Introduction
Both Sera-Mag™ speedbead carboxylate-modified 3 μm magnetic particles and Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles can be used as a solid support in immunoassays. They provide high binding capacity, high signal to noise ratio, colloidal stability, and slow gravity settling.
Magnetic particle binding capacity is important to understand and to ensure the analyte of interest can be captured. In the case of streptavidin particles, the binding capacity is related to the amount of biotin that can bind to the streptavidin on the magnetic particles. However, steric interactions with the biotinylated capture molecule, typically immunoglobulin antibodies (IgG), can also impact the binding capacity. Therefore, we used a competitive binding assay to evaluate the relative binding capacity of the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles to biotin-IgG in addition to three other commercially available 3 μm magnetic particles. The performance of the different magnetic particles was also evaluated using a commercially available full sandwich immunoassay for the detection of sex hormone binding globulin (SHBG). In both assay formats, a high signal was achieved using the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles.
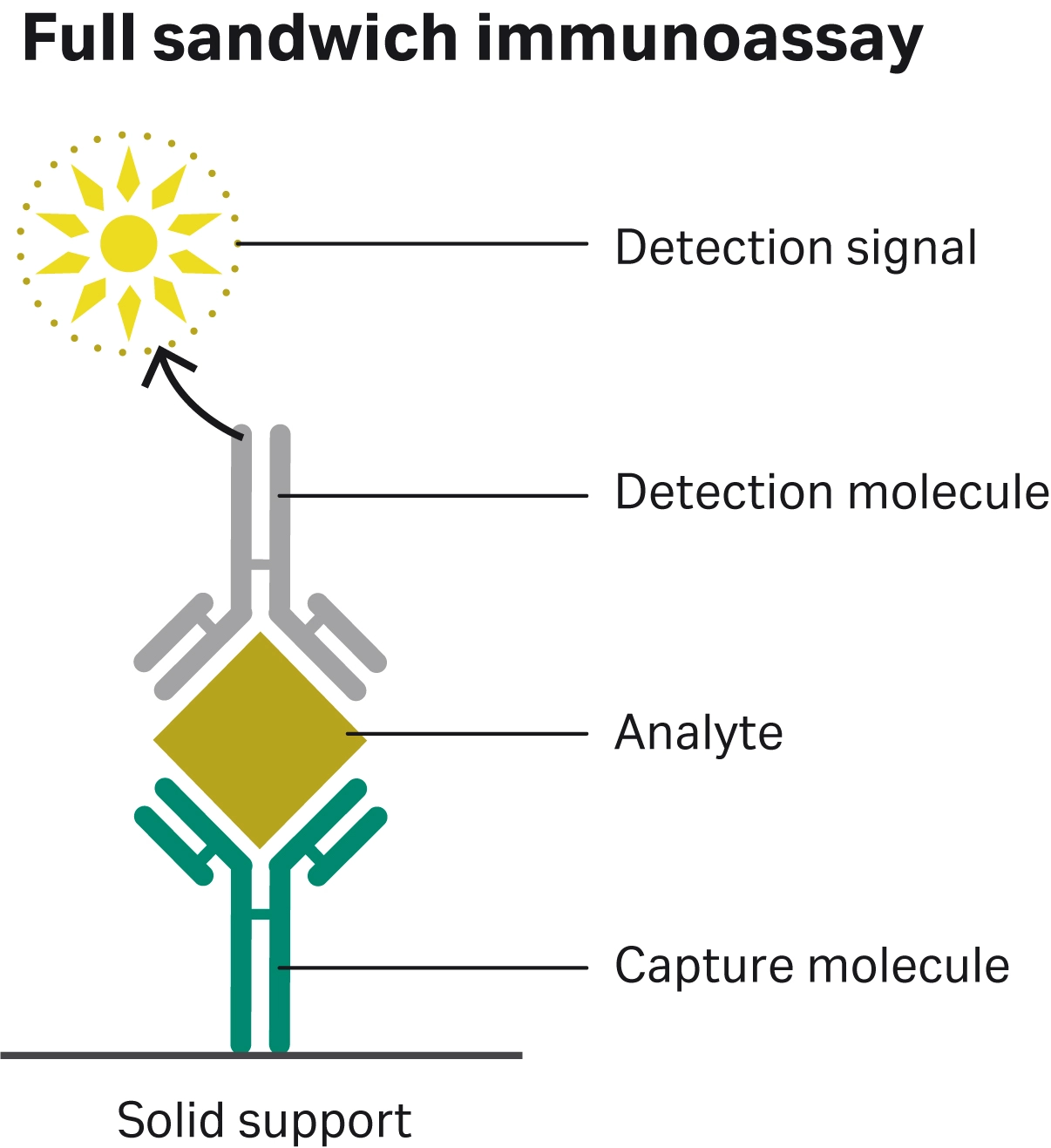
Fig 1. Full sandwich immunoassay schematic depicting the components of an assay: solid support, capture molecule, analyte of interest, detection molecule, and detection signal.
