Evelyne Nguyen, Cell Line Development Marketing Manager – Viral Vector
Christian Niehus, Molecular Biology and Analytics
Åsa Hagner Mcwhirter, Principal Scientist
Introduction
Adeno-associated virus (AAV)-based vectors show significant promise in treating a wide range of diseases. Alongside other gene transfer vectors, they represent a new and emerging therapeutic paradigm with a potential impact on human health comparable to that of recombinant proteins and vaccines. However, a major challenge for the current pipeline of AAV-based investigational products as they progress through clinical development is the identification, characterization, and control of process- and product-related impurities. Particularly problematic are recombinant adeno-associated virus (rAAV) vector product-related impurities that closely resemble the vector itself and, in some cases, that have no clear standard methods for removal in established biotherapeutic products.
In the production of recombinant adeno-associated virus vectors, cell line development as well as upstream and downstream processes play vital roles in ensuring safety, quality, and efficiency. Cell line development enabled the transition from adherent cell culture to even more scalable production systems utilizing suspension cell lines. The choice of cell line significantly impacts both rAAV vector quality and yield. Through genetic engineering, cell lines can be further optimized to obtain cell substrates with highly favorable rAAV production characteristics. Upstream efforts aim to increase the production of desired capsids (while minimizing the formation of empty or partially filled ones) by optimizing cell culture conditions. On the other hand, downstream processes focus on purifying rAAV by removing impurities. This removal involves several steps, including clarification, ultrafiltration (UF) and diafiltration (DF), capture, and polishing. While the capture step specifically binds vector particles, it currently cannot discriminate between empty and full capsids and is therefore not effective in removing the empty ones.
Types of impurities in recombinant adeno-associated virus products
In this article, we describe the major types of product-related impurities in adeno-associated virus production and their associated safety risks. We also elaborate on the main strategies for reducing and measuring these impurities and describe the Food and Drug Administration’s (FDA's) recommendations. A summary of this information is shown in Table 1.
Table 1. Summary of rAAVs impurities, strategies to control and measure them, and FDA guidelines for their presence in therapeutics.
| Full | Empty | Partial | PTMs | Residual hcDNA | |
|---|---|---|---|---|---|
 |
 |
 |
 |
   |
|
| Definition | Capsids with complete therapeutic transgene | Capsids with no DNA payload | Capsids with truncated transgene | Capsids harboring post translational modifications (glycosylation, phosphorylation, deamidation, and ubiquitination) | Non-encapsidated and encapsidated DNA |
| Method of measurement | qPCR/ELISA; mass photometry; SEC; MALS, analytical AIEX; AUC; cryo-TEM | qPCR/ELISA; mass photometry; SEC; MALS; analytical AIEX; AUC; cryo-TEM; ddPCR | AUC; mass photometry; NGS; ddPCR | Bottom-up mass spectrometry | qPCR; NGS; ddPCR |
| Risks and benefits | N/A | Immune responses | Immune responses; impact not yet fully understood | Impact (both positive or negative) on vector infectivity and packaging efficiency; may dictate systemic host response | Potential immunogenic and oncogenic effects |
| Strategies explored for control | Cell line engineering, reagents, process optimization, downstream optimization (to protect full) | Cell line engineering, upstream optimization, downstream methods such as AIEX, CsCl-gradient ultracentrifugation | Cell line engineering, upstream optimization. Challenge to remove in downstream chromatography, but recent data suggest optimization in the polishing step could reduce partial capsids. | Area still under investigation. | Non-encapsidated hcDNA or pDNA are removed using downstream processes. Cell line engineering is under investigation to reduce encapsidated hcDNA. Some new plasmid technologies with reduced backbone show a reduction in reverse packaging of these sequences. |
| FDA guidelines | N/A | While regulatory guidance does not prescribe specific thresholds, it is expected that the sponsor defines an appropriate threshold as part of establishing the critical quality attributes (CQAs), which is then used to ensure batch-to-batch consistency. | Similar to empty capsids | Existing guidelines on PTMs but for therapeutic protein in general and not specifically applied for rAAVs; Aim at derisking aggregation | Total residual DNA from non-tumorigenic cells to be less than 10 ng/dose; additional testing for transformed tumorigenic cells such as E1 testing for HEK293 cells; plus product control strategy for potentially encapsidated plasmid DNA |
Empty capsids
Empty adeno-associated virus particles are similar to the desired product but lack a DNA payload. These empty particles can be generated in high levels, commonly 40% to 80% of the total AAV particles in cell culture harvest. If not removed, final vector preparations can contain an excess of empty capsid particles and have undesired effects in the patient. Variation in percent full capsids between production batches may lead to high lot-to-lot variability.
