A compact and economical solution for maximal virus yield and robust protection of downstream purification steps
Seitz™ V100P depth filter sheets were developed for clarification of virus product feed streams. Here, Seitz depth filter V100P was tested on two live influenza harvests and demonstrated high product yield with high impurity removal capacity. These results show how Seitz™ filters could provide you with an economic filter solution for post-bioreactor feedstreams > 2000 L scale.
Introduction
For most biological drugs, including viral vaccines, removing cells and cell debris after cell culture is a critical step, which impacts the further purification process. An efficient clarification step separates the virus from the cells, cell debris, and many impurities including insoluble precipitants, aggregates, and other materials found in typical cell cultures. This step must combine high capacity for impurity removal, high product yield, ease of scale-up and maximally protect any further downstream operation, making the overall process as efficient and economical as possible.
Cellulose-based depth filters are the first-choice clarification solution for cell culture feed streams, for example, in monoclonal antibody production. With virus and nanoparticle products, however, standard cellulose-based depth filters have been shown to perform suboptimally for some applications, demonstrating particularly low product yields. Here, we demonstrate the use of a special depth filter sheet, grade V100P, based on our established Seitz P-series depth filter sheet range. The V100P grade sheet demonstrates good removal of contaminating particles combined with low retention for viruses, increasing the virus yield post-filtration.
The 0.45 µm filter sheet membrane reduces biorburden and turbidity and protects your downstream purification steps. A throughput above 200 L/m2 for fluids with limited dirt capacity (~ 120 NTU) is expected. For higher turbidities, prefiltration with commercially available depth filters, such as our Seitz™ K 900 P, potentially in a double-layer configuration, is recommended.
Materials and methods
Influenza harvest material
Material A: Influenza virus A/Puerto Rico/8/1934 (H1N1) was produced by infection of human embryonic kidney (HEK)-293 cells as described in reference 1. The material was centrifuged before filtration in the following experiments.
Material B: Influenza virus A/Puerto Rico/8/1934 (H1N1) was produced by infection of Madin-Darby canine kidney (MDCK) cells grown in roller bottles on Glasgow’s minimal essential medium (GMEM) supplemented with 10% fetal calf serum. The material was used without further processing as bulk for the following experiments.
Table 1. Overview and description of filters used
| Filter | Description | Retention rating* | Permeability at 100 kPa (L/m2/min) |
|---|---|---|---|
| Seitz™ V100P depth filter sheet | Cellulose-based depth filter media containing perlite | 2–4 µm | 149 |
| Seitz™ K 100 P depth filter sheet | Cellulose-based depth filter media containing perlite and diatomaceous earth | 1–3 µm | 149 |
| HDC II filter | Polypropylene ‘tapered pore’ high dirt capacity filter | 4.5–1.2-0.6 µm (series) | 70 |
| Fluorodyne™ II grade DBL filter | PVDF bioburden reducing membrane filter | 0.45 µm | 255 |
* For depth filters, it is not possible to talk about an exact retention rate. The given values are estimates only indicate the range.
Table 2. Surface area of different filter formats and recommended operation flow rates
| Surface (cm2) | Flow rate LMH (L/m2/h) | Flow rate (mL/min) | |
|---|---|---|---|
| Supracap™ 50 depth filter capsules | 22 | 100 | 3.6 |
| Membrane disc holder 47 cm | 11 | 100 | 1.8 |
| VELApad™ 60 filter housing |
22 | 100 | 3.6 |
| Supracap™ 100 depth filter capsules | 1000 | 100 | 167 |
Equipment and setup
We carried out the filtration trials on a filtration system as described in Figure 1. A peristaltic pump with compatible tubing, adapted to the required flow rate, pressure sensors, and a weigh balance or graduated cylinder were used.