Materials and methods
Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles and three commercially available 3 μm magnetic particles: Dynabead M270 Streptavidin (Thermo Fisher Scientific), Dynabead M280 Streptavidin (Thermo Fisher Scientific), Magnosphere MS300/Streptavidin (JSR Life Sciences) were used in this study. All chemiluminescence assays were completed using the IDS-iSYS Multi-Discipline Automated System (Immunodiagnostic Systems).
Relative biotin-IgG binding capacity assay
A biotin-IgG binding capacity assay was developed to evaluate the relative amount of biotin-IgG binding capacity of the magnetic particles. The biotin-IgG was labelled with acridinium ester (AE), a common detection label used in chemiluminescence in order to assess the biotin-IgG bound.
The binding capacity was evaluated using a competitive assay format to stay within the limit of detection of the IDS-iSYS Multi-Discipline Automated System. For all conditions, a constant amount of 3 ng per test of labelled biotin-IgG-AE was used and mixed with a titration of unlabelled biotin-IgG at 0 ng, 6 ng, 100 ng, 300 ng, 600 ng, 1000 ng, and 1500 ng per test. The mixture of biotin-IgG-AE and biotin-IgG was then added to the beads and incubated for 15 minutes (min). The change in AE signal as the amount of unlabelled biotin-IgG increased in the mixture was then monitored to determine the relative binding capacity of the beads tested (Fig 2). All conditions were tested with 32.5 μg of particles per test using a buffer of PBS, 0.02% antifoam, and 1% Pluronic F-127.
