Key takeaways:
- Sterile filtration is a physical filtration process that uses membranes with defined pore sizes to remove bacteria and other microorganisms from liquids or gases.
- Applications of sterile filtration: Sterile filtration is widely used in aseptic pharmaceutical manufacturing, biotechnology processes, and laboratory research to maintain sterility without heat sterilization.
- Types of sterile filters: Sterile filters come in different materials, pore sizes, and formats, such as membrane filters, syringe filters, and cartridge filters, designed for specific applications.
- How to choose the correct sterile filter: Selecting the right sterile filter involves considering factors such as pore size, chemical compatibility, flow rate, sample volume, and application requirements.
- Integrity testing and regulatory considerations: Integrity testing verifies that sterile filters function properly, while regulatory guidelines ensure filtration processes meet quality and safety standards.
- What you will learn: By the end of this article, you will understand how sterile filtration works, where it is used, and how to select and validate the right sterile filter for your application.
Overview of sterile filtration
Sterile filtration is vital in biopharmaceutical manufacturing, ensuring the removal of microorganisms from process fluids to maintain drug product safety. In both aseptic processing and lab research, sterile filtration is generally used for protecting sensitive liquids from contamination without compromising product yield or filter integrity.
From upstream cell culture media preparation to downstream formulation and filling, sterile filtration provides a reliable method for achieving sterility, meeting stringent regulatory requirements, and supporting the consistent production of sterile biotherapeutics.
Core principles of sterile membrane filtration
Sterile membrane filtration is different from other methods of sterilization in that, instead of using heat, radiation, or chemicals to destroy microbes, it removes contaminants by passing the fluid solution through a membrane. This membrane traps microbes, effectively removing them from the process fluid and creating a sterile effluent.
Sterilization by filtration removes harmful or contaminating microorganisms from liquids and gases, such as bacteria and fungi. This is achieved using filters with a pore size of 0.2/0.22 microns (µm), which is small enough to physically prevent organisms from passing through the filter membrane. In fact, a sterilizing-grade filter is defined as a filter that can produce sterile effluent when challenged with 107 colony-forming units (CFU) of Brevundimonas diminuta (B. diminuta) per cm2 of filtration media. B. diminuta is one of the smallest bacteria, making it an ideal process model species to challenge the sterility claim of a filter.
Finer filters (0.1 µm) can be used to target smaller contaminating microorganisms like mycoplasma. These 0.1 µm filters are used to ensure cell culture media is sterile when making additions to bioreactors.
Prior to filling vials or syringes with a biotherapeutic drug, the bulk drug product is filtered through a 0.2/0.22‑micron sterilizing‑grade membrane filter. This step removes contaminating microorganisms and ensures sterility of the final filled product—without using heat or chemicals that could potentially damage or degrade the final drug product formulation. This is especially true for large or complex proteins and vaccines.
How does sterile filtration work?
Unlike terminal sterilization, which sterilizes the product after it’s been put into the final dispensing container, sterile filtration happens upstream of drug filling. While sterile filtration is always used at the final filling stage for sterile injectables, it can be used at different points during the bioprocessing workflow to achieve a level of sterility.
Sterile filtration plays a role in both upstream and downstream processes, but not all filters are sterilizing grade. Filters used for clarification, pre-filtration, and bioburden reduction are not considered sterile filters but are used in manufacturing drug substance to maintain a “low bioburden” environment, ensuring product yields/purity are not affected. These intermediate filters help reduce any potential contamination during the drug manufacturing process.
Depth filters (used for clarification or pre-filtration) remove larger particles after cell harvest, clarifying the process fluid for downstream purification steps but not producing a sterile effluent. Bioburden‑reduction filters reduce the level of microbes before final sterilizing filtration in the bulk filling process. These non-sterilizing-grade filters reduce microbial burden but don’t meet the regulatory requirement for a “sterilizing‑grade” filter, as the typical pore size is larger than .2/.22 microns.