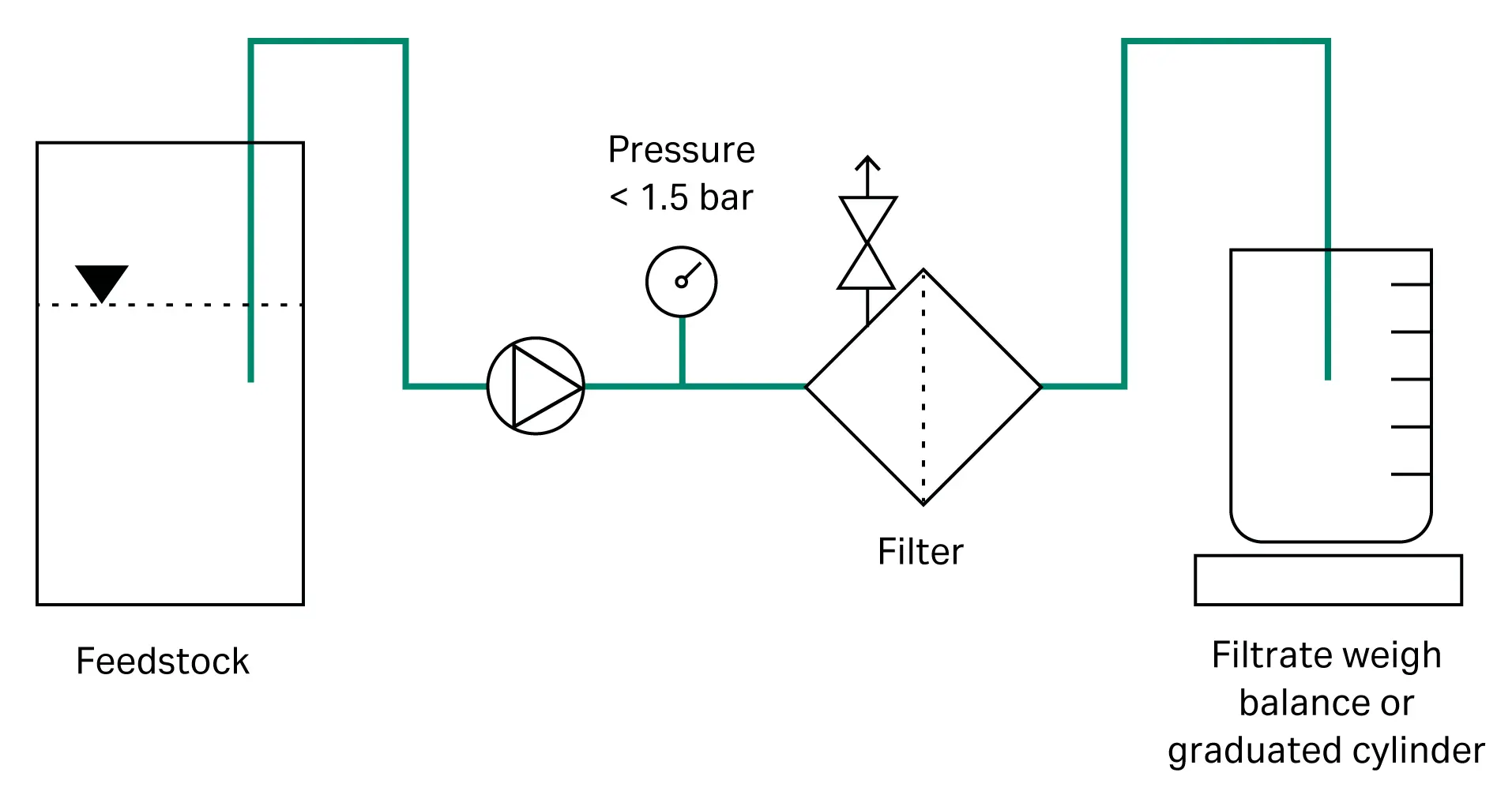
Fig 1. The experimental setup.
Buffers and solutions
Prefiltration and post-filtration, the filters were flushed with PBS buffer, pH 7.4, 15 mS/cm.
Filter installation
We installed the filters according to the setup shown in Figure 1 and the instructions for use issued with the products. Filters were prerinsed with a minimum volume of 50 L/m2 buffer. The capsule was vented during filling by allowing the air to escape through the vent. The vent was tightened as soon as all excess air had escaped the assembly, and the liquid had reached the level of the vent. After rinsing, the filtration device was drained by opening the vent and pouring the excess liquid out.
Filtration and sampling
The filtration unit was filled and vented as described above. During filtration, the flow rate was set to 100 LMH. A sample of the initial feedstock was taken for virus titer determination and turbidity measurement. During filtration, we monitored pressure and turbidity as a function of time, and the filtration stopped when either a pressure of 1.5 bar (7.25 psi, 0.05 MPa) was exceeded, when a significant increase in turbidity was noticed when the feedstock ran out or a predefined volumetric throughput was reached (200 L/m2).