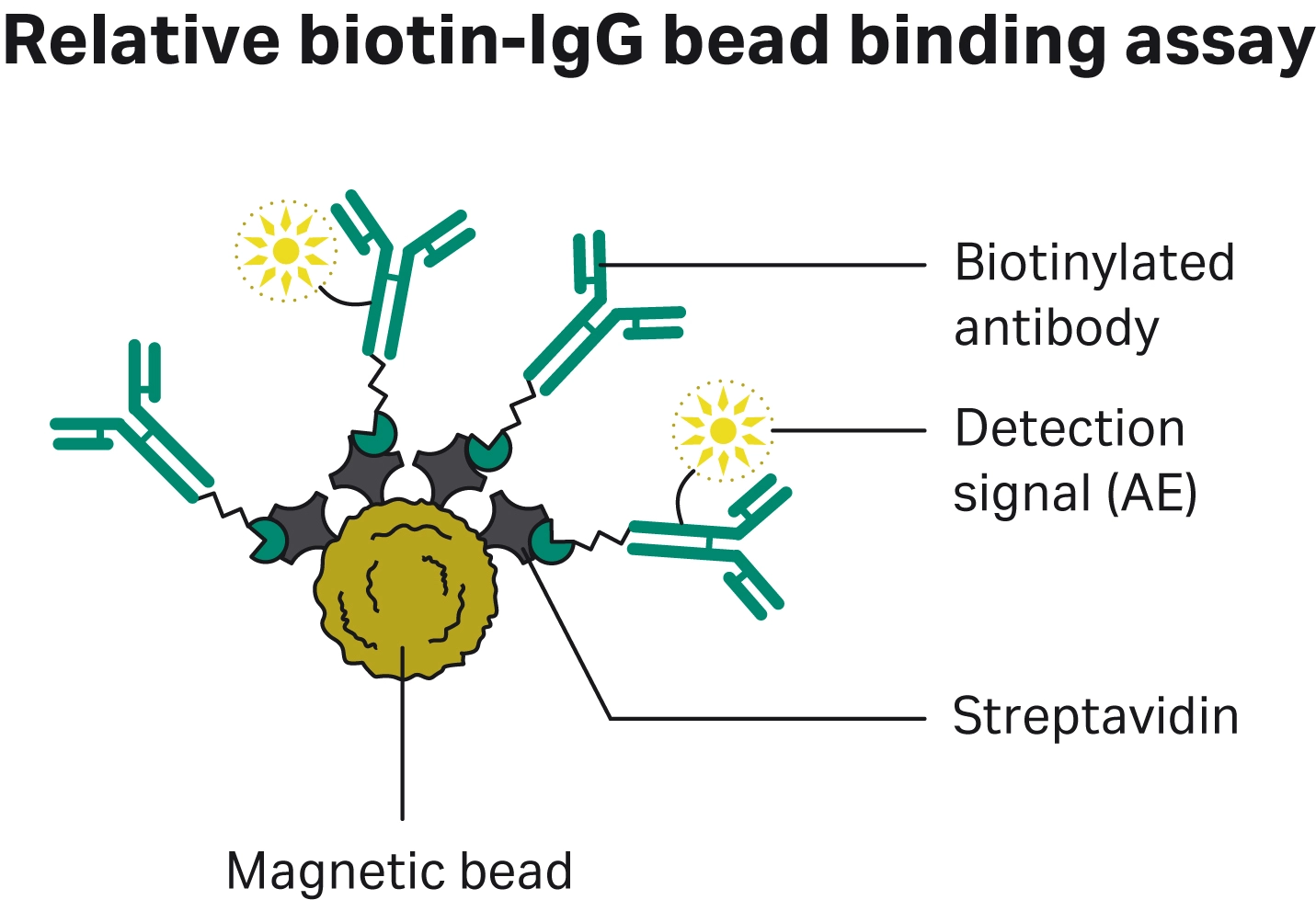
Fig 2. Schematic of biotin-IgG binding capacity where a competitive assay with a mixture of biotin-IgG and biotin-IgG-AE is used to determine the relative binding capacity of different streptavidin magnetic particles.
SHBG full sandwich immunoassay
The magnetic particles were also evaluated in an automated chemiluminescence immunoassay (CLIA) using IDS SHBG assay for the measurement of sex hormone binding globulin (SHBG). SHBG is a glycoprotein responsible for the transport of testosterone and oestradiol and its levels can be used to diagnose androgen imbalance, among other medical conditions.
The measurement of SHBG in 10 different serum samples obtained from Biomex GmbH (Germany) was assessed for each magnetic particle. The magnetic particles provided with the SHBG kit were replaced with the 3 μm magnetic particles evaluated in this study. For the initial evaluation all conditions were evaluated at the same magnetic particle concentration of 2 mg/mL. All magnetic particles were washed three times prior to addition into the assay to wash out the bead storage buffer.
Results
Relative biotin-IgG binding capacity