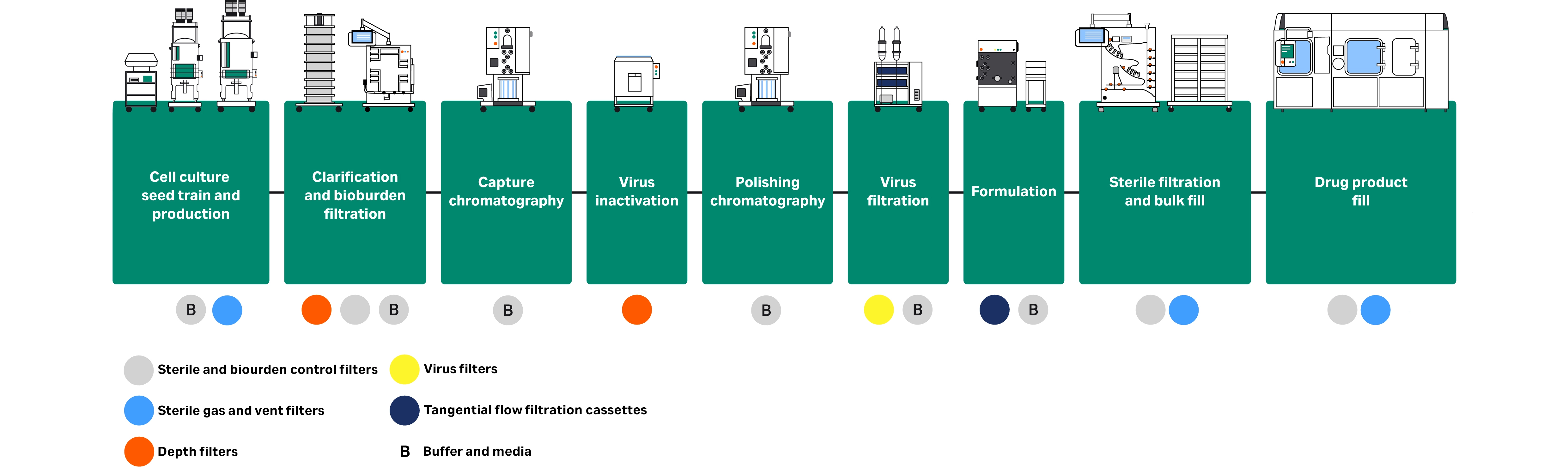
Some bioburden filters are considered sterilizing grade (0.2 µm); however, to make a sterility claim, the filter would need to be integrity tested, which typically occurs during the bulk filling and drug product filling process stages. Sterile filtration is used across the drug substance workflow, and sterilizing-grade filtration is used at the final stage, before drug product filling (Fig 1).
Fig 1. Application of liquid sterile filtration in a typical mAb downstream process.
Bioprocess vs lab filtration: what’s the difference?
Sterile filtration has many applications, including biotech and pharmaceuticals, food and beverage, and healthcare. As it pertains to biotech and pharma, sterile filtration is a critical step in both bioprocessing, including aseptic drug manufacturing, and lab research.
- Bioprocess filtration: Sterile filtration is applied at multiple points along the drug manufacturing workflow, from growth media, buffers, intermediate process fluids, bulk drug substance, and more. For aseptic drug manufacturing, the most important step of filtration is the final filtration step before filling a drug product. Sterilizing-grade filters for aseptic processing must undergo stringent integrity testing, including pre-use, post-sterilization integrity testing (PUPSIT). Additionally, their use in aseptic processing must be validated according to clinical regulatory requirements.
- Lab filtration: Filtration is important for various stages of cell culture, including sterilization of liquids, elimination of particulates, reduction of mycoplasma (a common source of contamination), and clarification of solutions. Sterilizing liquids in cell culture lowers the risk of contamination from media, reagents, and other additives.
