As process development of biologics becomes more and more data and simulation driven (Fig 1), interest in mechanistic modeling is also increasing. This approach allows you to simulate and predict chromatographic behavior and experiments in silico.
Mechanistic modeling is part of smart process development, a collection of approaches to improve process outcomes and speed up process development. Together with statistical models based on multivariate data analysis (MVDA) such as DoE, it can be a powerful tool to save time and create more robust processes, which is the goal of the overarching QbD framework.
The approach can be a shortcut to more robust process outcomes, but it is in no way a straight path. In this article, I’ll outline both the current opportunities and challenges for using mechanistic modeling for process development of chromatography steps.
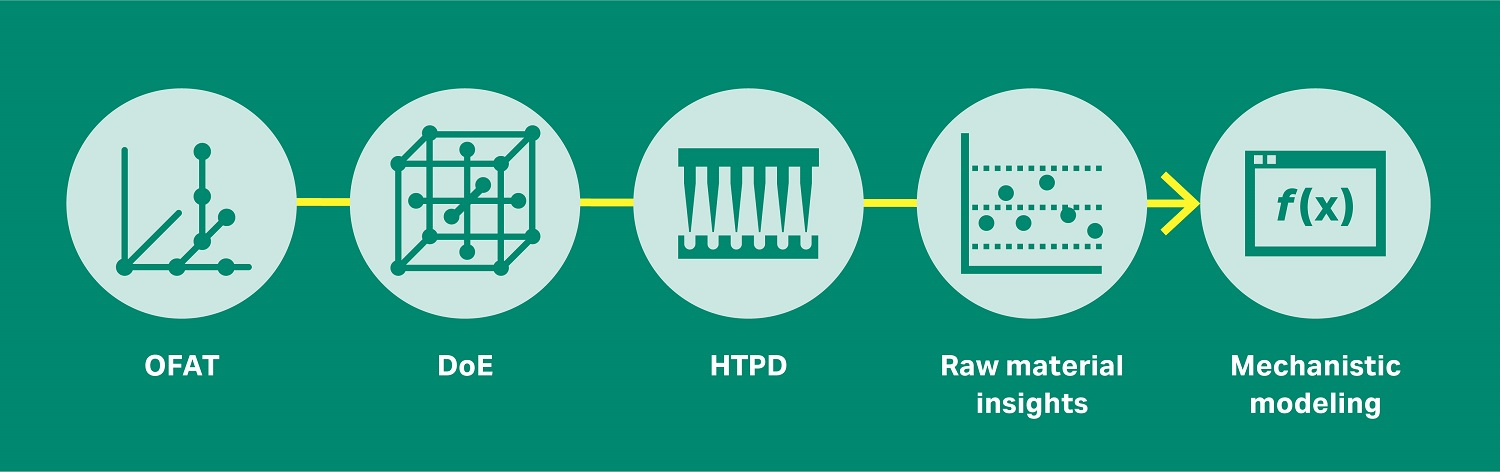
Fig 1. The evolution of process development, from one-factor-at-a-time (OFAT), through design of experiments (DoE), to high-throughput process development (HTPD), raw material insights and quality by design (QbD), and now mechanistic modeling.
What is mechanistic modeling in chromatography?
Mechanistic models use computer simulations to decrease the number of experiments needed during process development. The simulations are based on known physiochemical phenomena involved in chromatography.
Mechanistic modeling is a mathematical representation of the physiochemical transport and interactions that occur during chromatography. For example, this methodology uses differential equations that describe how molecules move between resin beads and inside the bead pores. It also uses adsorption isotherms to quantify how molecules compete for ligands when binding.
The methodology is not exactly new, but its use is moving from academic research to implementation in the biopharma industry, driven by a need to improve outcomes and speed up process development. Thanks to greater computer power and improved software, mechanistic modeling is now more accessible to process developers in the industry.
Why use mechanistic modeling in downstream process development?
Mechanistic modeling is a complement to other process development approaches, such as HTPD and MVDA. Unlike in MVDA, physiochemical effects are also considered in mechanistic modeling and allow interpretation of the model parameter values for increased process understanding.
With mechanistic models, process developers can get a better understanding of both the entire process and the parameters that can influence the process with a smaller number of experiments. That way, process development can become faster, more scientific, and more reliable. The ability to anticipate and model scale effects is another advantage of mechanistic modeling over MVDA. Mild extrapolations are also covered in the mechanistic modeling framework.