Post-rinsing
Our preliminary trials indicated that the virus yield could significantly increase with a post-filtration buffer rinse step (Fig 2). Therefore, after filtration, the filter capsules were emptied (see Filter installation above) and rinsed with ~ 23 L/m2 buffer (recommended PBS buffer or process-specific buffer). We pooled this rinse with the filtrate and the virus titer was determined on the pool.
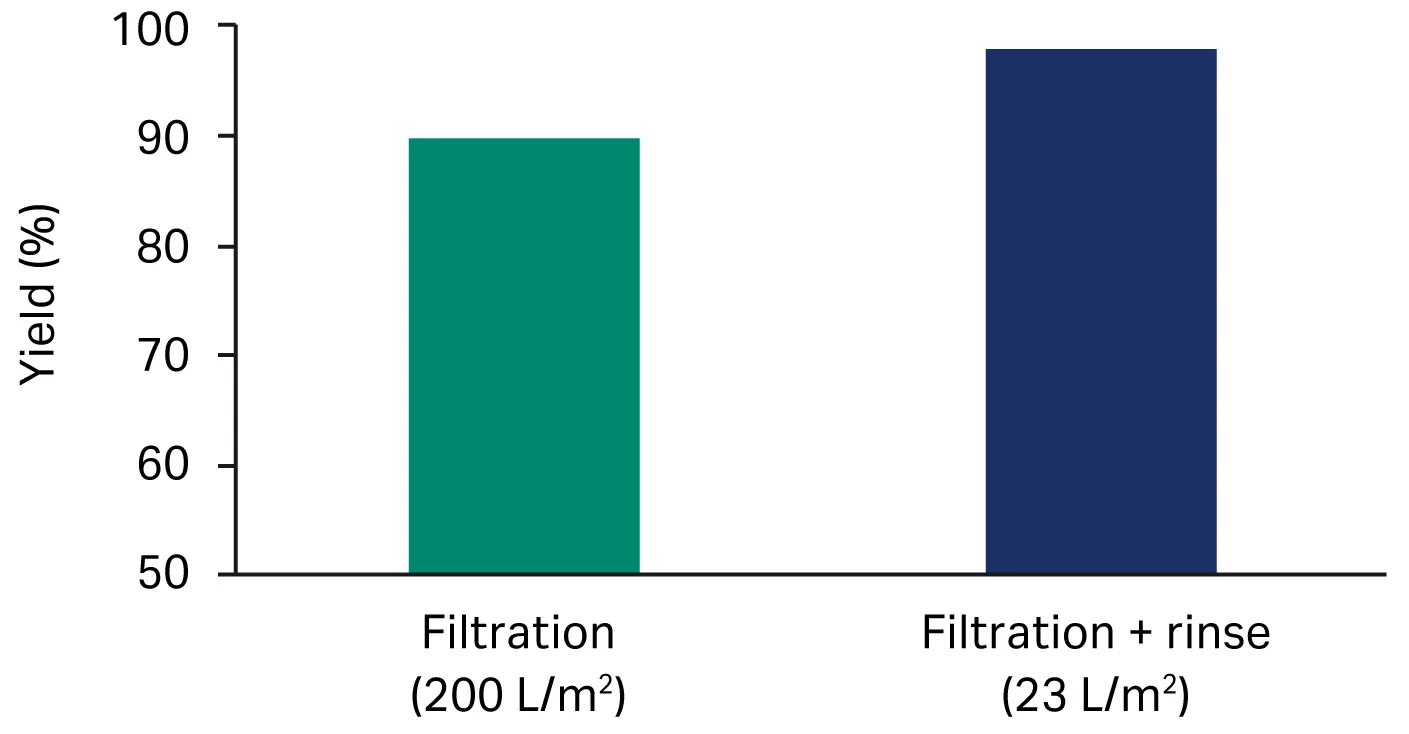
Fig 2. Impact of post-filtration rinse on the overall product recovery for material A on Seitz™ depth filter sheet V100P.
Assays
We quantitated influenza virus using a classical hemagglutination assay on chicken red blood cells. The assay was performed according to the protocol described in reference 2 and Its standard deviation was generally 0.18 log HA units/mL. Turbidity was measured using a Hach 2100P turbidimeter.
Results and discussion
Performance of Seitz™V100P compared to other Cytiva products
Current viral vaccine manufacturing platforms often incorporate a train of polypropylene-based membrane filters with decreasing porosity, to remove the cells. We used a reference train combining HDC II series membranes (4.5 µm + 1.2 µm + 0.6 µm) was used as a benchmark throughout the experiments.