The relative biotin-IgG binding capacity of the magnetic particles was evaluated using a typical competitive immunoassay format to stay within the limit of detection of the IDS-iSYS Multi-Discipline Automated System (3). The competition was achieved by mixing a constant amount of labelled biotin-IgG and increasing amounts of unlabelled biotin-IgG and then adding this mixture to the magnetic particles (see Materials and methods). Therefore, a slower drop in signal with increasing unlabelled biotin-IgG in the mixture indicated a higher relative binding capacity of the magnetic particles (Fig 3).
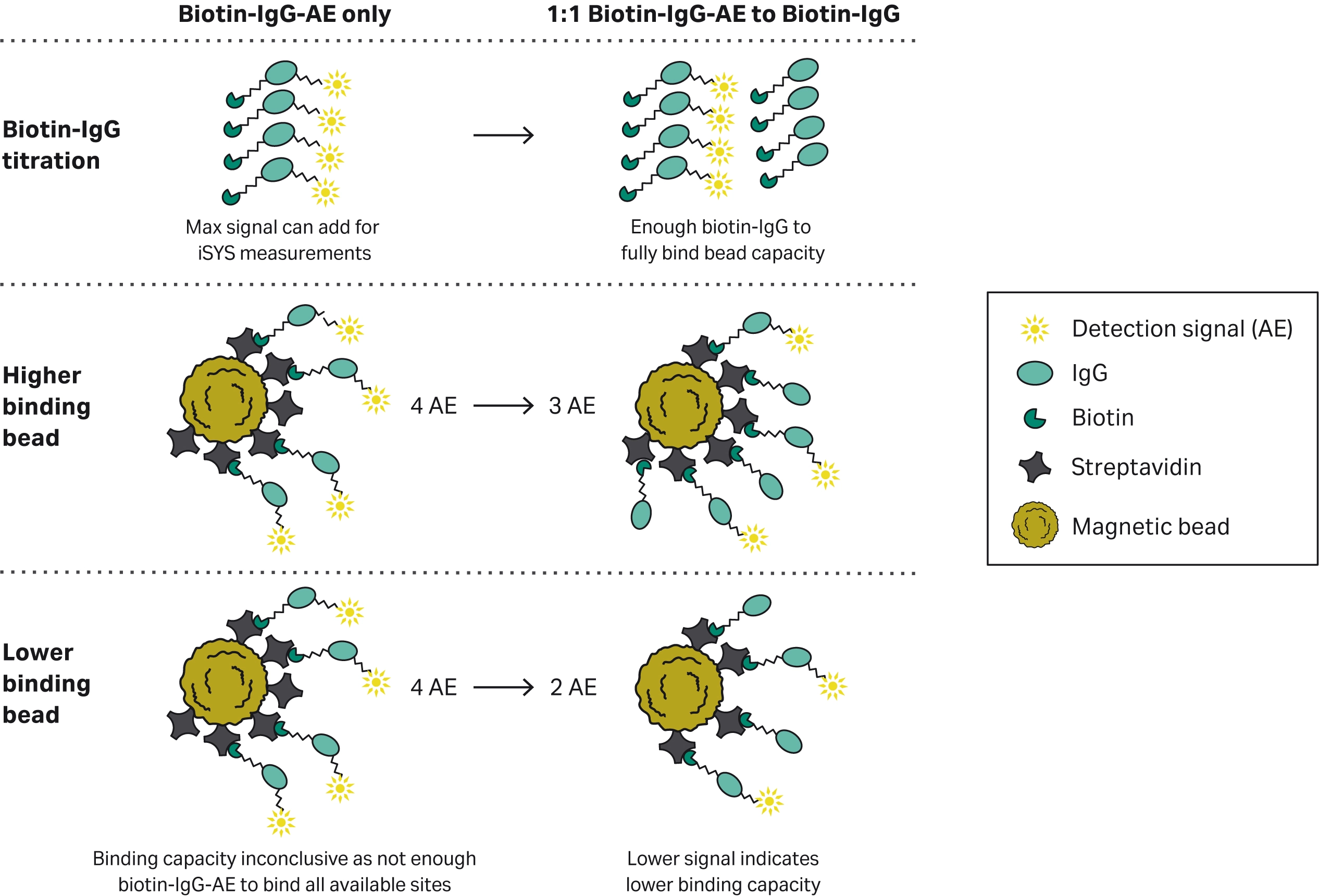
Fig 3. Schematic of the signal output of the competitive biotin-IgG binding capacity assay. In order to stay within the limit of detection of the IDS-iSYS Multi-Discipline Automated System a competitive assay was used to determine the relative binding capacity. A set amount of 3 ng/test of labelled biotin-IgG-AE was used to provide signal. Increasing amounts of unlabelled biotin-IgG was then added to ensure all available binding sites were used. Beads with a lower binding capacity would therefore see a faster drop in signal at lower concentrations of unlabelled biotin-IgG concentrations.
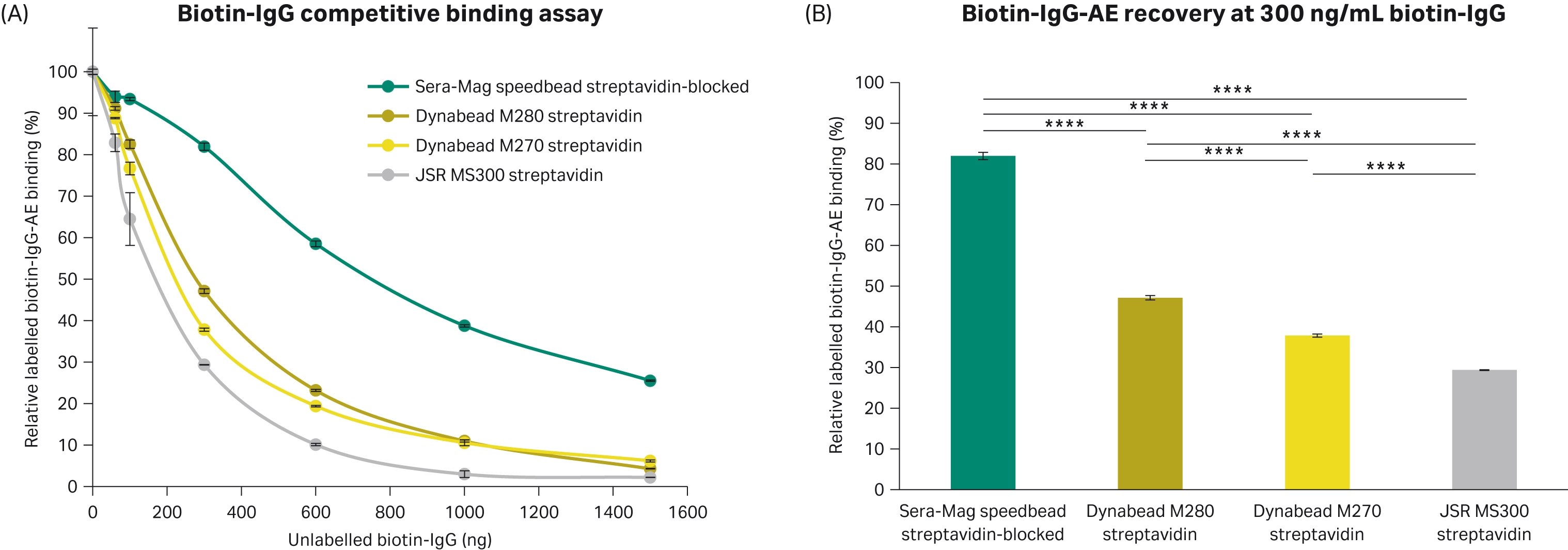
This 1-step biotin-binding assay, showed the labelled biotin-IgG-AE was displaced at a slower rate with increasing amounts of unlabelled biotin-IgG for the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles (Fig 4).