Possible applications include prediction of step elution conditions for ion exchange chromatography, facilitation of tech transfer by predicting scale-up from lab to process chromatography columns and explaining deviations in manufacturing.
What are the current opportunities with mechanistic modeling?
As mentioned, the advantage of mechanistic modeling is that it can speed up process development and improve processes. The time invested in calibrating the model reduces the number of experiments needed, saving both time and resources such as sample and resin.
The deeper process understanding obtained from this methodology also results in more robust processes and improved performance, as evaluated by yield or throughput, for example. It is also beneficial for tech transfer, as it enables handover of a scalable predictive model instead of just a large set of data. An added advantage is the ability to use historical data, to gain even deeper understanding of existing processes.
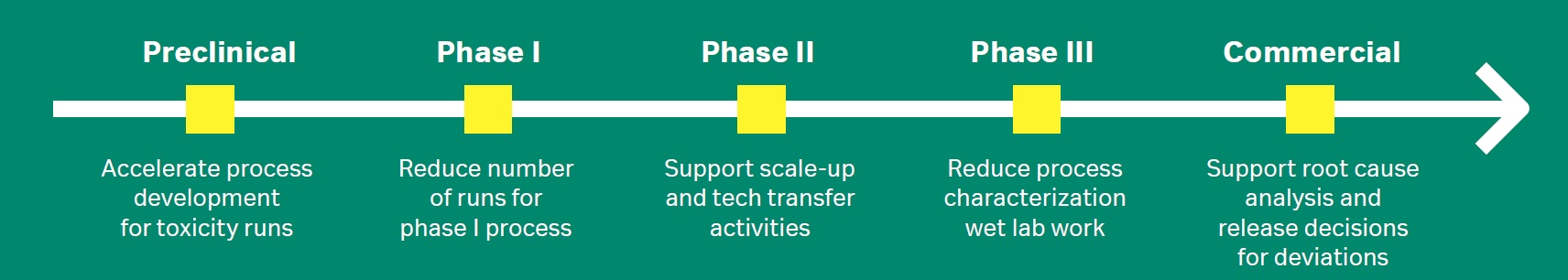
There are several ways mechanistic modeling can bring value to drug development work. At present, the approach is typically used during late-stage development, but the possibilities to use it in early-stage process development are increasing. Accelerating early process development allows you to shorten the timeline to toxicology and first-in-human studies.