To evaluate its performance, we compared various lots of the depth filter V100P to the HDC II membrane (Fig 3) and to K100P, another cellulose based depth filter with similar permeabilities or retention rates (Fig 4). Overall, depth filter sheet V100P resulted in a virus product yield ranging from as good as, to slightly below the HDC II membrane benchmark. Furthermore, it had a larger volumetric throughput than the benchmark, leading to a compact sizing of the manufacturing scale product and a better turbidity reduction, which translates to better protection of the downstream process steps.
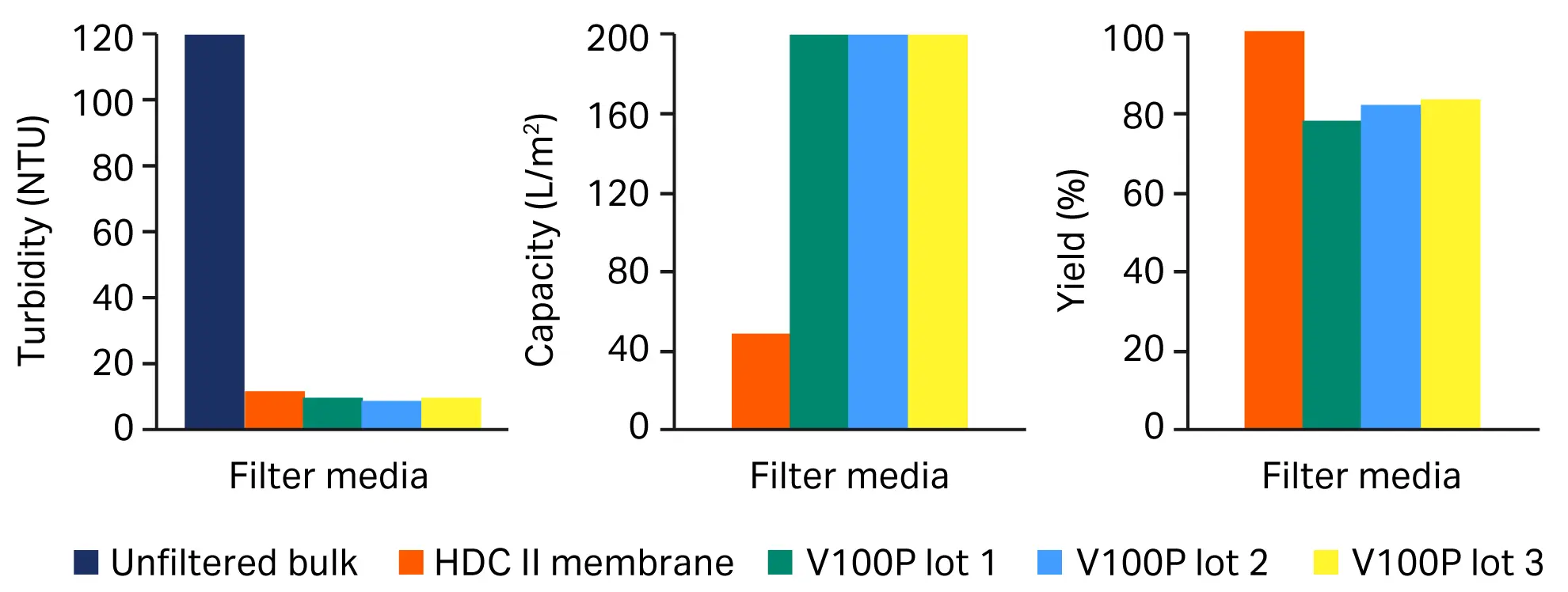
Fig 3. Comparison of performance of different lots of the depth filter sheet V100P vs benchmark filter train (HDC II membrane). (A) turbidity, (B) capacity, (C) yield.
