Introduction: The importance of reliable cryogenic shipping
Timing and temperature are crucial factors when delivering sensitive cell therapies to critically ill patients. Whether the treatment is a commercial formulation or part of a clinical trial, there are typically multiple sites and partners involved in the delivery process, making logistics complex. While cryopreservation affords extra time and flexibility, the process comes with unique challenges.
Cryogenic transport of cell therapy starting material and finished product is currently carried out in dry shippers that use liquid nitrogen (LN2). It can be difficult, however, to confidently predict how cold contents can be kept in these devices, and for how long.
Held in a ‘dry’ phase within a sponge-like material, the rate of evaporation determines the cryogenic stand-by time for a LN2-based shipper and depends on the unit’s age, the quality of cleaning of the sponge, the condition of its seals, and positional factors in transport, such as tilting. Once the LN2 has evaporated, these systems have a fast warm-up profile1 and require a charge process that makes managing unforeseen events and delays during transit challenging. They also require dedicated infrastructure for cleaning. The finite nature of LN2 and associated limitations put pressure on scheduling logistics and can define the timing of a therapy’s administration, which often occurs as soon as the final product is received at the clinical site.
To address these challenges, Cytiva developed the VIA Capsule™ system. This smart, liquid nitrogen-free cryogenic shipper offers the conditions and functionality required to maintain cell viability before, during, and after transport. Instead of LN2, the system is charged using electricity from a standard outlet. This charge process powers a cryocooler Stirling engine which pre-cools the VIA Capsule™ shipper to -192°C and, while connected to a standard electricity source, can maintain the temperature of the shipper’s chamber at cryogenic levels.
In transit, the VIA Capsule™ shipper’s high quality vacuum and insulation give a stand-by time of at least five days below the key threshold of -120°C2,3 from full charge. Should weather or another unexpected event cause an unscheduled delay during shipment, access to a power outlet and a cryocooler allows the shipper to maintain the cryopreserved sample’s temperature until the shipment can continue. The ability to hold the shipper unit at cryogenic temperatures while “on-charge” removes the scheduling pressures inherent to LN2-based shipments. A cryocooler-connected VIA Capsule™ shipper sits primed and ready for the sample, providing a temporary storage facility (validated up to 6.5 weeks). Currently just over half of the sites which are sent cryogenic shipments have their own on-site temporary cryogenic storage facilities4. This capability eliminates the requirement to transfer the sample to temporary cryogenic storage facilities or to a back-up pre-charged dry shipper, if a delay occurs, simplifying the chain of custody and reducing the risk of sample mix-ups.
CSL Behring, a biopharmaceutical company that manufacturers biologic therapeutics, evaluated the potential logistical benefits of the VIA Capsule™ system for the cryogenic transportation and short-term storage of cryopreserved cell products. They gathered data from two independent shipments from Pasadena, California (West Coast) to King of Prussia, Pennsylvania (East Coast) and back to Pasadena, California using the VIA Capsule™ shipper and compared results to control samples that remained at the original site in LN2. This exercise was designed to determine if there was any variation in biological quality that could be attributed to using the VIA Capsule™ shipper for sample transport.
Methods: CSL Behring’s cryogenic transport trial using the VIA Capsule™ system
Sample preparation and cryopreservation:
On two separate occasions (designated Trial 1 and Trial 2), CSL Behring formulated CD34+ enriched fractions at a final dimethyl sulfoxide (DMSO) concentration of 5% (CryoStor™ CS5 media, STEMCELL Technologies). Each formulation was then aliquoted in 2× 250 mL-type cryobags for cryopreservation. Cyrobags were frozen using a VIA Freeze™ Quad controlled-rate freezer with a standard protocol of 1°C/min to -100°C and transferred to vapor-phase LN2 storage. One of the two samples would be loaded in the VIA Capsule™ shipper for cryogenic transport, while the other would be maintained in cryogenic storage as an unshipped control.
- Trial 1 used 2× cryobag samples frozen at 2 × 106 cells/mL in a final volume of 33 mL
- Trial 2 used 2× cryobag samples frozen at 1 × 106 cells/mL in a final volume of 30 mL
Evaluation trials:
CSL Behring booked shipments with World Courier using Chronicle™ software. The Chronicle™ platform offers electronic standard operating procedures (eSOPs) for loading and unloading the VIA Capsule™ shipper, with a QR code-based system digitally linking each shipment to the specific sample being shipped and the shipper used along with associated shipment documents. This process allowed for chain of identity and chain of custody to be digitally recorded. A GPS tracker (PT300D, Sendum) associated with the VIA Capsule™ system (shipper base: SBA-1219004; cryocooler: SEN1219004) and integrated into the Chronicle™ platform allowed CSL Behring to record and follow the shipment’s location while monitoring other critical parameters in real time, such as temperature of the chamber, ambient temperature, tilt angle, humidity, and light or shock data, all from the Chronicle™ shipment page. An additional feature allows alerts to be sent to predetermined users by email and/or text message when critical shipping parameters reach specified threshold levels, enabling fast action and response in the event of an issue. It’s also worth noting that World Courier strategically positions VIA Capsule™ charge hubs with cryocoolers and staff trained to use them throughout their network.