Fig 4. Relative biotin-IgG binding of 3 μm streptavidin magnetic particles. (A) Labelled biotin-IgG-AE was displaced when increasing amounts of unlabelled biotin-IgG was added to the 3 μm streptavidin magnetic particles. (B) Percentage of labelled biotin-IgG-AE bound when 300 ng/mL biotin-IgG was added relative to when no biotin-IgG-AE was added. All conditions completed in triplicate (n = 3). Error bars represent standard deviation. Statistical comparisons completed using an ANOVA with Tukey’s pairwise post hoc test (**** p<0.0001).
To further understand the relative binding capacities, logarithmic regression models were fit to the biotin-IgG titration for each bead. It was predicted it would take more than double the amount of biotin-IgG to displace 50% of the labelled biotin-IgG-AE when using Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles. Good fits were achieved with R2 values of 0.91, 0.98, 0.99, and 0.96 for the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles, Dynabead M270 Streptavidin, Dynabead M280 Streptavidin, and JSR MS300 streptavidin magnetic particles, respectively. Using the equations for the fitted lines, the amount of bioting-IgG to displace 50% biotin-IgG-AE was calculated. The percentage of biotin-IgG required was calculated relative to Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles as an indication of the relative biotin-IgG binding capacities.
Table 1. Calculated predicted biotin-IgG required to displace 50% of biotin-IgG-AE. Logarithmic regressions were fit to the biotin-IgG binding assay results
| 3 μm magnetic particles | Calculated biotin-IgG to displace 50% biotin-IgG-AE (ng) | Biotin-IgG to displace 50% biotin-IgG-AE relative to SeraMag™ (%) |
| Sera-Mag™ speedbeads streptavidin-blocked | 695 | 100 |
| Dynabead M270 streptavidin | 270 | 34 |
| Dynabead M280 streptavidin | 236 | 38 |
| JSR MS300 streptavidin | 173 | 25 |
These results indicate Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles had a higher biotin-IgG binding capacity compared to Dynabead M270 Streptavidin, Dynabead M280 Streptavidin, and JSR MS300 Streptavidin magnetic particles in this assay. Higher biotin-IgG binding of the magnetic particles could enable more analyte to be captured per magnetic particle or increase the chances of capturing analytes at low concentration levels.
