By Christopher J. Woolstenhulme, Nathan D. Israelsen, Damien Bellos, Alexandre Pereiras Magariños, and Andra Kunzler
Abstract
Chemically defined cell culture media have been widely adopted in biomanufacturing processes to produce biotherapeutics such as biosimilars, vaccines, and monoclonal antibodies. Performance of these biotherapeutics can be greatly affected by trace impurities introduced by the media raw materials. The additive effect of trace impurities can dramatically increase concentrations of components including metal salts and other trace impurities. Therefore, precise control of trace impurities is crucial for drug consistency and performance. Here we present a novel approach for manufacturing chemically defined media with precision control of trace impurities. We use a combination of innovative raw material analysis, data analytics, and a proprietary algorithm to select raw material lots to manufacture media containing metal and other trace impurity concentrations within customer-defined limits. To date, we have successfully manufactured numerous lots using this technique.
Introduction
Chemically defined media (CDM) for cell culture were created to ensure consistent yields and performance from cell culture products, including biotherapeutics. Unlike serum, CDM provides flexibility and traceability of raw materials through complete customization of formula components. However, more components mean more sources of impurities. Every raw material used in CDM typically meets some compendial grade requirements. For example, USP requires materials be ≥ 98.5%–99% pure. Usually the 1.0%–1.5% of impurities are not characterized. By themselves, these impurities may not affect the overall performance of the media. However, CDM formulations can contain more than a hundred raw materials. The additive effects of impurities from so many components can affect cell culture performance. At best, they cause inconsistency. At worst, they cause complete product failure. The additive effects are perhaps best demonstrated by trace metals and their effect on cell performance and protein quality.