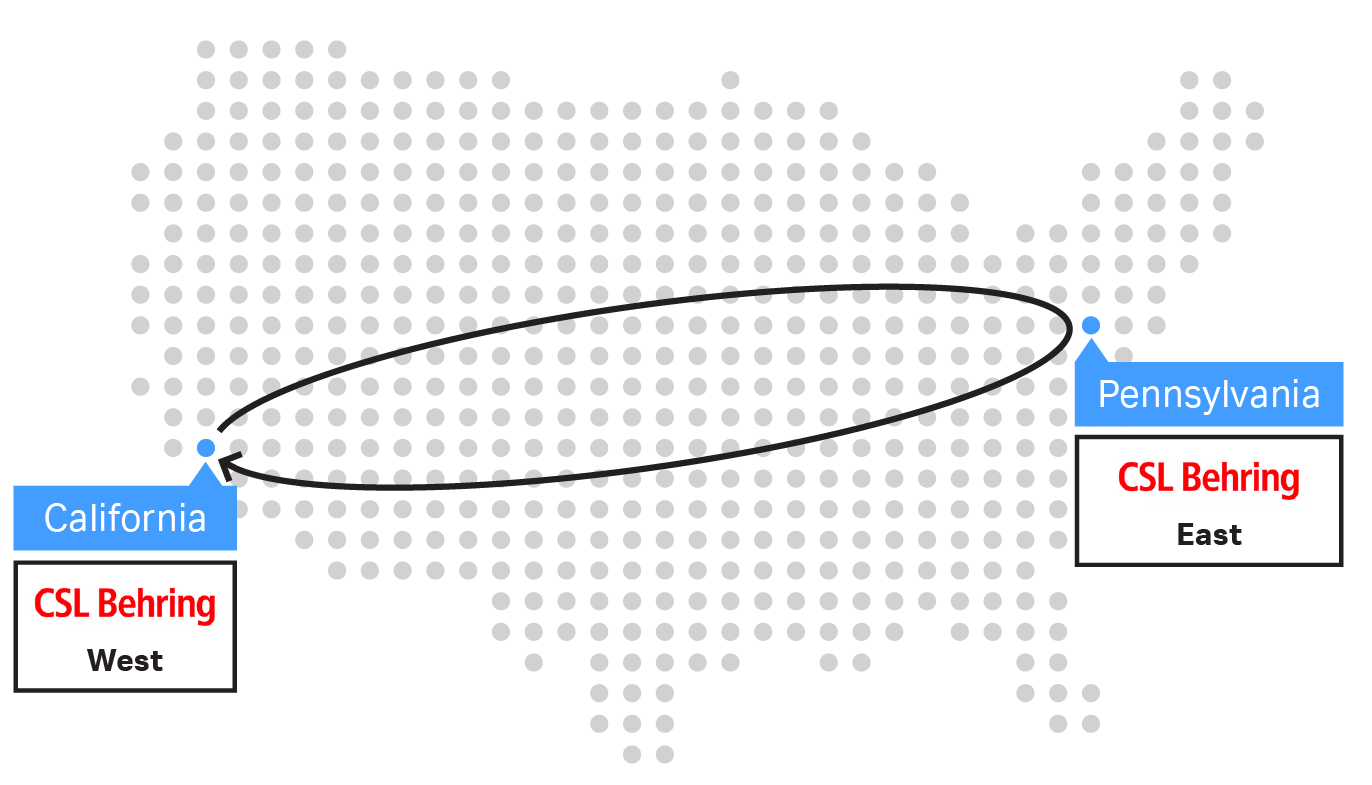
Trial 1 consisted of a direct return shipment from the West Coast to the East Coast and back, lasting three days. Trial 2’s shipment followed the same route but also included a short-term storage period of five days at the East Coast site, lasting seven days total. The short-term storage simulated a logistic scenario in which the receiving site did not have LN2 facilities. To simulate short-term storage, a cryocooler delivered a charge to the VIA Capsule™ shipper before returning to the original site on the West Coast. Cryobags were immediately returned to vapor-phase LN2 storage upon return to the original site.