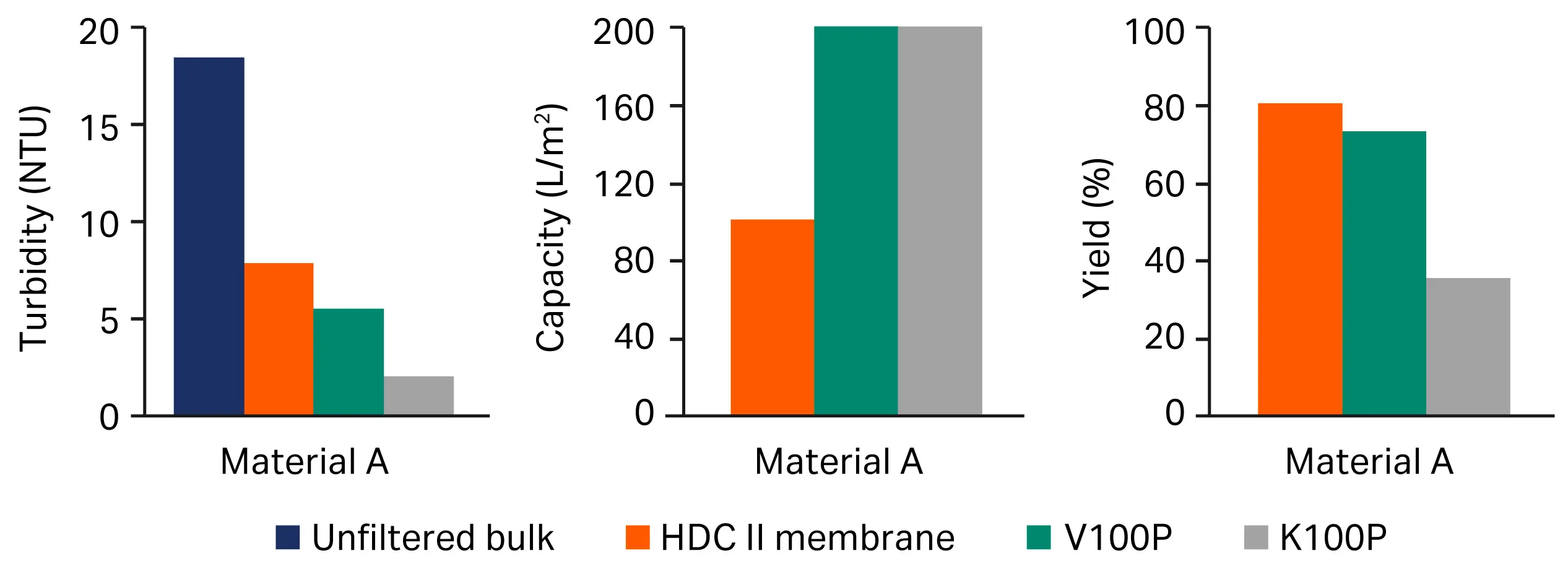
Fig 4. Comparison of performance of the Seitz™ depth filter sheet V 100P vs benchmark of filter train (HDC II membrane) and Seitz™ K 100 P depth-filter membranes. (A) turbidity, (B) capacity, (C) yield.
Scalability of performance
Our range of Supracap™ 50, Supracap™ 100, and Stax™ encapsulated depth-filter modules provide great flexibility and assurance of scalability for process volumes from < 1 to > 1000 L for typical cell-removal applications. The scale-up performance of the range (Fig 5) shows scalability to within ± 15% of capacity (3).
We compared the performance of Seitz™ depth filter V100P using the Supracap™ 100 module (0.1 m2) to that of the scale-down tool Supracap™ 50 capsule (22 cm2). The latter is typically used for initial screening studies and is also the tool we used in the experiments described above. Comparable performance was observed for filtrate turbidity and overall filter capacity (Fig 6). The overall yield was noticeably higher for the higher scale capsule, which could be attributed to either HA assay variation or optimized flow distribution through the larger capsule.

Fig 5. The current range of scalable depth filter tools is available from bench to manufacturing.