Numerous studies have investigated the effects of trace metals on different cell lines (1). Many of these metals are crucial to cellular metabolism, metalloenzymes, transport pathways, cell life cycle, and monoclonal antibody (mAb) glycosylation patterns (1). The latter is of particular interest to pharmaceutical companies because mAb glycosylation patterns impact drug efficacy and consistency. Studies have shown that small changes in trace metal concentrations can greatly affect glycosylation patterns in certain cell lines (2). To minimize risk of inconsistent and less efficacious therapeutics, strict control of raw materials in CDM must be achieved.
Here we present a novel approach for precision control of trace impurities when manufacturing CDM. We use innovative analytical techniques to build a database of impurities for all our raw materials. Our proprietary algorithm then selects specific lots to manufacture media with metal and other trace impurity concentrations within customer specified ranges. To date, we have produced more than 20 lots of complex CDM using this technique and met our customers’ targets for every lot.
Trace impurities
Impurities in raw materials and CDM
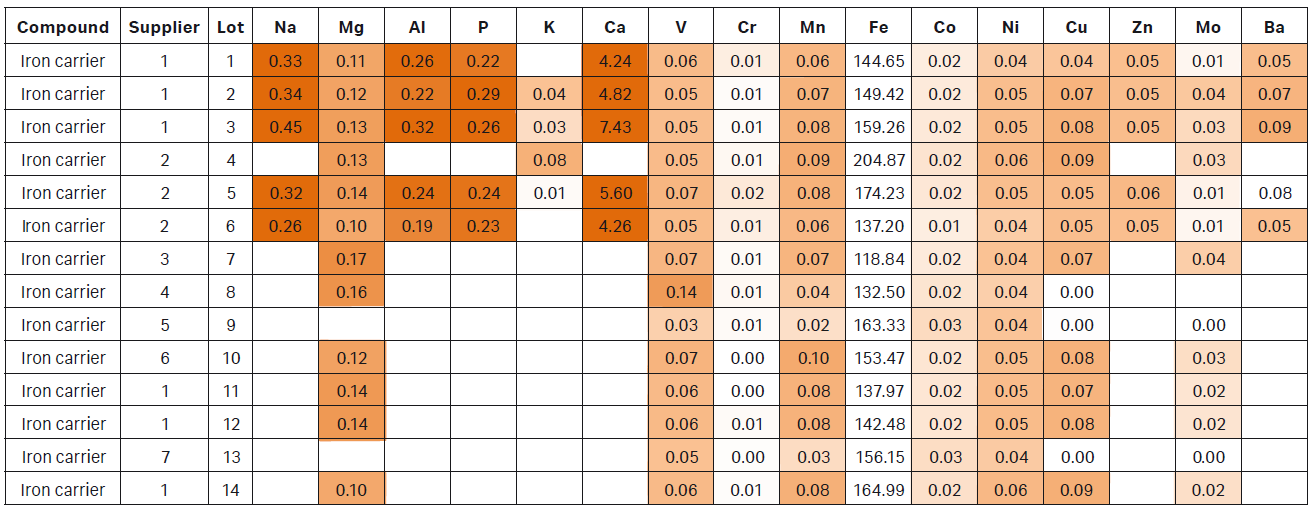
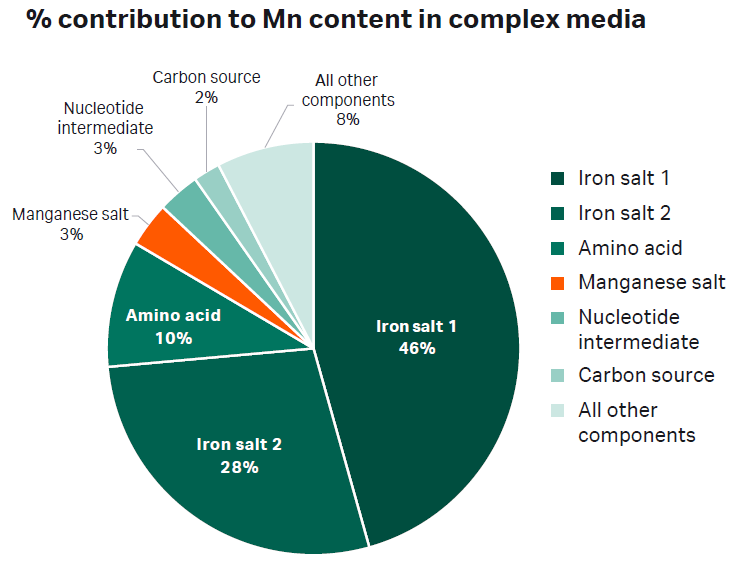
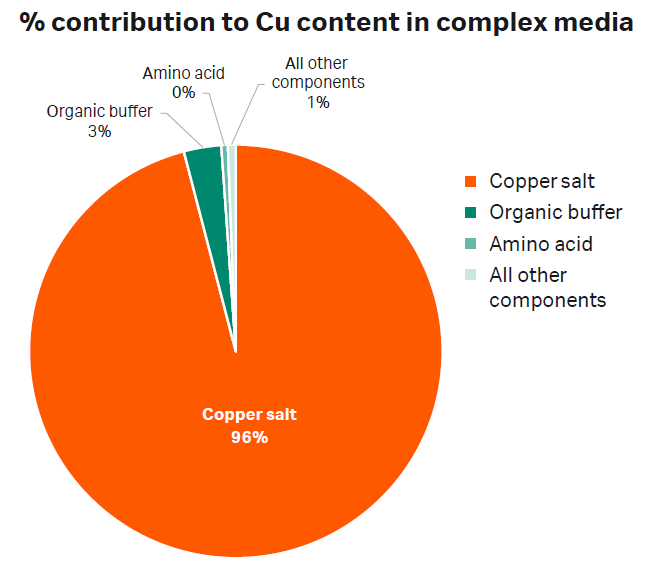
Trace impurities found in raw materials vary depending on how the material was manufactured, synthesized and/or mined. Different suppliers have different impurity profiles and variation even exists among different lots from the same supplier. Our analysis demonstrated that some materials contain a wide range of impurities (Table 1). The accumulation of these impurities can significantly alter the concentrations of metals and amino acids in the final medium. To demonstrate this, we analyzed a batch of complex CDM for its metal content. We discovered that manganese salt contributed only 3% to the overall manganese (Mn) content (Fig 1) whereas copper salt was the main contributor to copper (Cu) content (Fig 2).
Table 1. Heat map of trace metals found in iron carriers. Analysis was completed on lots from different suppliers and different lots from the same supplier. A wide range of impurities can be found in all lots tested. Reported results are in parts per million (ppm).
