Source: Cytiva
Introduction
As we began 2020, no one could have foreseen that just a few weeks into the year, we would be facing a pandemic threatening unprecedented damage to human health and the world economy. Businesses are currently grappling with disrupted supply chains, restrictions (and even shutdowns) in air travel and freight, and serious challenges in meeting their day-to-day business objectives and longer-term financial obligations.1
In facing such an unanticipated situation, it is crucial to have strong protocols and programs in place that will allow business operations to take place uninterrupted, or as close to normal as possible. This is where a proactive business continuity management (BCM) program can make a significant impact in securing supply, reducing risks and downtime, and maintaining quality in manufacturing operations.
Fig 1. We are maintaining deliveries despite COVID-19 challenges, and business continuity management has a key role delivering these capabilities. In this image, 43 tons of scientific instruments leave Stockholm Arlanda airport, bound for our customers in the USA.
What business continuity management means in practice
Implementing a robust BCM program requires a solid understanding of the processes involved in continuous and sustainable business. This allows the team to properly map the processes and assess the risk associated with each process (and the resources pertaining to that process). Once you understand your risks well, it is easier to prepare for them and develop a plan that can be executed at the time of a crisis.
Some key components of a BCM program include:
- Carrying out a Business Impact Analysis (BIA) on the BCM processes and mapping critical resources
- Carrying out a risk assessment on the resources
- Developing a business continuity plan to show what steps will be taken to reduce interruptions in the case of a disaster
- Implementing the business continuity plan and executing necessary exercises to test the plan
- Conducting an internal review and reevaluating the plan regularly to allow for continuous improvements
Part of running a successful BCM program is ensuring that your protocols are up to international standards. Particularly for business continuity, organizations can obtain accredited certification against ISO 22301, a recognized management systems standard developed by the ISO Technical Committee 223 for societal security.2 This standard helps build a structured and documented approach to minimize the risk and impact of incidents preventing manufacture and assures that the certified organization adheres to good practices in BCM.
International certification ensures quality in business continuity management
In 2021, Cytiva completed recertification to ISO22301:2019 on all of its mission-critical production facilities worldwide. This includes seven manufacturing sites: three in Europe, two in the US, one in Asia, and one in Oceania. Having the same BCM certification at multiple sites has enabled us to effectively maintain a uniform standard across our entire organization, while also setting an important precedent for the industry, in terms of quality in BCM.
Fig 2. Between 2015 and 2021, we were able to recertify all of our key manufacturing sites with ISO 22301 Business Continuity Management certification. This image shows our cell culture media facility in Singapore.
We have learned that thanks to the uniformity in the framework of our BCM program, it is easier to manage teams during a crisis – switching personnel to different sites becomes an almost seamless operation, with minimal confusion and disruption. Additionally, our customers can be confident that we strictly adhere to the quality standards defined by ISO 22301 and that we have the preparedness to act quickly and efficiently through well-developed plans, should a crisis situation emerge.
Solutions driven by a successful BCM program
Cytiva’s BCM program consists of concrete solutions that help mitigate risk and ensure security of supply. It is important to understand that each product portfolio has different supply chain, quality, and manufacturing needs, and the solutions are tailored according to those needs. Here are three examples:
(1) Strategic reserves: For our chromatography resins, we have built a strategic reserve of 90 final products, available at two locations (geographically separate from our manufacturing sites) to cover a gap in production during a crisis. This backup inventory is in place to ensure continued supply to customers who depend on our products for the production of regulatory approved human biopharmaceuticals and vaccines. The strategic reserve is reviewed annually and the content rotated on a regular basis to maximize shelf-life. It is not earmarked for any specific customer.
(2) Multi-site manufacturing: We have established multiple manufacturing sites around the globe, which allows us to rotate production if we face challenges at one location, for example, due to a crisis. Examples are the facilities for cell culture media production in Pasching (Austria), Utah (USA), and Tuas (Singapore), all of which have the same pin-milling technologies that generate equivalent results3 and a harmonized quality management system. With multiple sites having the same capabilities, we can increase the manufacturing capacity, while also reducing logistic complexity when we do need to move production around.
Fig 3. The three sites for cell culture medium production all have the same pin-milling technologies that generate equivalent results.
(3) Supporting Suppliers and Global Warehouses: We have encouraged and supported two of our most critical suppliers and global strategic warehouses to develop business continuity programs that, in turn, help us ensure our supply in a more organized fashion.