Empty capsids add to the overall vector load during clinical dosing, and higher vector loads can intensify capsid-triggered immune responses. It has been observed that there is a correlation between adverse events and increased vector load (1) in clinical trials for AAV-based therapeutics. In addition, therapeutic rAAVs are dosed based on vector genome (vg) titer, so while non-functional empty capsids increase the vector load, they do not enhance clinical efficacy (2). From a regulatory standpoint, manufacturers must account for the ratio of empty capsids to full capsids. This requirement is outlined in FDA guidance, which does not specify a precise threshold. Instead, manufacturers are expected to define their own critical quality attributes (CQAs) to characterize their product and establish risk-based acceptable percentage ratios of full-to-empty capsids in their batches (3).
Removing empty capsids is challenging due to their structural similarity to the desired vector product. Two-step approaches need to be considered when looking to reduce empty capsids. The first approach involves cell line engineering. For instance, by understanding and optimizing the packaging mechanism, it may be possible to reduce the number of empty capsids. Upstream process optimization may also reduce empty capsids (4). But even with an optimized upstream process, the proportion of full capsids in the harvested material is usually below 60%. Therefore, the second step is a polishing step that is part of the downstream process. Traditionally, CsCl-gradient ultracentrifugation—which separates capsids based on density—has been used to purify full rAAV vector capsids at small scale. But this method doesn’t scale up well. On the other hand, anion exchange chromatography (AIEX) offers scalability, making it suitable for large-scale production (5). This method utilizes the small difference in isoelectric points (pI) between full and empty capsids to achieve separation.
Measuring the proportion of empty versus full capsids in rAAV production often utilizes quantitative polymerase chain reaction (qPCR) combined with enzyme-linked immunosorbent assay (ELISA). Quantitative PCR measures vg titer whereas ELISA is used to measure the total viral particles (vp) titer. The ratio can then be obtained by dividing the number of vg by vp. However, using a ratio between two assays—each with its own inherent variability—risks amplifying overall variation. It is recommended to use at least one orthogonal method to complement and confirm the qPCR:ELISA results. Mass photometry, size exclusion chromatography (SEC), in combination with UV detection or multiangle light scattering (MALS), and analytical AIEX are commonly used, but analytical ultracentrifugation (AUC) (6) and cryogenic transmission electron microscopy (cryo-TEM) are the gold standard techniques to determine percent full capsids.
Partially filled genome capsids
Partially filled genome capsids have their DNA payload truncated and present significant challenges similar to those posed by empty capsids. Truncated gene of interest (GOI) species lack the complete genetic sequence necessary for proper therapeutic function, leading to inefficiencies (7) in gene delivery. High levels of truncated GOI products, which can constitute a substantial portion of the total recombinant adeno-associated virus particles, result in variability and reduced efficacy of the final vector preparations.
The impact of partial GOI genomes on the safety and efficacy of rAAV products is not well understood. Their presence in clinical preparations may pose similar risks to those associated with empty capsids, particularly if they are nonfunctional (8). Even though no hard criteria have been defined by the FDA, the recommendation is to investigate and compare batches because partial genome capsids are still considered as impurities (3).
Due to the high diversity of truncated versions, removing truncated genome capsids using current downstream purification methods is extremely challenging. Therefore, the primary focus will be on cell line engineering and upstream process development to minimize truncation occurrences. For instance, optimizing the coding sequence of the GOI can reduce the likelihood of truncation events.
To measure partial capsids, AUC (9) is the method of choice as it can differentiate empty particles from full and partials. Next-generation sequencing (NGS) and ddPCR are PCR-based methods that can also provide deeper insights into genome integrity and detect chimeric or truncated sequences. Mass photometry is gaining traction as it offers higher throughput, requires less sample while still being GOI and serotype agnostic. This technique utilizes interference of light scattering from individual particles to determine their masses, enabling the distinction between empty, full, and partially filled capsids.
PTMs on capsids
Post-translational modifications (PTMs) on adeno-associated virus capsids are an emerging area of interest in gene therapy research. All AAV capsids inherently contain post-translational modifications (PTMs). It is essential to ensure that the PTM profile does not adversely impact biological activity or downstream processing. These modifications, which include glycosylation, phosphorylation, deamidation, and ubiquitination, can significantly impact the properties and performance of rAAV vectors such as vector infectivity and packaging efficiency and may dictate systemic host response (10).