Benefits of using sterile filtration
All in all, sterile filtration offers many benefits, some of which can be summarized as follows:
Prevents contamination
Sterile filtration maintains product safety and quality by removing contaminants from fluids and gases.Improves product integrity
By removing these harmful microorganisms, sterile filtration helps to ensure regulatory compliance standards requiring an absolutely sterile drug product.Reduces waste
Sterile filtration prevents contamination, which leads to batch loss, thereby reducing waste.Improves process efficiency
Sterile filtration streamlines bioprocessing and reduces batch loss, promoting faster, more precise results.Supports regulatory compliance
Sterile filtration ensures a quality standard that meets GMP and regulatory requirements for safe and effective drug products.
Limitations and risks of sterile filtration
On the other hand, sterile filtration does present some risks and limitations, which can affect product quality and safety, as well as the reliability of the filtration process:
Filter integrity
Sterilizing‑grade filters can become compromised or fail if they are damaged during handling or assembly, they are installed incorrectly, or pressure limits are exceeded. This can lead to contamination of the final product, and it is why integrity testing (pre‑ and post‑use) is required in GMP manufacturing.Filter fouling (clogging)
When proteins, lipids, or other particulates end up blocking filter pores, these can lead to reduced flow rates, increased pressure and subsequent damage, and ultimately batch failures. It is most problematic with viscous or high‑protein biologics.Filter incompatibility
Choosing the right filter or membrane is key to preventing product loss from adsorption of proteins, ensuring complete sterilization with the right pore size for the organism of concern, and discouraging extractables and leachables from entering the product.
What types of membrane filters are used in sterile filtration?
There are specific types of filters that can be applied for different sterile filtration needs. They can be categorized based on size exclusion, membrane type, and further by application, drug type, and scale.
Size exclusion
Membrane filters with pores around 0.2 µm physically block bacteria, fungi, and other microbes while allowing the drug solution to pass through. Sterile filtration to remove bacteria uses 0.2/0.22 µm filters whereas virus filtration requires smaller membranes with pore sizes as small as ~20 nm (0.02 µm).Membrane materials
Choosing the appropriate membrane type for your sample is crucial for success. Hydrophilic membranes such as polyethersulfone (PES) and polyvinylidene fluoride (PVDF) are commonly used for liquid filtration, while hydrophobic membranes are applied to gases.
PES membrane filters are hydrophilic and can filter aqueous and some organic solvents in many different types of environments. PES is also good for applications where there is high pH. PVDF membrane filters are resistant to a broad range of organic solvents. PVDF is naturally hydrophobic, but it can be modified to show hydrophilic properties.
Polytetrafluoroethylene (PTFE) membranes are hydrophobic, inert, and durable; the wide operating temperature range and resistance to the damaging effect of many chemicals make this a popular filter material.
Nylon membrane filters are naturally hydrophilic with wide chemical compatibility and good resistance to organic solvents. Nylon is particularly suitable for high-pH samples, but it should be avoided in any protein recovery applications.
How to select sterile filters?
Selecting the right sterile filter depends on several factors, including sterilization grade, process fluid, and workflow needs.
The pore size of your filter must meet sterility requirements; for example, sterile filters can reduce bioburden by removing bacteria of a certain size, but they might not be validated to the level of sterilizing-grade needed for aseptic filling. Viscous fluids will require specific filter types, as will solvents with specific chemical compatibility.
To select a sterile filter, you should consider:
- Sterility grade (level of sterility)
- Fluid characteristics
- Membrane/filter type (including size, pressure limits, etc.)
- Chemical compatibility
- Integrity testing capability
- Process risk and single‑use/closed system needs
- Supplier validation data and documentation
Filter integrity testing and filter validation for regulatory compliance
Filter integrity testing (IT) verifies that a sterilizing‑grade filter is functioning as stated by the manufacturer. IT can discover filter defects as well as setup issues like improper sealing. It is used before (pre-use) and after (post-use) filtration as part of GMP sterility assurance to ensure the filter has not been damaged during sterilization or processing.