Communication is key
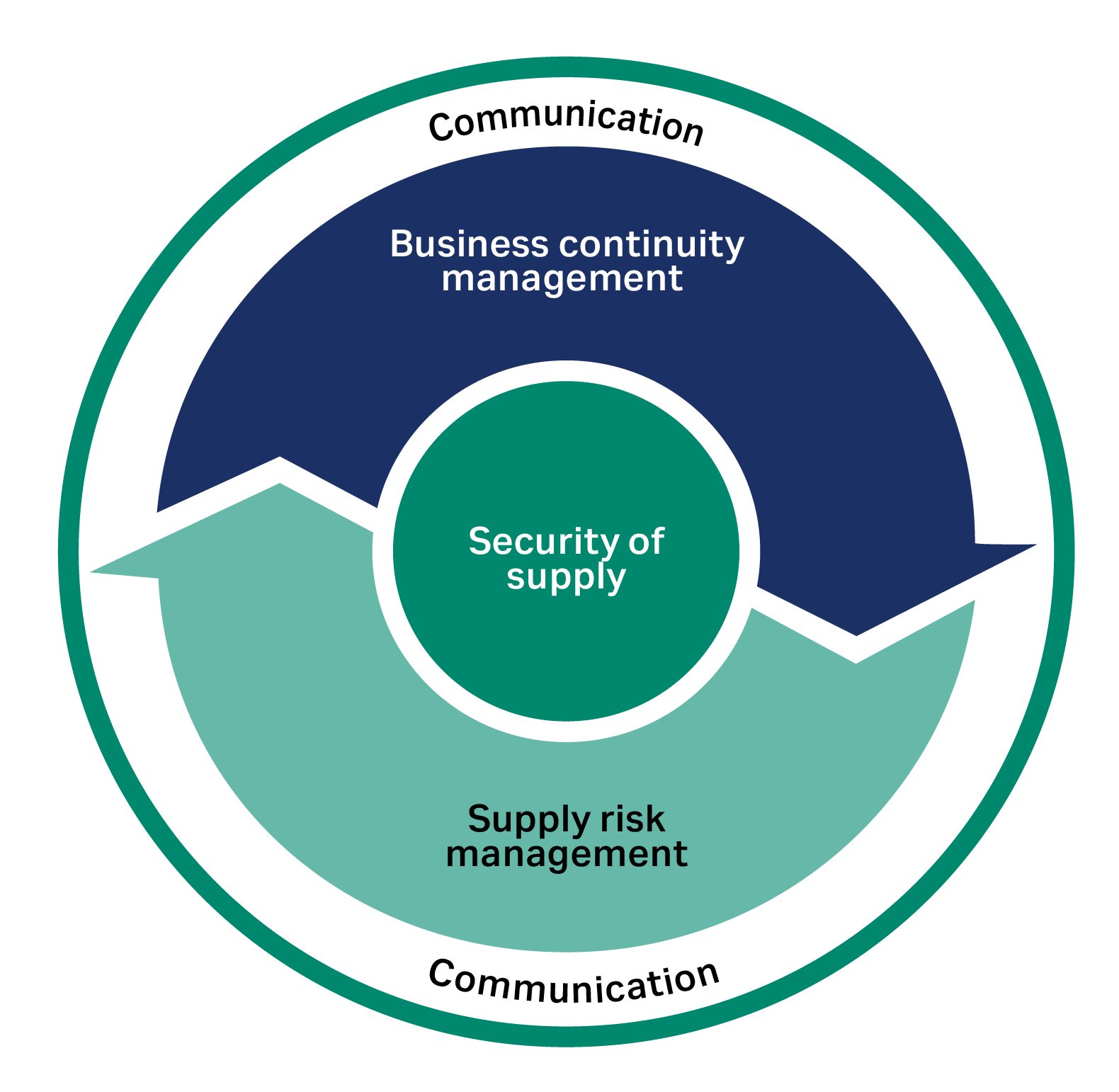
Securing supply is dependent not only on managing business continuity, but also on managing supply risk and maintaining transparent and frequent communication. Business continuity management is essentially about focusing on internal matters such as manufacturing sites and operations, while supply risk management helps identify effective activities to mitigate supplier risks (i.e., reduce the number of high-risk suppliers).
Communication is the glue that holds the whole enterprise together and ensures security of supply through a disruption. It is vital that accurate information is passed internally and externally, and reaches customers quickly during an external crisis, to establish trusted supplier and customer partnerships.
Fig 4. The three pillars of security of supply: supply risk management, business continuity management, and communication.
But communication goes both ways – without support and information from customers, it is not possible to maintain uninterrupted business and supply. Thus, customer input plays a pivotal role in the management process.
At Cytiva, we encourage our customers to share more information about their business forecasting, so that we are well prepared to accommodate requirements and changes in their business needs. Updates from customers go a long way in adjusting to mid-/long-term growth, because we can plan our manufacturing capacity accordingly and get a head start on ramping up facilities to meet the demands of the future. Currently, as part of our BCM program, we have processes in place (which are being continuously optimized) to capture forecasting data and get timely input from customers.
Implementing BCM: requirements, challenges, and thoughts for the future
The most important requirements for implementing BCM successfully are leadership commitment and awareness in the work culture. Top-level management has to drive the process, from making the initial decision, to allocating resources such as managers and project leaders to facilitate the program and provide supervision at the different sites. But the program is actually built from the bottom up, because you need support and awareness from employees at every level.
We have refined our BCM processes through implementing them across seven different sites – learning the challenges from one site and incorporating solutions at the next site. It is an added challenge when working globally, as you need to accommodate and adjust to different work cultures and ways of working.
There is no end to the BCM program – we must continually optimize our processes to tackle new problems that arise, such as issues with new materials, new production lines, new regulations, etc. It is a work in progress that needs to be updated and improved, at least on a yearly basis.
More information about Security of supply at Cytiva
References
- Hutt. R., The economic effects of COVID-19 around the world, World Economic Forum (Feb. 2020).
- Tangen. S., Austin, D., Business Continuity - ISO 22301 When Things Go Seriously Wrong, International Organization for Standardization (June 2012).
- Dry powdered medium milling equivalency study, Cytiva.