PTMs on therapeutic proteins can elicit immune responses by inducing aggregates (11) (a known issue for high concentration rAAV vectors and for certain serotypes), altering stability or functional activity, or modifying antigen processing and presentation (12). Glycosylation, for instance, is a particularly strong modulator of immunogenicity (13). In addition, it has been observed that producing rAAV vectors in insect cells like Spodoptera frugiperda can pose immunotoxicity risks (14). Nonmammalian N-glycans can trigger allergic responses in humans. In contrast, rAAV vectors produced in human cells are more potent and safer, as they avoid these immune reactions and enhance product solubility. There is still much to learn about the effects of PTMs on capsids, and it may be possible to fine-tune these modifications to enhance viral infectivity or packaging efficiency. The FDA has existing guidelines when reviewing other therapeutic protein products, and while these guidelines do not yet apply to rAAV vectors, they provide insight into potential areas of focus as the scientific community learns more about the impact of these chemical modifications on AAV vectors (15, 16). The aim is to ensure that biological therapeutic products are safe, effective, and similar to their reference products in terms of PTMs.
Few studies have explored PTMs on capsids, and characterizing these PTMs remains challenging due to the limited number of techniques available and the specific technical expertise required. Bottom-up mass spectrometry has been the predominant method for their characterization thus far (10, 12).
Residual hcDNA and pDNA
In addition to empty capsids and truncated GOI, residual host cell DNA (hcDNA) and plasmid DNA (pDNA) impurities are also a source of concern and constitute a particular two-part problem in rAAV preparations where they might get carried over outside and inside of the viral capsid. Unpackaged host cell components like DNA, RNA, proteins, and lipids are called process-related impurities, and they can be effectively removed by conventional downstream process purifications (capture and polishing). Product-related impurities on the other hand, involve host cell or plasmid backbone components that are incorporated in the viral capsid and are therefore intrinsically resistant to classic purification methods. Among these encapsidated impurities hcDNA and pDNA represent the most problematic stowaways. Encapsidated hcDNA might arise when fragments of host cell genomic DNA, rather than the virus genome, are unintentionally enclosed into virus particles.
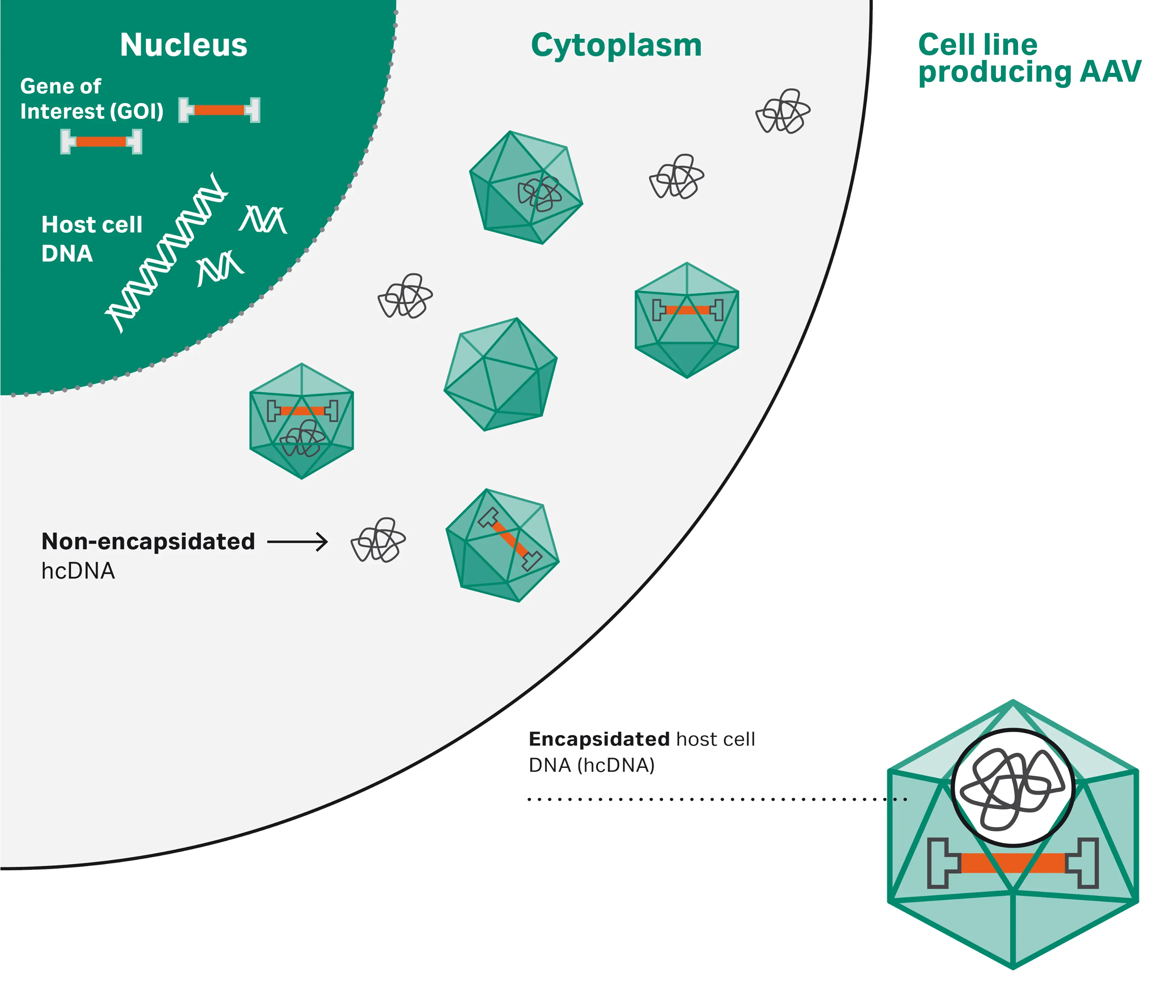
Fig 1. Formation of encapsidated and non-encapsidated hcDNA