Fig 1. Chart depicting the percent (%) contribution of each component in a complex chemically defined medium to the overall Mn concentration.
Fig 2. Chart depicting the percent (%) contribution of each component in a complex chemically defined medium to the overall Cu concentration.
Precision manufacturing
Predictive modeling
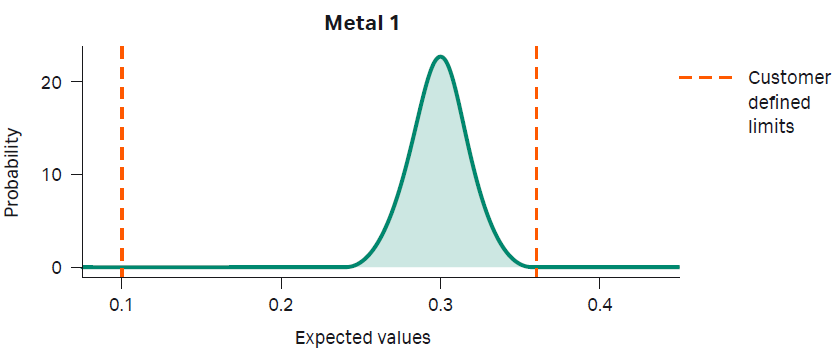
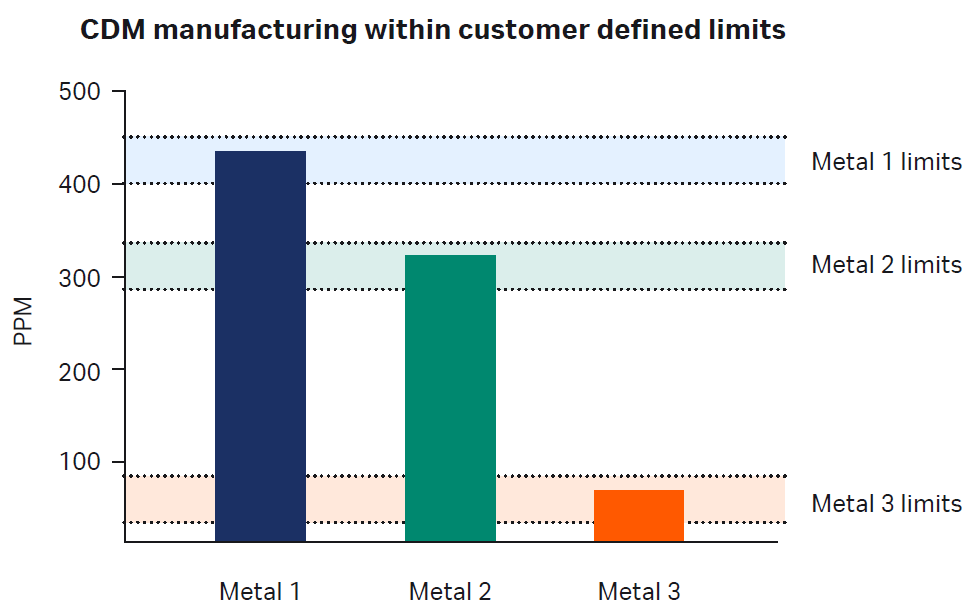
Precision manufacturing involves two phases: predictive modeling and manufacturing. In the first phase, predictive modeling, all raw materials have been thoroughly tested and data are stored on secured databases. Next, the customer determines the desired target range for metals and/or other trace impurities. We then statistically model the probability of our ability to manufacture CDM based on current and historical data (Fig 3). Once we confirm our capability to produce the material, we proceed to the next step.
Fig 3. An example of a normal distribution of historical data modeling the probability of producing medium with Metal 1 content between upper and lower limits specified by our customer.