Regulatory bodies require documented evidence of filter integrity for sterile filtration processes. In fact, in 2022 the European Union (EU) issued its updated Guidelines for Good Manufacturing Practice (GMP) for Medicinal Products for Human and Veterinary Use, Annex 1: Manufacture of Sterile Medicinal Products (1), requiring pre-use, post-sterilization integrity testing (PUPSIT). PUPSIT is now mandatory for drugs marketed within the EU unless the manufacturer can justify not doing it based on a thorough risk assessment. Currently, US Food and Drug Administration (FDA) guidelines require only post-use integrity testing (2).
Sterile filtration is subject to stringent regulatory oversight. Manufacturers must perform filter validation studies of their filter performance through a standardized and inclusive program of tests, including:
- Integrity testing: Confirms filter performance by performing IT before and after use.
- Extractable and leachables: Ensures there are not unsafe levels of compounds extracted from a filter or migrated into a drug product.
- Chemical compatibility: Ensures filter materials do not interact negatively with the product.
- Adsorption: Evaluates potential product loss due to binding to the filter’s surface.
- Bacterial retention: Demonstrates the filter’s ability to retain microorganisms under process-specific conditions.
Conclusion: sterile filtration is crucial for drug purity and patient safety
Sterile filtration is key to aseptic processing, as it ensures absolute sterility of a final drug product. As therapeutic modalities continue to evolve and manufacturers continue to innovate on membrane materials, sterile filtration processes and techniques will become even more critical to improving timelines and delivering a reliable, safe drug product.
Read more related articles:
>Frequently asked questions
1. What is the purpose of sterile filtration?
Sterile filtration removes microorganisms such as bacteria and fungi from process fluids to maintain drug product safety.
2. What pore size is considered sterile filtration?
Sterile filtration removes microorganisms using filters with a pore size of 0.2/0.22 microns (µm).
3. Does sterile filtration remove bacteria and viruses?
Sterile filtration removes bacteria, but not viruses. Viruses are an order of magnitude smaller than microorganisms so a specialist virus filter with pore size of 20 nm (0.02 µm) is typically used during a drug substance manufacturing process step.
4. Which filters are used for sterile filtration?
Membrane filters with pore size of 0.2/0.22 µm physically block bacteria, fungi, and other microbes. Membrane materials include polyethersulfone (PES), polyvinylidene fluoride (PVDF), polytetrafluoroethylene (PTFE), and nylon.
5. How is sterile filtration different from other sterilization methods?
In aseptic processing, sterile filtration uses a sterilizing‑grade filter (typically pore size of 0.2/0.22 µm) to remove microorganisms from a drug product solution. It is ideal for heat‑sensitive or radiation‑sensitive products that cannot withstand terminal sterilization. On the other hand, terminal sterilization sterilizes the drug after it has been filled and sealed in its final container, using heat or radiation.
6. What are the common uses for sterile filtration?
Sterile filtration is applied at multiple points along the drug manufacturing workflow, including clarification of process solutions, bioburden reduction, and the final sterilizing-grade filtration step before filling a drug product. Sterile filtration is important in cell culture, including clarification of solutions, elimination of particulates, and reduction of mycoplasma.
7. What is sterile filtration validation?
Filter validation is a series of tests designed to demonstrate that the sterile filtration step will reliably produce a product that meets sterility and quality requirements. Regulatory bodies expect process‑specific validation of sterilizing‑grade filters under worst‑case, realistic conditions.
References
- EudraLex - Volume 4 - Good Manufacturing Practice (GMP) guidelines; Annex 1 – Manufacture of Sterile Medicinal Products. European Commission. August 22, 2022. Accessed March 9, 2026. https://health.ec.europa.eu/document/download/e05af55b-38e9-42bf-8495-194bbf0b9262_en?filename=20220825_gmp-an1_en_0.pdf
- Guidance for Industry: Sterile Drug Products Produced by Aseptic Processing—Current Good Manufacturing Practice. US Food and Drug Administration (FDA). September 2004. Accessed March 9, 2026. https://www.fda.gov/media/71026/download