The presence of encapsidated hcDNA in rAAV vectors poses significant risks including potential immunogenic and oncogenic effects (17, 18). Encapsidated hcDNA can integrate into the host genome, potentially disrupting tumor suppressor genes and leading to oncogenesis. To mitigate the risks related to foreign DNA inside viral vectors, the FDA recommends stringent control measures and risk assessments. The FDA advises that hcDNA levels should be minimized and that the level of residual DNA from cell substrates should be less than 10 ng per dose (19) with a length shorter than 200 bp.
Residual DNA impurities are to be characterized by PCR-based techniques such as NGS and ddPCR (20). Following vector particle purification, the capsids are digested, and PCR methods are employed to quantify host cell DNA by amplifying sequences specific to the host cell. To further characterize residual hcDNA, NGS (preferably long-read techniques) is performed on DNA from purified particles to further inform risk assessments.
Few strategies have been published on reducing plasmid DNA and host-genome cell line contaminants encapsidated. This lack indicates that our understanding of these specific contaminants is still limited, and much work remains to be done to fully comprehend this phenomenon before effective reduction strategies can be applied. For example, some new plasmid technologies with reduced backbone demonstrated a reduction in reverse packaging of these sequences (21).
Conclusion
In conclusion, to effectively reduce recombinant adeno-associated virus vector impurities, it is essential to focus on cell line development, upstream processes, and downstream processes. While the downstream development and processes for rAAV vector production have been well established and understanding is robust, there is still significant work to be done in optimizing the upstream processes and developing high quality production cell lines. Upstream efforts and cell line engineering are crucial for minimizing the formation of unwanted capsid species and are more tightly linked to vector/host interactions. There is still much to learn and understand about the molecular mechanisms and conditions needed to achieve reduction of all contaminants. Also, the development of advanced analysis methods to fully characterize the rAAV vectors will be key in the future. By continuing to advance knowledge and techniques in these areas, the overall quality and efficacy of rAAV vectors can be improved, paving the way for more effective gene therapies.
Related content
Related services
References
- Kishimoto TK, Samulski RJ. Addressing high dose AAV toxicity – ‘one and done’ or ‘slower and lower’? Expert Opin Biol Ther. 2022;22(9):1067-1071. doi:10.1080/14712598.2022.2060737
- McColl-Carboni A, Dollive S, Laughlin S, et al. Analytical characterization of full, intermediate, and empty Aav capsids. Gene Ther. 2024;31(5-6):285-294. doi:10.1038/s41434-024-00444-2
- Guidance for Industry Q8(R2) pharmaceutical development. 2.3 Risk Assessment: Linking Material Attributes and Process Parameters to Drug Product CQAs. US Food and Drug Administration. November 2009. Accessed July 24, 2025. https://www.fda.gov/media/71535/download.
