If you could reach GMP filling readiness earlier, what would it mean to your company’s bottom line? The success of your product launches?
In this blog, we explain why Cytiva’s filling technology provides speed to good manufacturing practices (GMP) filling readiness compared with conventional options – isolator barrier systems and restricted access barrier systems (RABS).
There are several reasons for the speed advantage:
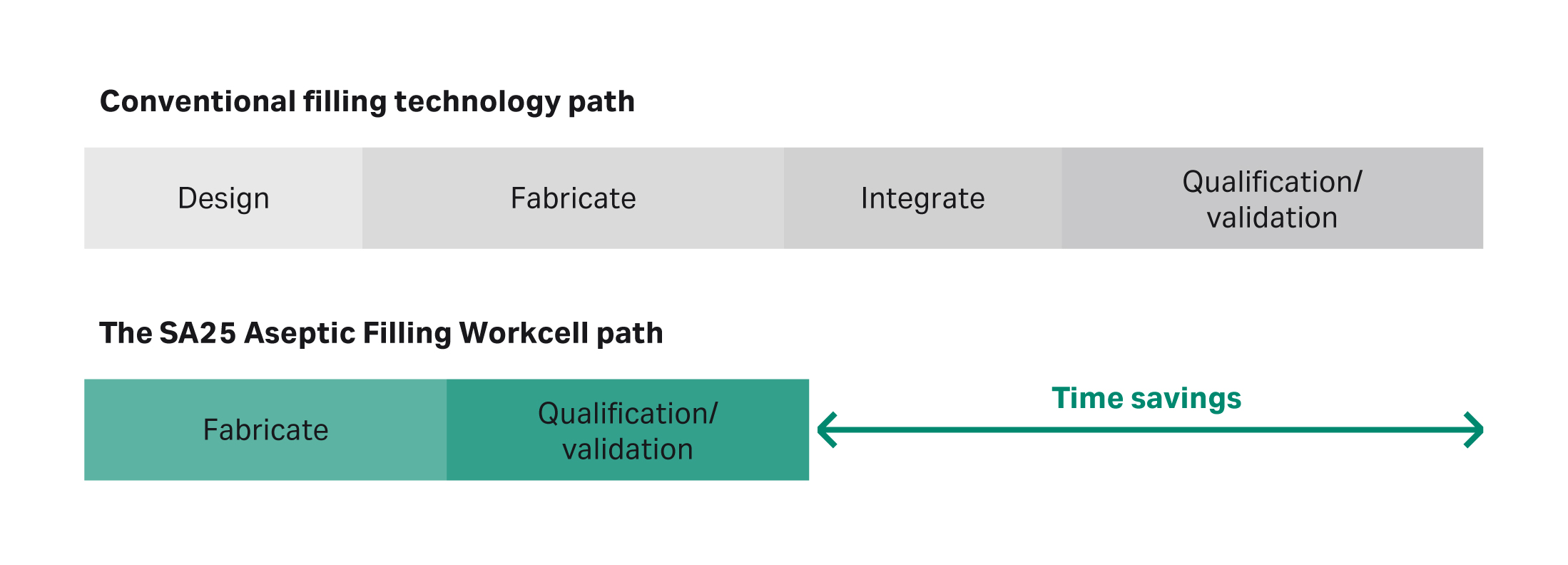
- Standard machine designs allow faster build and installation times, eliminating the design and integration stages of projects (Fig 1);
- The aseptic process was designed by pharmaceutical manufacturers to emphasize flexibility and sterility assurance – which avoids modular systems and design and redesign ’by committee’;
- Cytiva’s systems require a smaller Grade C cleanroom space that is faster and less expensive to build; and
- A machine can be built in parallel with modular cleanroom facilities, so you could potentially have a complete facility in months, not years.
Fig 1. Illustration comparing the path to build and install Cytiva's aseptic filling technology vs the path for conventional options.
Data to support this approach
Here are some data points to support this approach to gaining speed to market:
- Over a three-year period, the average time from purchase order to factory acceptance test (FAT) was 26 weeks (6 months). Compare that against 12 to15 months for conventional systems.
- In the same period, customers averaged 7 weeks from the completion of FAT to completion of site acceptance test (SAT).
- Customers, such as Emergent BioSolutions routinely reach GMP within 12 to 15 months of their purchase order with multiple vial, syringe, or cartridge formats validated.
What’s the cost of being early vs late?
