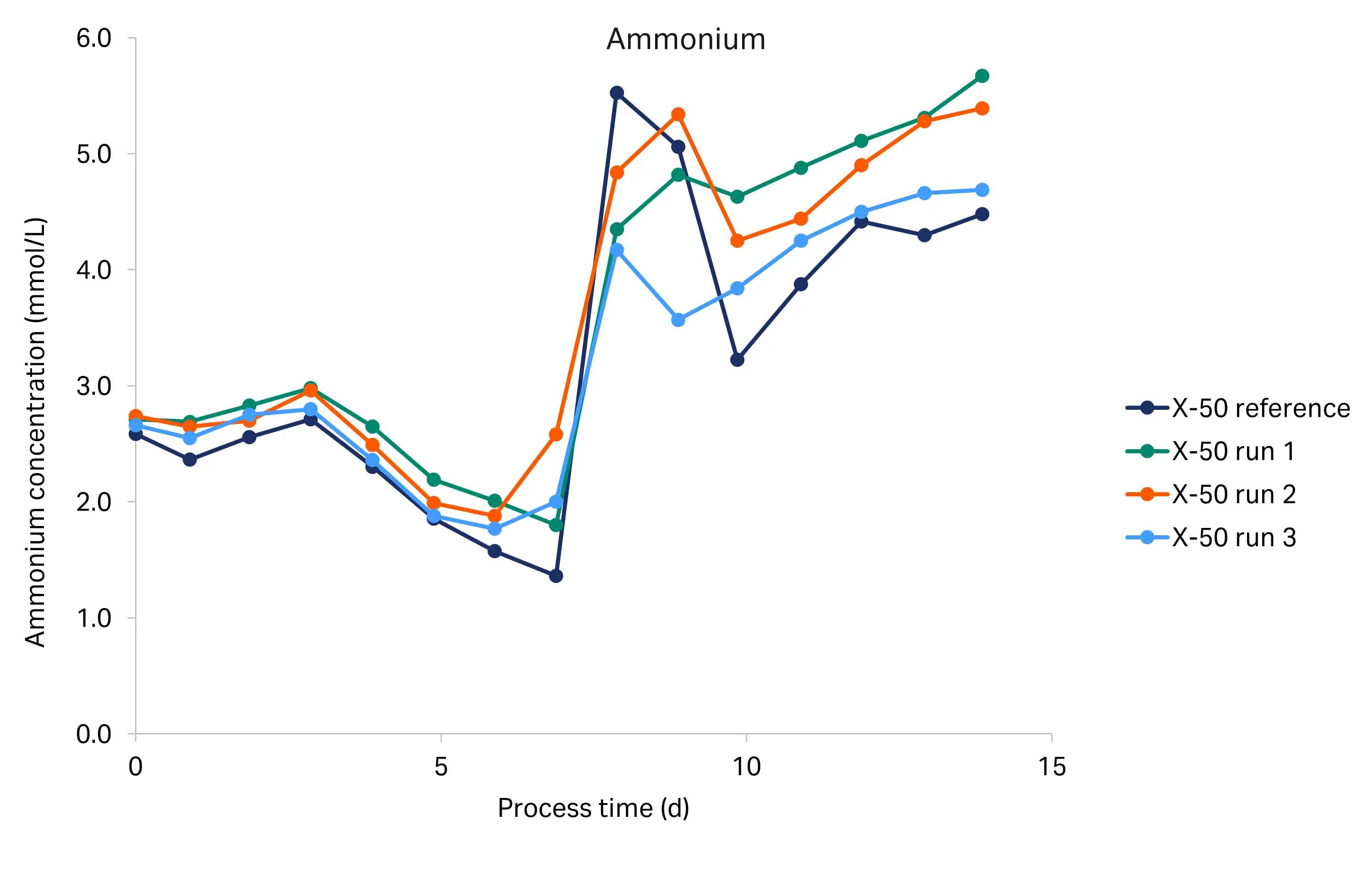
The glucose, lactate, and ammonium profiles from the historical and tech transfer runs are shown in Figure 5. We used the BioProfile Flex 2 cell culture analyzer for the tech transfer runs, and the Cedex Bio HT analyzer for the historical run. The glucose, lactate, and ammonium profiles were comparable between the runs, indicating a high degree of reproducibility. Glucose concentration decreased from 6.0 g/L to below 0.5 g/L at day 6. From day 6 to the end of the culture (day 14), the glucose concentration remained low. Lactate was produced during the initial growth phase and began to drop at day 6, indicating it was metabolized. The ammonium concentration was constant from inoculation to day 3, then decreased until day 7, after which the concentration rapidly increased from around 1.4–2.6 mM up to 4.2–5.5 mM, where it remained until the end of the culture. The rapid increase coincided with glucose and glutamate (data not shown) depletion.
Fig 5. Glucose, lactate and ammonium profiles for the historical X-50 and tech transfer X-50 runs (R1-R3).