Figure 2 shows an overview of the opportunities throughout the different drug development phases. The methodology is particularly useful for process development and characterization as well as tech transfer.
Fig 2. Examples of how different steps in drug development can benefit from chromatography mechanistic modeling.
What can you get out of mechanistic modeling of chromatography?
In chromatography, mechanistic modeling can be used to simulate elution profiles under different conditions such as column dimensions, load ratio, flow rate, pH, and conductivity. Simulations can be performed for both major and minor sample components. The output can then be used to optimize the separation.
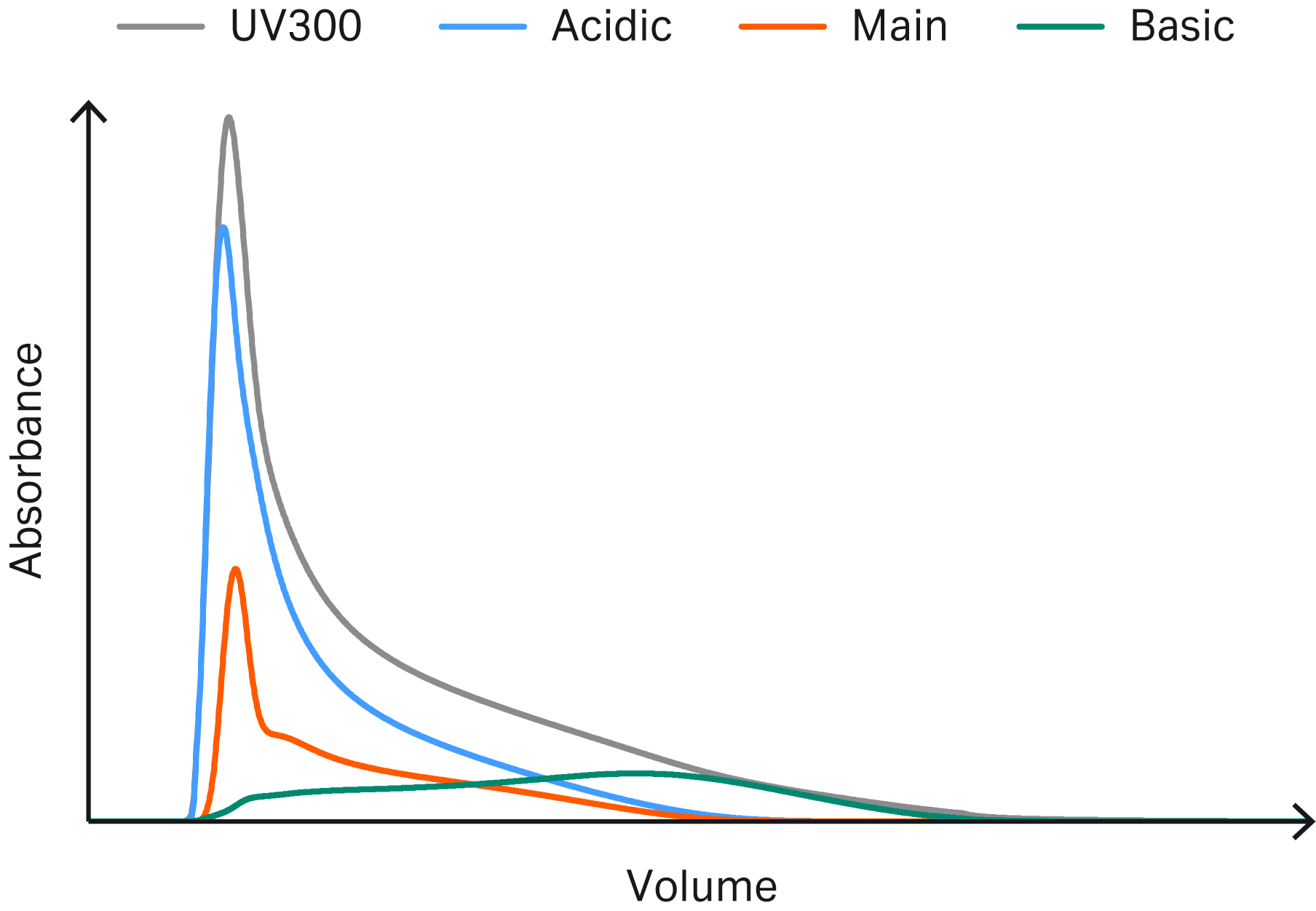
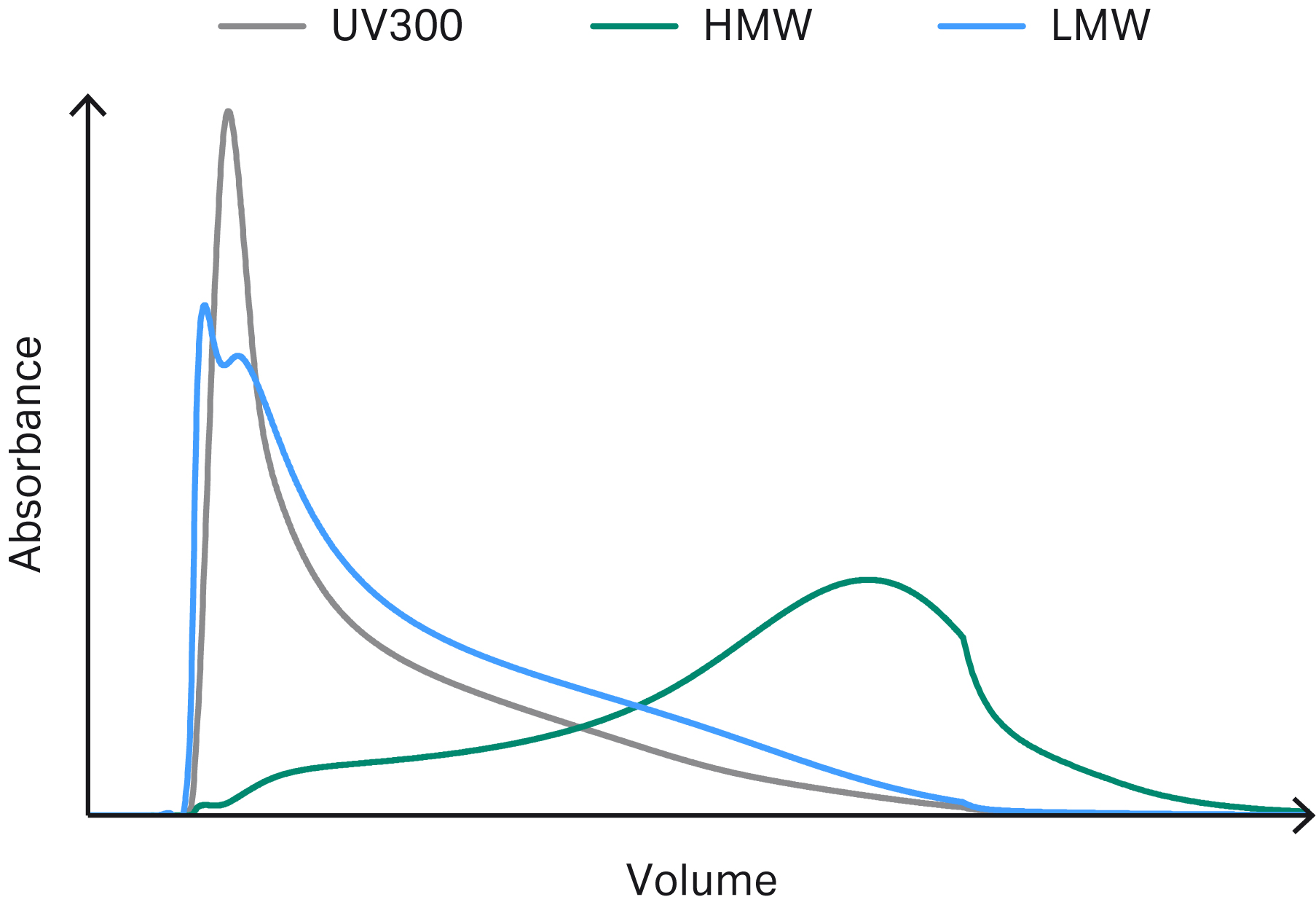
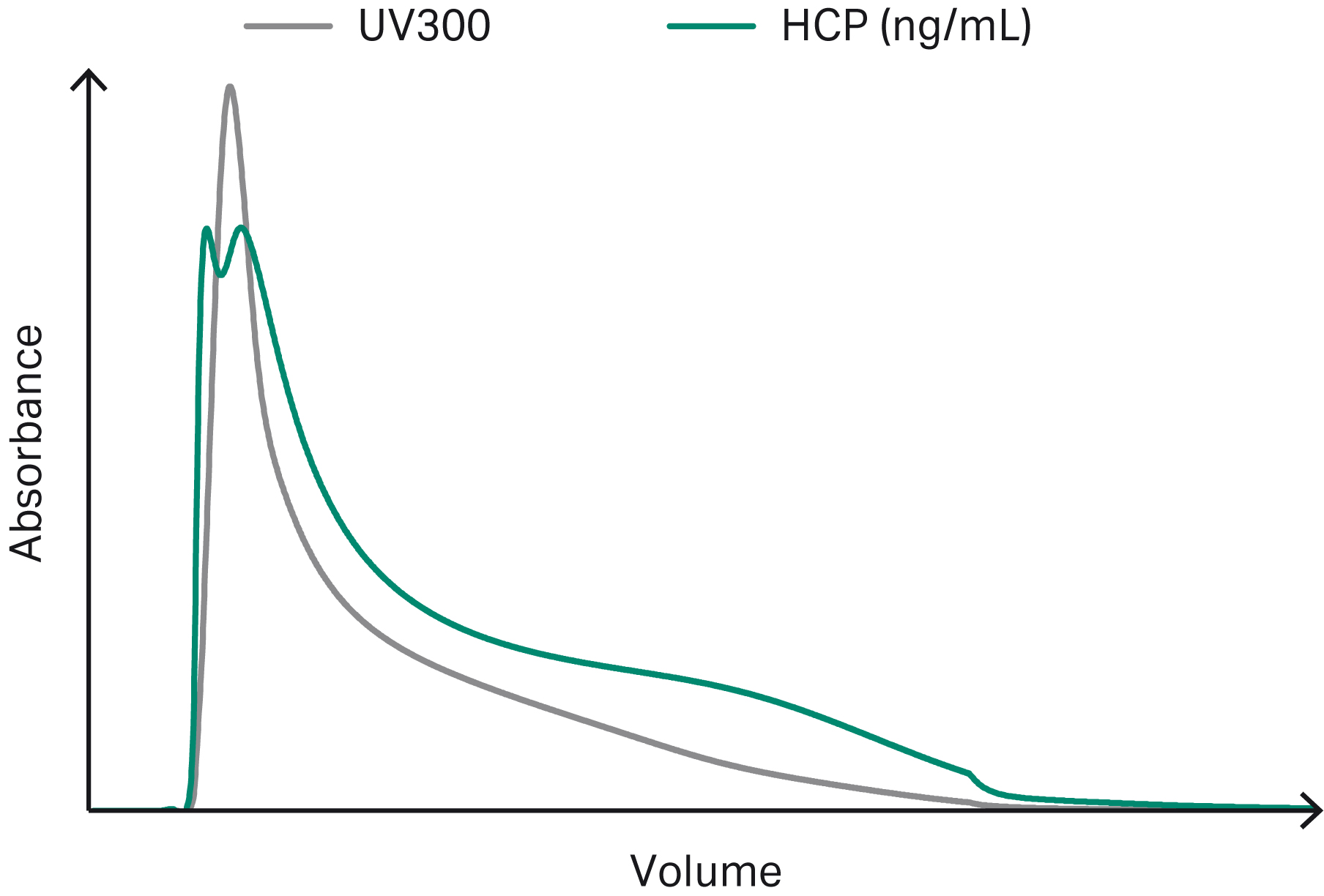
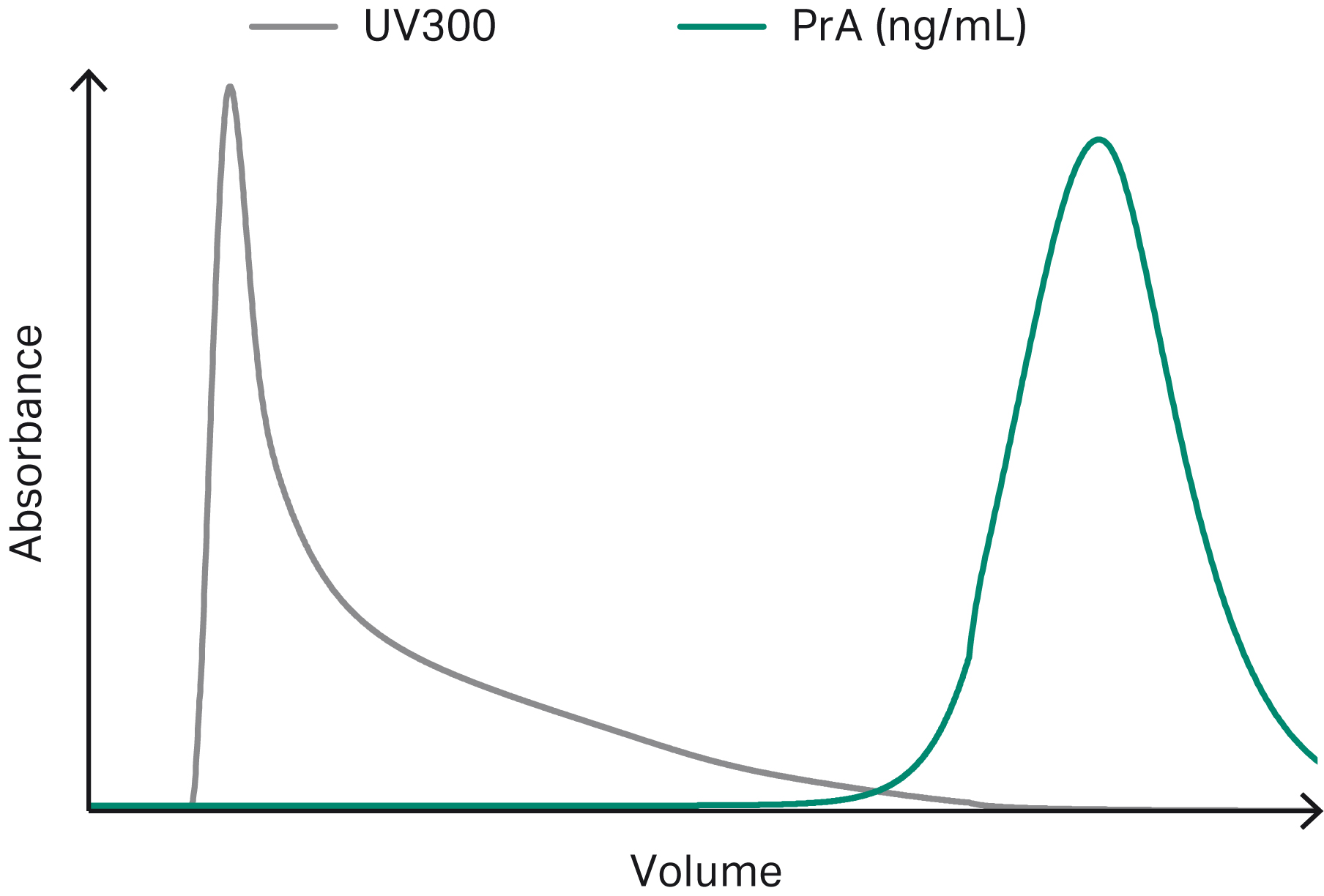
Figures 3–6 show example chromatograms from simulations of mAb elution profiles on Capto™ S ImpAct resin. The figures show profiles for charge variants, aggregates and fragments, host cell proteins (HCP), and leached protein A.