Manufacturing
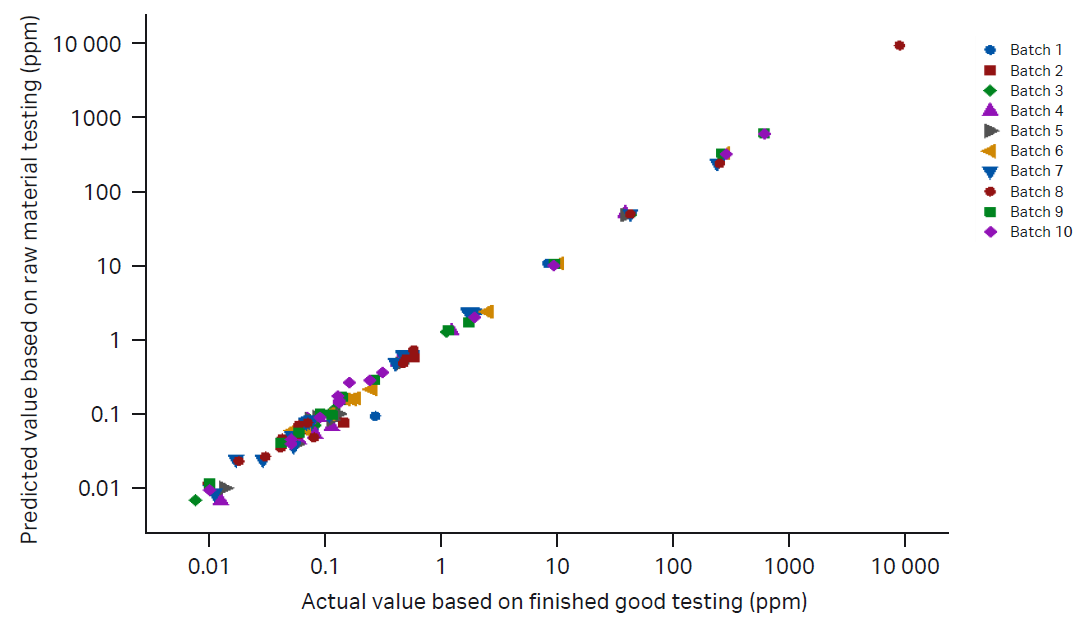
The next phase of precision manufacturing involves selection of lots by a proprietary algorithm. The algorithm chooses the best lot combination that will produce medium within the desired limits (Table 2). Once the medium has been produced, it is tested to confirm metal and/or other trace impurity content (Fig 4). Historical analysis of product data showcases a high degree of correlation between predicated values based on raw material testing and finished good product testing (Fig 5).
| Component 1 | Component 2 | Component 3 | Component 4 | Component 5 |
|---|---|---|---|---|
| Lot 1 | Lot 1 | Lot 1 | Lot 1 | Lot 1 |
| Lot 2 | Lot 2 | Lot 2 | Lot 2 | Lot 2 |
| Lot 3 | Lot 3 | Lot 3 | Lot 3 | Lot 3 |
| Lot 4 | Lot 4 | Lot 4 | Lot 4 | Lot 4 |
| Lot 5 | Lot 5 | Lot 5 | Lot 5 | Lot 5 |
Table 2. The algorithm chooses lots for each component of the medium that will produce a product within customer-specified limits.
Fig 4. Final products are tested for metal content to ensure they meet our customers’ criteria.
Fig 5. Correlation graph of metal content from actual batches produced using this method in comparison with predicted values (R2 > 0.9).
Conclusions
We developed an innovative approach for controlling trace impurities in cell culture media with precision accuracy. With precision manufacturing, we can control metal and amino acid content in media and manufacture to customer specifications with a high degree of accuracy. Our capacities include:
- Track and trend trace impurities in raw materials from lot to lot and supplier to supplier
- Determine percent contribution of our raw materials to our overall metal content in CDM
- Model ability to produce CDM within customer-defined limits
- Produce CDM within customer-defined limits as verified by analytical testing
- Potential application to other types of impurities
Learn more about how Cytiva can help with cell culture raw material characterization to minimize your process variability.
References
- Graham RJ, Bhatia H, Yoon S. Consequences of trace metal variability and supplementation on Chinese hamster ovary (CHO) cell culture performance: A review of key mechanisms and considerations. Biotechnol Bioeng. 2019;116(12):3446–56.
- Gangwar N, Priyanka P, Rathore AS. Achieving charge variant profile of innovator molecule during development of monoclonal antibody based biosimilars – Use of media components. Biochem Eng J. 2022;182:108438.