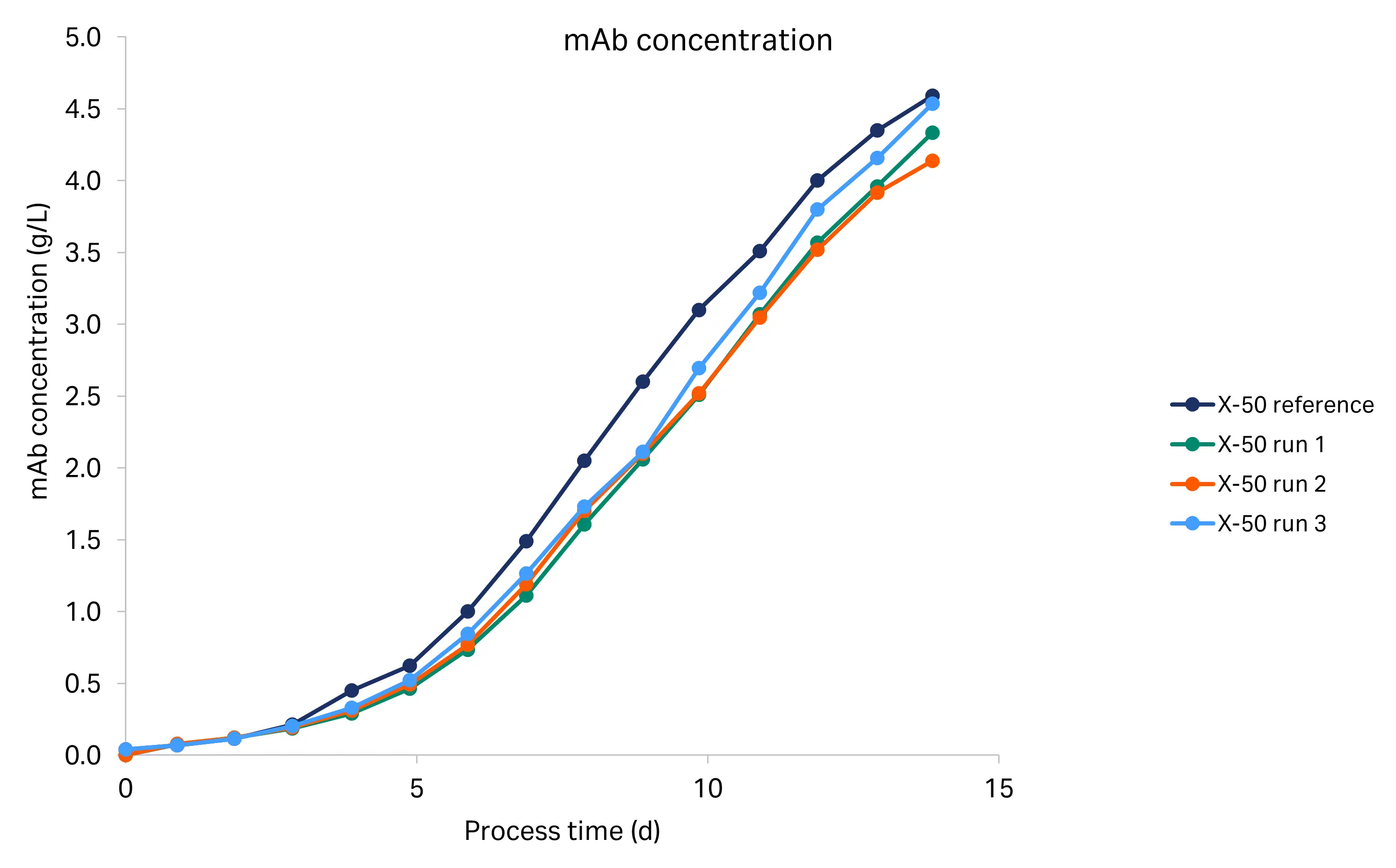
The mAb titers analyzed by HPLC for the tech transfer and historical runs are shown in Figure 6. We used the Cedex Bio HT analyzer for the historical run titer analysis and protein A HPLC for the tech transfer run titer analysis. We observed titers of 4.1–5.1 g/L across all runs, with the harvest titers of the tech transfer runs within 11% that of the historical run.
Fig 6. mAb titer profiles for the historical and three tech transfer X-50 runs (R1-R3).
Summary
We transferred a fed-batch cell culture process in an Xcellerex™ X-50 bioreactor to a second facility using established workflows. The three tech transfer cultures were successfully executed, and the resulting nutrient/metabolite, cell growth, and titer data was comparable across runs, confirming a successful tech transfer. It also highlights consistent performance achieved in different locations (Sweden and UK) and with different operators. Also important to note that we performed the first run at the chosen working volume without the need for benchtop or shake flask cultures to test the process first.
This success was due to the work done upfront, understanding the process, putting together the study plan along with the robustness and reproducibility of the Xcellerex™ X-50 bioreactor. Additionally, with the aid of Chronicle™ automation software, we were able to do this using multiple scientists running the cell culture.
HG902-URP-01
CY51150