By inspecting the simulated elution profiles, it is possible to generate potential pooling criteria that can be tested and optimized in silico prior to experimental verification.
Fig 3. Example chromatogram showing simulated elution profiles for charge variants.
Fig 4. Example chromatogram showing simulated elution profiles for aggregates and fragments. The UV curve is plotted on a separate y-axis scale.
Fig 5. Example chromatogram showing simulated elution profiles for HCP. The UV curve is plotted on a separate y-axis scale.
Fig 6. Example chromatogram showing simulated elution profiles for leached protein A (PrA). The UV curve is plotted on a separate y-axis scale.
What are the current challenges with mechanistic modeling?
There are many possibilities to speed up process development work with mechanistic modeling, but applying this approach is in no way a quick fix. In order to reap the benefits of the methodology, you need to make long-term investments in time, knowledge, and people. The learning curve is rather steep, even with current improvements to software packages.
In other words, it takes time to get to the point where you have enough knowledge to take maximum advantage of this approach. It can also be challenging to find the right people for the job, as it is a specialized skillset with high demand.
Another challenge worth mentioning is the increased analytic workload that this approach creates. Using simulations results in less experimental work for process developers. However, it may also rely on more analytical work, as you need to collect a large number of fractions for product quality and yield analysis to describe the mechanistic dynamics.
I described this shift in analytical workload in a presentation at the HTPD conference in 2019. In one case study, we estimated that the number of chromatographic runs for model determination would be reduced by 62% compared to that of a hypothetical traditional central composite DoE three-factor design. However, we estimated that the number of fractions to analyze for product quality would increase by 69%, though this could represent a better total lifetime investment in analytic effort for the process step. One could also note that the level of process understanding gained is higher for the mechanistic modeling approach.
What about challenging separations?
Today, good models exist for ion exchange separations performed in both bind and elute and flowthrough mode, and models for hydrophobic interaction and multimodal chromatography are often just as straightforward to use. Models describing other adsorber chemistries, including ceramic hydroxyapatite, reversed phase, and affinity systems might not be as developed, but substantial effort is going into application in these modalities. It’s not yet fully clear how useful mechanistic modeling will be for more challenging separations.
A sharp tool to add to the smart process development toolbox
Mechanistic modeling is not a quick fix for faster process development for any separation. There is a learning curve, and the first studies might be rather time consuming. However, together with other smart process development approaches, such as HTPD and MVDA, mechanistic modeling will ultimately help speed up chromatography process development and provide deeper process understanding.
Learn more or contact us about mechanistic modeling.
Author: Gunnar Malmquist

|
Dr. Gunnar Malmquist is a Senior Principal Scientist within our BioProcess R&D section and has over four decades of chromatography experience. His current focus is resin design strategies, quality by design, and process analytical technology together with empirical and mechanistic modelling of chromatography data for smarter process development and increased process understanding. |