Development of robust purification processes requires significant time and resources. The quality-by-design initiative by the FDA puts additional demands on process development work because a higher degree of process understanding is needed. The choice of tools is therefore critical to keep development time short, while increasing the amount of information available and keeping sample consumption low.
The choice of the tool will of course differ depending on whether you want to screen for the most suitable buffer conditions in early process development or perform process robustness studies.
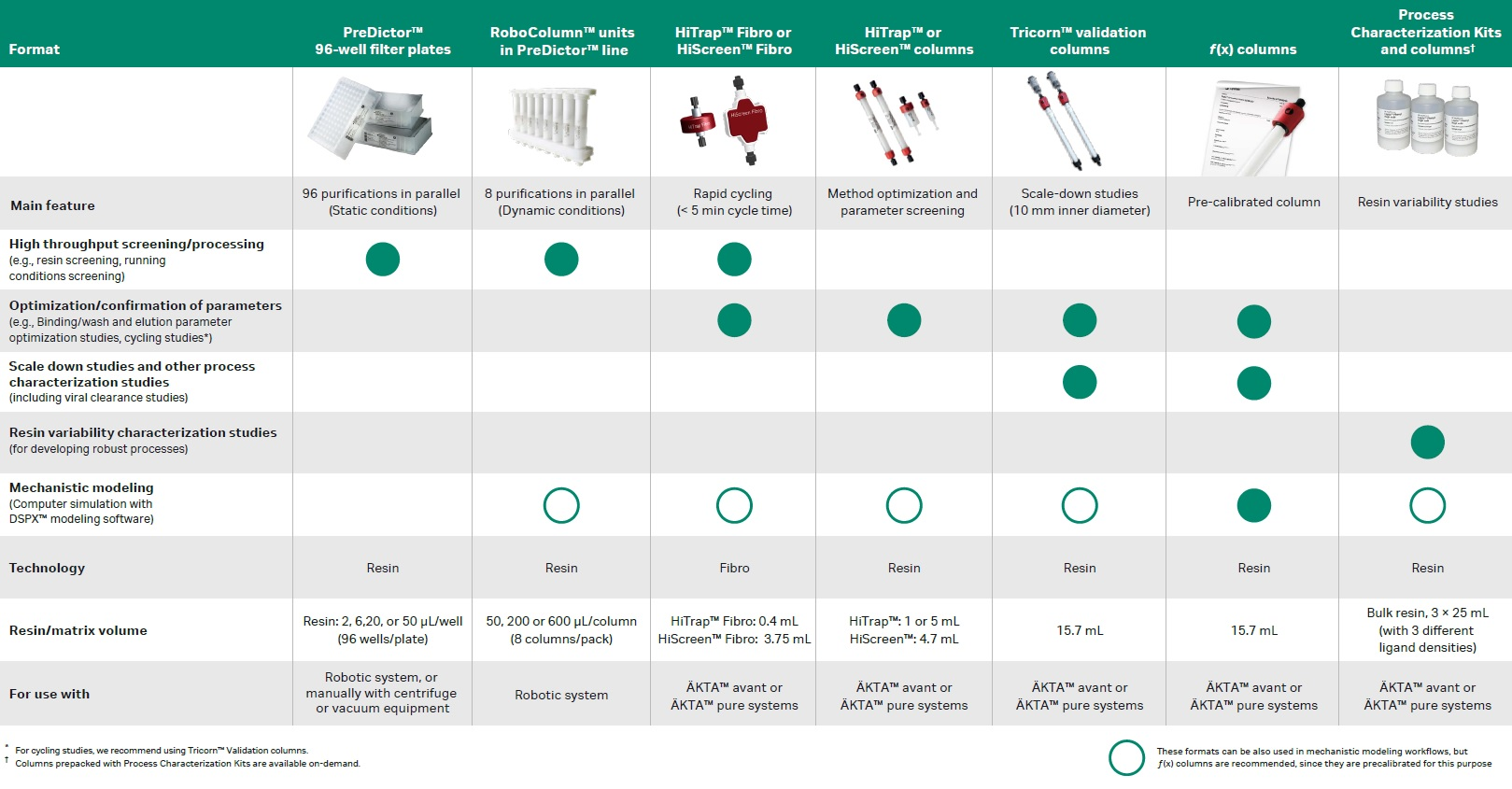
The table below helps select the chromatography tools available to help you make your process development as efficient as possible.
PreDictor™ 96-well plates
PreDictor™ plates are disposable, 96-well filter plates for high throughput process development (HTPD). They are designed for parallel screening of resins and chromatographic conditions such as binding (static), wash, and elution conditions. They can be used with centrifugation or vacuum manifold, manually or in automated robotic liquid handling systems.
Three main categories of PreDictor™ plates:
- Single resin plates: same volume, same resin in all wells of the plate for binding, wash, or elution studies on a single resin.
- Resin screening plates: for binding, wash, or elution studies on multiple chromatography resins.
- Adsorption isotherm plates: contain a single resin in all wells, but with different volumes in different wells.
PreDictor™ RoboColumn™ units
PreDictor™ RoboColumn™ units are prepacked, miniaturized columns that support HTPD using a robotic liquid handling workstation, for fully automated and parallel chromatographic separations. HTPD work can be performed using PreDictor™ RoboColumn™ units alone, or as a complement to PreDictor™ 96-well filter plates. Dynamic binding capacity data can be obtained with PreDictor™ RoboColumn™.
HiTrap™ Fibro and HiScreen™ Fibro units
HiTrap™ Fibro and HiScreen™ Fibro are ready-to-use fiber adsorbent units designed for research and early process development and are optimized for screening and optimization of process conditions. The open pore structure allows for high-binding capacities at very short residence times, and purification cycle times are performed in minutes instead of hours needed for resin-based chromatography. You can operate both formats with either a peristaltic pump or an ÄKTA™ chromatography system.
HiTrap™ and HiScreen™ columns
The columns can be operated with a syringe (HiTrap™ columns), a peristaltic pump, or a chromatography system such as the ÄKTA™ systems. The column formats are well-suited for preparative purifications as well as for screening and optimization of purification conditions when developing a new method.
Validation columns
High-quality, prepacked, scale-down columns for robust and reproducible process validation and process development. Because of their dimensions—narrow 10 mm inner diameter, combined with 20 cm bed height typically used in biomanufacturing—these columns are especially suited for scale-down studies such as viral clearance studies.
f(x) columns for mechanistic modeling of chromatography
Prepacked and precalibrated chromatography columns for fast and accurate mechanistic modeling during downstream process development.
Process Characterization Kits
These kits allow you to study the potential effect that resin ligand density might have on the process outcome. The kits consist of three 25 mL bottles with different ligand densities, representing a low, average, and high value in the manufacturing envelope to get critical insights on resin variation within ligand density specification interval.