Quantification of SHBG in Full Sandwich Immunoassay
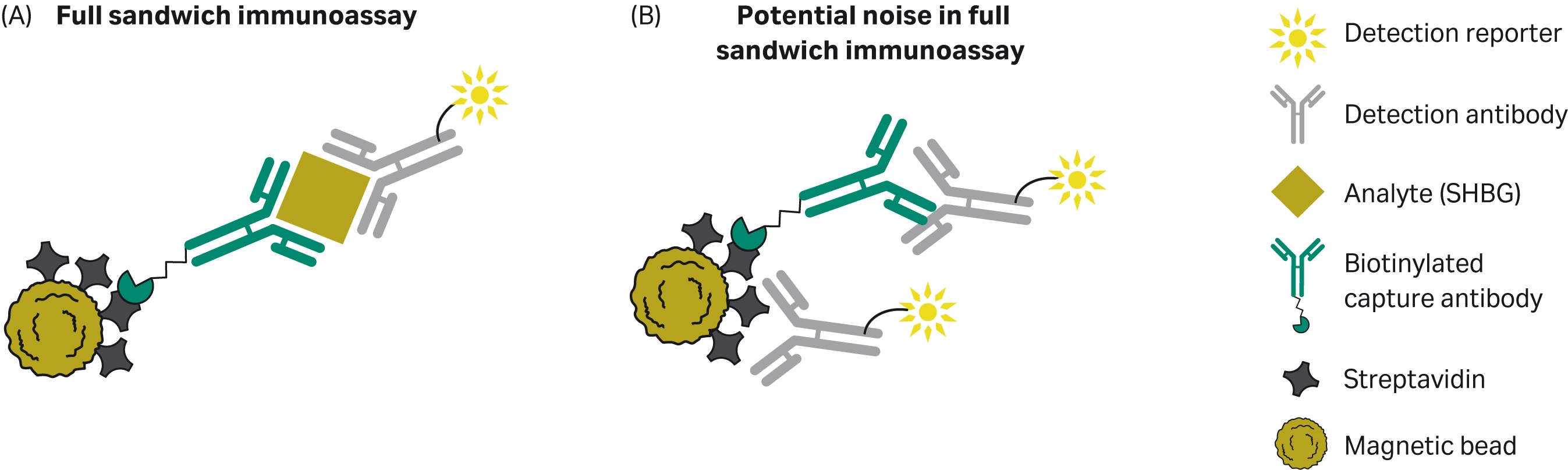
The performance of the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles was also evaluated in a full sandwich immunoassay using a commercially available SHBG CLIA immunoassay kit. Due to the complexities of interactions between the multiple components of a full sandwich immunoassay, typically it is best to optimize all components of the assay (4). Interactions between any of the assay components can contribute to noise in the assay and assay sensitivity (Fig 5).
Fig 5. (A) Diagram of a typical magnetic particle-based full sandwich immunoassay, and (B) noise in the assay due to non-specific binding of the assay components.
For the purposes of this comparison, the magnetic particles supplied in the IDS SHBG chemiluminescence immunoassay were exchanged with the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles and the other commercially available 3 μm streptavidin magnetic particles evaluated in this study. While holding the magnetic particle concentration constant, each type of magnetic particle was used to measure the level of SHBG in 10 different serum samples.
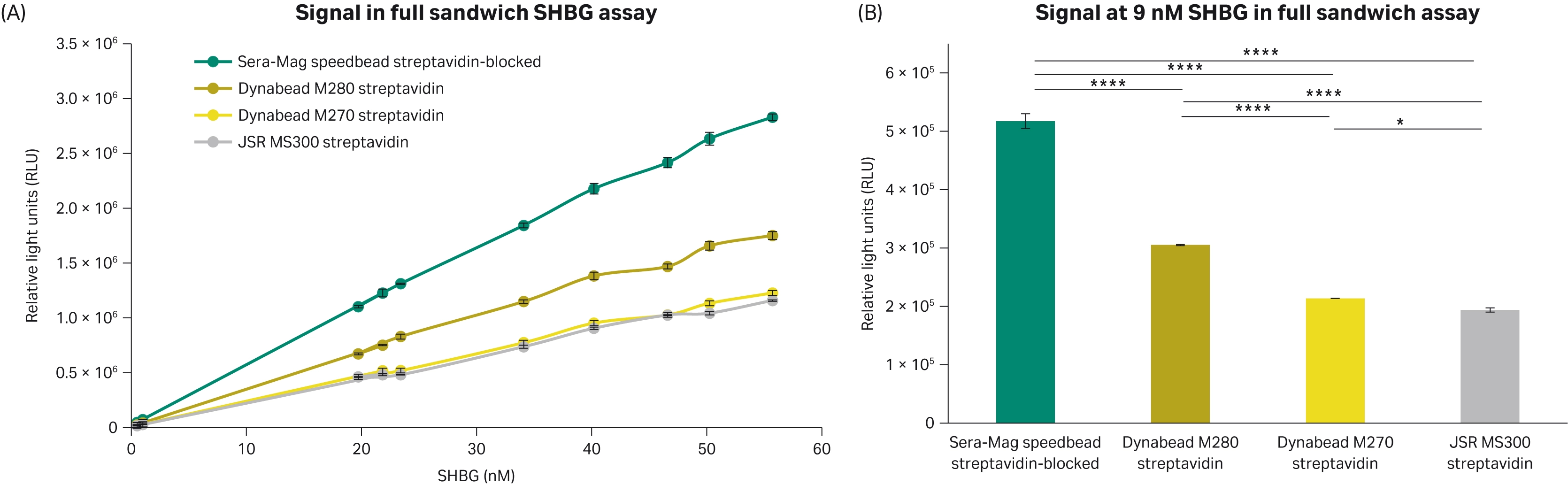
When using the SHBG CLIA kit, the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles were shown to have higher signal (Fig 6). This aligns with the results from the previous biotin-IgG binding capacity studies. This is of particular note at lower SHBG concentrations when the capture of the analyte may be more difficult (Fig 6B).