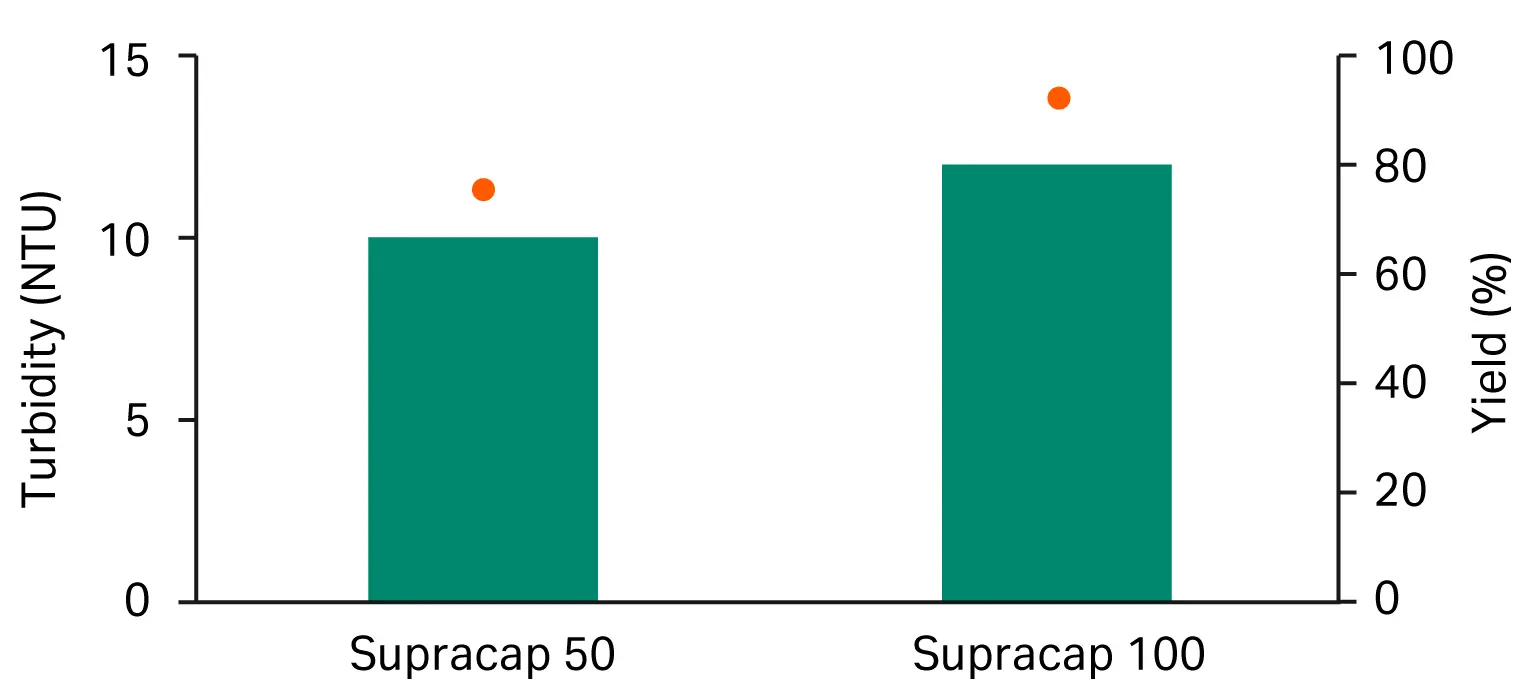
Fig 6. Comparison between filtrate turbidity (bars) and yield (dots) for the scale-down tool, Supracap™ 50 capsule, and the manufacturing tool, Supracap™ 100 capsule.
Batch-to-batch variation: Process robustness
Batch failure rates in the manufacture of biologicals including vaccines are relatively high compared to other industries and are often a consequence of non-robust manufacturing processes. This can lead to a shortage of important biological products as reported by the Food and Drug Administration (FDA) (4).
The performance of depth filter V100P was compared to the reference filter train for three different manufacturing batches of material B. The results in Figure 7 indicate that the V100P filter has a more reliable and reproducible performance and can neutralize some of the upstream variations for the downstream process.