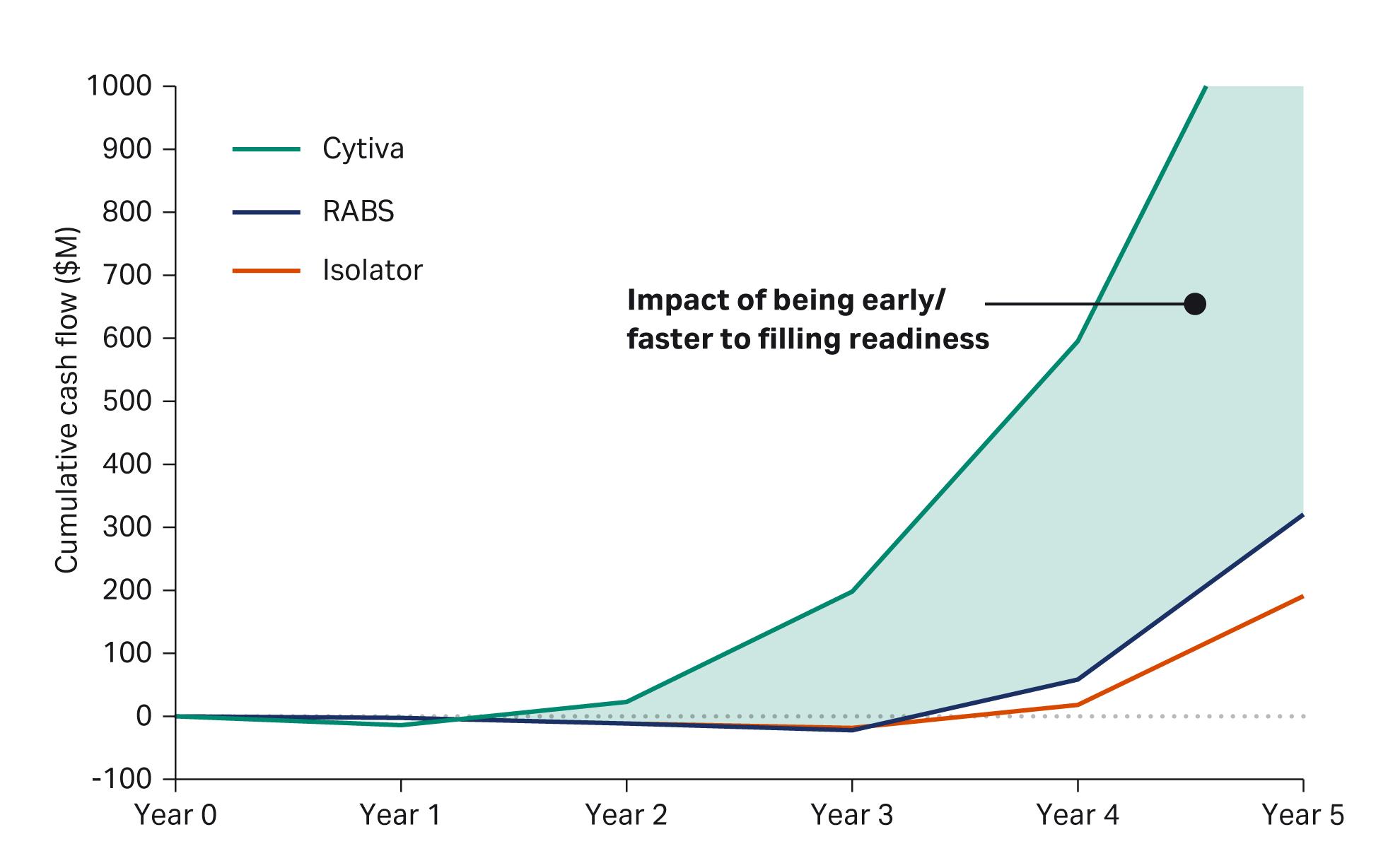
To put a number on the cost of differing market entries, see the model in Figure 2, which assumes you have a portfolio of products ready to manufacture or are moving to launch new products.
When comparing the available barrier technologies for aseptic filling, we can see a similar cumulative cash flow in the first two years of each project as they work toward GMP manufacturing.
With Cytiva's technology as shown in this model, you would reach positive cash flow earlier and with less risk of going over budget and over your project timeline. The model assumes you have passed validation between months 12 and 18 and are producing commercial products.
Fig 2. Model showing cumulative cash flow over five years for three aseptic filling technologies.
In this model, because of the standard design and rapid lead time with Cytiva’s technology, that GMP milestone is reached two years earlier than in the other projects.
And what happens in the alternate scenario of being late? Your company misses out on all of the benefits of being early, likely overspending your budget and possibly underperforming in a product launch versus competition.
Last Updated: November 14, 2024