Fig 6. Signal achieved using the SHBG full sandwich assay. (A) Signal achieved with 4 different 3 μm streptavidin magnetic beads with 10 different serum samples with different levels of SHBG. (B) Signal from the different magnetic particles at 9 nM SHBG in the assay calibration curve. All conditions completed in triplicate (n = 3). Error bars represent standard deviation. Statistical comparisons completed using an ANOVA with Tukey’s pairwise post hoc test (*p<0.05, **p<0.005, ***p<0.0005, **** p<0.0001).
To further evaluate the sensitivity of the SHBG assay with the magnetic beads tested, the reaction slope was calculated by dividing the normalized signal by the SHBG concentration. The Sera-Mag™ speedbeads streptavidin-blocked 3 μm magnetic particles were calculated to have a higher slope (Table 2). This represents a larger increase in signal with increasing SHBG levels which could allow for smaller changes in the analyte to be distinguished.
Table 2. Reaction slope of the signal achieved per SHBG in the full sandwich assay. The relative reaction slope of each type of magnetic particles was calculated as a percentage of the Sera-Mag™ streptavidin-blocked 3 μm magnetic particles.
| 3 μm magnetic particles | Reaction slope (RLU/nM SHBG) | Reaction slope relative to Sera-Mag™ 3 μm |
| Sera-Mag™ speedbead streptavidin-blocked magnetic particles | 52 865 | 100 |
| Dynabead M270 streptavidin | 22 709 | 43 |
| Dynabead M280 streptavidin | 32 647 | 62 |
| JSR MS300 streptavidin | 20 979 | 40 |
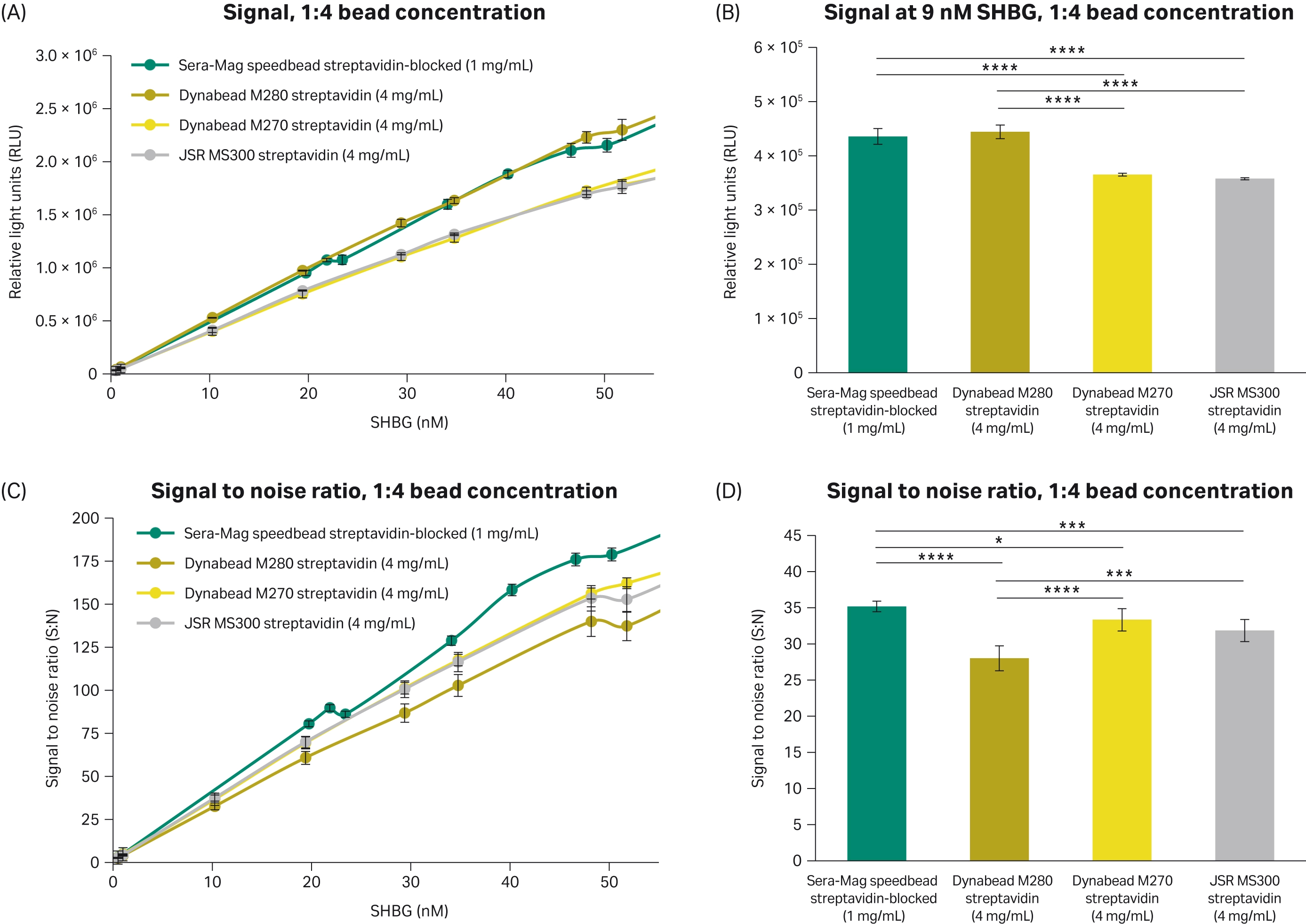
Since the observed signal achieved with the chemiluminescence immunoassay was higher with the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles compared to the other beads evaluated, magnetic particle concentrations were compared to determine what concentrations would achieve similar signal levels. Comparable signal was achieved with 1 mg/mL of Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles and 4 mg/mL Dynabead M280 streptavidin magnetic particles. The signal achieved with the 1 mg/mL Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles was higher than the 4 mg/mL Dynabead M270 streptavidin, and 4 mg/mL JSR MS300 streptavidin magnetic particles (Fig 7).