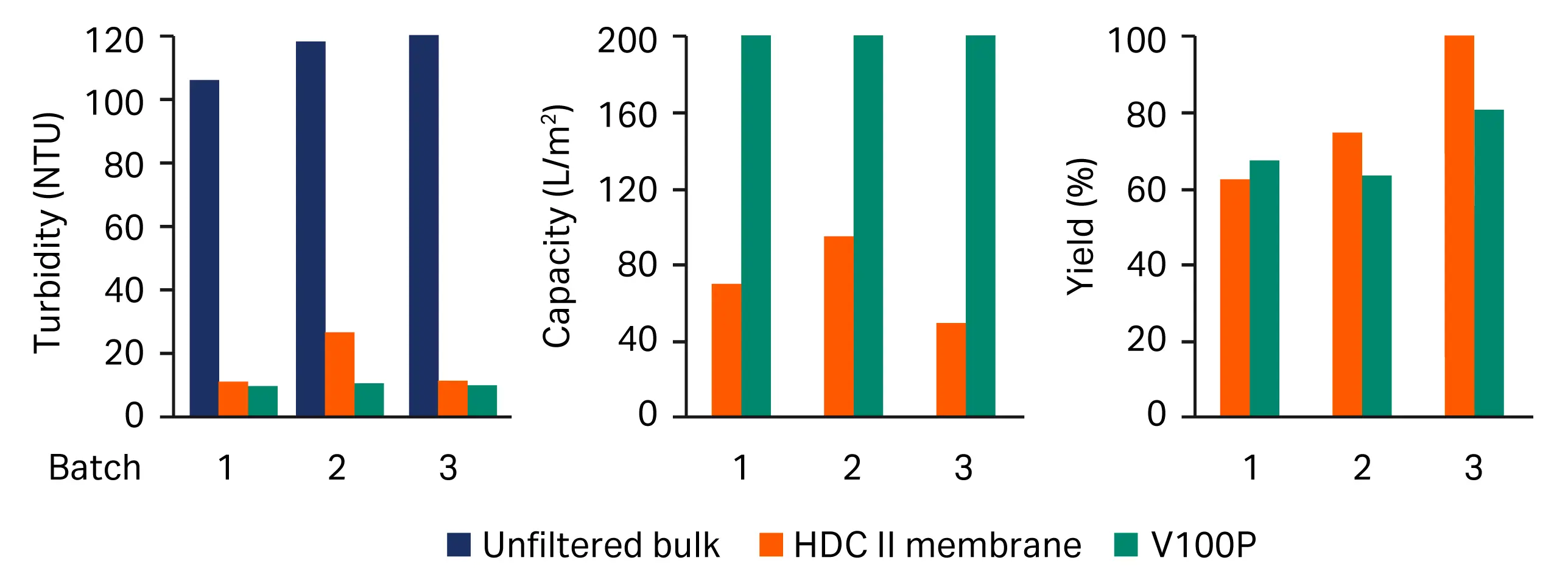
Fig 7. Impact of upstream variations on the cell removal efficiency of Seitz™ depth filter sheet V100P versus benchmark filter train (HDC II membrane). (A) turbidity, (B) capacity, (C) yield.
Protection of downstream processing: Post-clarification filtration on a bioburden-reducing membrane
The impact on the downstream process of the cell removal step using the V100P filter and the related economics was demonstrated using the Fluorodyne™ II grade DBL bioburden-reducing membrane (0.45 µm). We measured the membrane capacity during filtration without and with preceding cell removal on V100P, and found to be at least 10-fold higher, resulting in a 10-fold reduction of the sizing of this filter (Fig 8). Typically, the cost of bioburden-reducing and sterilizing-grade membranes or chromatography products is higher than that of cellulose-based depth-filter products. The overall benefit of implementing such a step is therefore cost reduction, in addition to achieving a more robust overall process.
Fig 8. Impact of a cell removal filtration on Seitz™ V100P on the downstream process.
Conclusion
- The Seitz™ V100P depth filter was tested on two influenza feed materials against current benchmark technologies and demonstrated high product yield with high contaminant capacity, that would provide for a robust depth filter solution for post-bioreactor feed solutions up to 2000 L scale.
- Overall, across the data generated, it is clear there is a balance between increased turbidity reduction and reduced virus yield, probably linked to size exclusion of the virus particles. However, for depth filter sheets with equal permeability, it can be observed that the Seitz™ V100P improves the yield.
References
- Le Ru A, Jacob D, Transfiguracion J, Ansorge S, Henry O, Kamen AA. Scalable production of influenza virus in HEK-293 cells for efficient vaccine manufacturing. Vaccine. 2010 May 7;28(21):3661-3671. doi: 10.1016/j.vaccine.2010.03.029.
- Genzel Y, Behrendt I, König S, Sann H, Reichl U. Metabolism of MDCK cells during cell growth and influenza virus production in large-scale microcarrier culture. Vaccine. 2004 Jun 2;22(17-18):2202-2208. doi: 10.1016/j.vaccine.2003.11.041.
- Collins, M, Lari, H., Anderson, S, Leibnitz, R, Kumar, A. and Kuriyel, R. Investigating flow distribution and its effects on scale-up. Performance of a multicapsule depth filtration system. BioProcess International. 2009 7(9):46-52.
- fda.gov/drugs/drug-safety-and-availability/drug-shortages. Accessed June 2021.
CY49592