- Coronel J, Patil A, Al-Dali A, Braß T, Faust N, Wissing S. Efficient production of Raav in a perfusion bioreactor using an ELEVECTA® stable producer Cell Line. GEN. May 1, 2024. Accessed July 24, 2025. https://www.genengnews.com/sponsored/efficient-production-of-raav-in-a-perfusion-bioreactor-using-an-elevecta-stable-producer-cell-line/.
- Heldt CL, Skinner MA, Anand GS. Structural changes likely cause chemical differences between empty and full AAV capsids. Biomedicines. 2024;12(9):2128. doi:10.3390/biomedicines12092128
- Wagner C, Fuchsberger FF, Innthaler B, Lemmerer M, Birner-Gruenberger R. Quantification of empty, partially filled and full adeno-associated virus vectors using mass photometry. Int J Mol Sci. 2023;24(13):11033. doi:10.3390/ijms241311033
- Enrique B, Marta B. Efficacy, effectiveness and efficiency in the health care: The need for an agreement to clarify its meaning. International Archives of Public Health and Community Medicine. 2020;4(1). doi:10.23937/2643-4512/1710035
- McColl-Carboni A, Dollive S, Laughlin S, et al. Analytical characterization of full, intermediate, and empty Aav capsids. Gene Ther. 2024;31(5-6):285-294. doi:10.1038/s41434-024-00444-2
- Wagner C, Fuchsberger FF, Innthaler B, Lemmerer M, Birner-Gruenberger R. Quantification of empty, partially filled and full adeno-associated virus vectors using mass photometry. Int J Mol Sci. 2023;24(13):11033. doi:10.3390/ijms241311033
- Mary B, Maurya S, Arumugam S, Kumar V, Jayandharan GR. Post?translational modifications in capsid proteins of recombinant adeno?associated virus (aav) 1?RH10 serotypes. FEBS J. 2019;286(24):4964-4981. doi:10.1111/febs.15013
- Center for Drug Evaluation and Research. Immunogenicity assessment for therapeutic protein products. U.S. Food and Drug Administration. August 2014. Accessed July 24, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/immunogenicity-assessment-therapeutic-protein-products.
- Rumachik NG, Malaker SA, Paulk NK. VectorMOD: Method for bottom-up proteomic characterization of Raav capsid post-translational modifications and vector impurities. Front. Immunol. 2021;12. doi:10.3389/fimmu.2021.657795
- Malaker SA, Penny SA, Steadman LG, et al. Identification of glycopeptides as posttranslationally modified Neoantigens in leukemia. Cancer Immunol Res. 2017;5(5):376-384. doi:10.1158/2326-6066.cir-16-0280
- Altmann F. The role of protein glycosylation in allergy. Int Arch Allergy Immunol. 2006;142(2):99-115. doi:10.1159/000096114
- Center for Drug Evaluation and Research. Development of therapeutic protein biosimilars: Comparative analytical assessment and other quality-related considerations guidance for industry. U.S. Food and Drug Administration. May 2019. Accessed July 24, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/development-therapeutic-protein-biosimilars-comparative-analytical-assessment-and-other-quality.
- Center for Drug Evaluation and Research. Quality considerations in demonstrating biosimilarity of a therapeutic protein product to a reference product guidance for industry. U.S. Food and Drug Administration. April 2015. Accessed July 24, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/quality-considerations-demonstrating-biosimilarity-therapeutic-protein-product-reference-product.
- Wang Z, Troilo PJ, Wang X, et al. Detection of integration of plasmid DNA into host genomic DNA following intramuscular injection and electroporation. Gene Ther. 2004;11(8):711-721. doi:10.1038/sj.gt.3302213
- Sheng L, Cai F, Zhu Y, et al. Oncogenicity of DNA in vivo: Tumor induction with expression plasmids for activated H-Ras and C-myc. Biologicals. 2008;36(3):184-197. doi:10.1016/j.biologicals.2007.11.003
- Guidance for industry: Chemistry, Manufacturing, and Control (CMC) Information for Human Gene Therapy Investigational New Drug Applications (INDs). U.S. Food and Drug Administration. January 2020. Accessed July 24, 2025. https://www.fda.gov/media/113760/download.
- Developing potential cellular and gene therapy products. U.S. Food and Drug Administration. November 2024. Accessed July 24, 2025. https://www.fda.gov/media/183631/download.
- Williams JA, Paez PA. Improving cell and gene therapy safety and performance using next-generation Nanoplasmid vectors. Mol Ther Nucleic Acids. 2023;32:494-503. doi:10.1016/j.omtn.2023.04.003