Comparing the ratio of signal-to-noise can also be an indication of sensitivity achieved with an assay. When using a quarter of the Sera-Mag™ speedbead streptavidin-blocked 3 μm magnetic particles, a higher signal to noise was achieved compared to the Dynabead M270 Streptavidin, Dynabead M280 Streptavidin, and JSR MS300 Streptavidin magnetic particles (Fig 7). Higher signal-to-noise could enable smaller changes in SHBG levels to be distinguished in the assay.
Fig 7. Performance in the SHBG assay when comparing the concentration of Sera-Mag™ speedbeads streptavidin-blocked 3 μm magnetic particles to other magnetic particles at a 1:4 ratio. (A) Signal achieved with four different 3 μm streptavidin magnetic particles with 10 different serum samples with different levels of SHBG. (B) Signal from the different magnetic particles at 9 nM SHBG in the assay calibration curve. (C) Ratio of signal-to-noise achieved with four different 3 μm streptavidin magnetic particles with 10 different serum samples with different levels of SHBG. (D) Ratio of signal to noise from the different magnetic particles at 9 nM SHBG in the assay calibration curve. All conditions completed in triplicate (n = 3). Error bars represent standard deviation. Statistical comparisons completed using an ANOVA with Tukey’s pairwise post hoc test (*p<0.05, **p<0.005, ***p<0.0005, **** p<0.0001).
Conclusions
We have shown that Sera-Mag™ SpeedBeads streptavidin-blocked 3 μm magnetic particles:
- Possesses a high biotin-IgG binding capacity which enables more analyte to be captured per magnetic particle or increases the chances of capturing analytes at low concentration levels.
- Achieved high signal and a high reaction slope at the SHBG concentrations tested.
These findings suggest that Sera-Mag™ speedbeads streptavidin-blocked 3 μm magnetic particles offer a promising solution for highly sensitive CLIA assay development, potentially improving diagnostic accuracy and patient outcomes.
All work completed in collaboration with Future Diagnostics
REFERENCES
- Tekin, HC, Gijs, MAM. Ultrasensitive protein detection: a case for microfluidic. Lab on a Chip. 2013;(24):4711-4739. doi: 10.1039/C3LC50477H
- Coarsey C, Coleman B, Kabir MA, Sher M, Asghar W. Development of a flow-free magnetic actuation. RSC Advances. 2019;(15):8159-8168. doi: 10.1039/C8RA07607C
- Darwish IA, Blake DA. One-Step Competitive Immunoassay for Cadmium. Anal. Chem. 2001;73(8):1889-1895. doi: 10.1021/ac0012905
- Tate J, Ward G. Interferences in Immunoassay. Clin Biochem Rev. 2004;25(2):105-120.