 B)
B)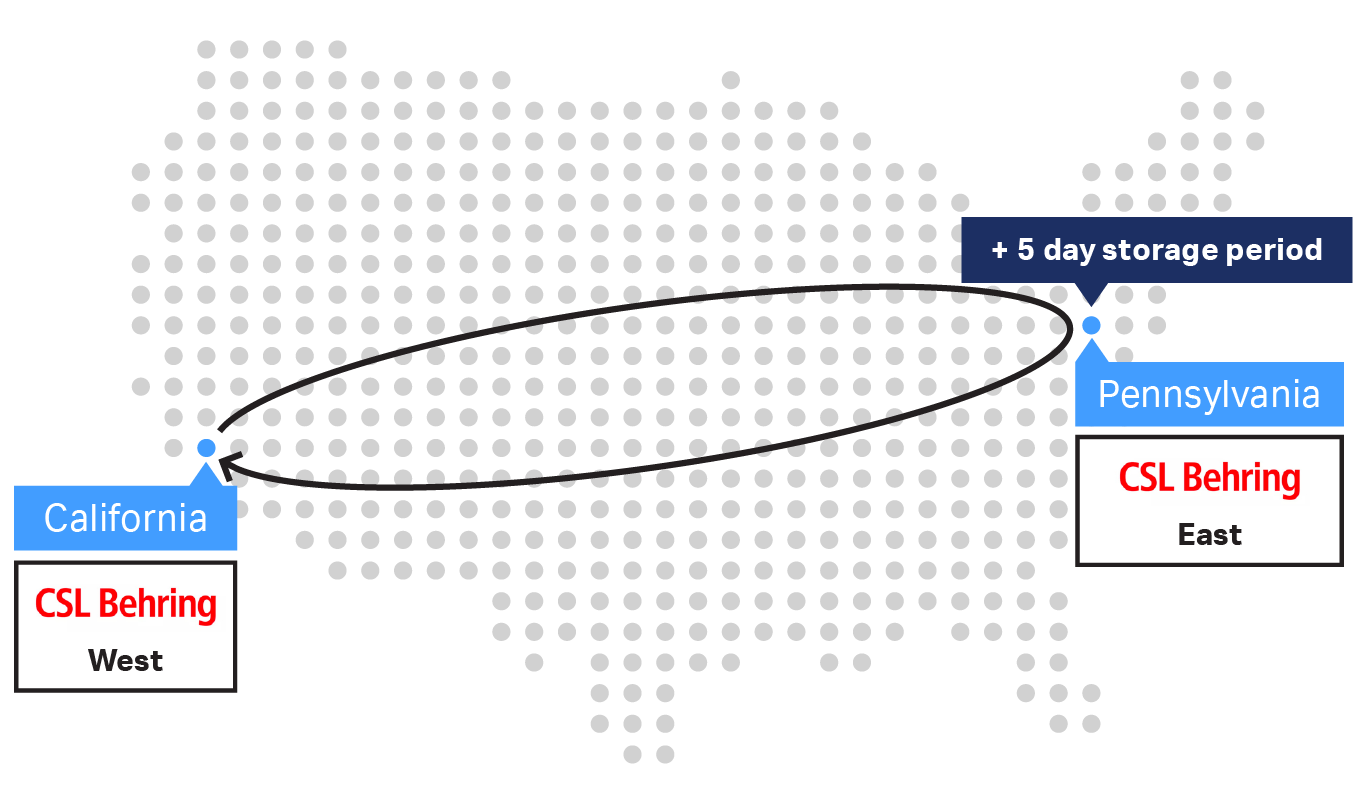
Fig 1. USA West Coast to East Coast sample shipments with the VIA Capsule™ system and control sample maintained in LN2 storage at the West Coast site throughout. (A) Trial 1 direct return shipment over 3 days. (B) Trial 2 lasted a total of 7 days, including five-day storage at the East Coast site.
Post shipment analyses:
CSL Behring operators used a VIA Thaw™ CB1000 with standard protocol to thaw each cryobag. They then analyzed the samples to determine cell count, viability, and recovery before plating for further bio-analysis.
In summary, thawed cryobags were spiked and cells were collected in a 50 mL syringe. Cells were added to 20 mL of X-Vivo™ media plus 2% human serum albumin (HSA) in two 50 mL centrifuge tubes and spun at 300×g for 10 minutes. Supernatants were discarded and pellets resuspended in 20 mL of X-Vivo™ media plus 2% HSA to determine cell count and viability. Cells were counted with an automated cell counter (Nexcelom Cellometer). For CD34 enumeration on day 2, cells were plated in a 24-well plate at ~106 cells per well in duplicate. The plate was incubated overnight at 37°C, 5% CO2. On day 2, each well was counted once for quantitation and viability. The CD34+ population was quantified for each well. Briefly, 100 µL of cell suspension was stained with 10 µL of CD34/CD45 antibody cocktail and 10 µL of 7-AAD. The suspension was then lysed and analyzed on a flow cytometer (MACSQuant™ Analyzer 10, Miltenyi Biotec) according to an enumeration technique that produces an absolute count excluding dead cells and CD34- events.
Acceptance criteria were qualitative, as the few number of samples did not allow for definitive statistical analysis. Data from of the VIA Capsule™ shipper was compared against the control that remained on-site. As a baseline for comparison, criteria were set at >70% recovery of cells and >80% viability upon thaw.
Results and discussion: VIA Capsule™ performance in shipping cryopreserved cell samples
Before the shipment process began and with minimal remote training from Cytiva, the CSL Behring team seamlessly managed the charging of the VIA Capsule™ system and learned how to use the smart monitor. Cytiva provided remote support for scheduling the shipments through Chronicle™ software, including booking the first return shipments as per the project agreement. No further assistance was necessary for booking the second return shipments. The team followed a Chronicle™ eSOP to deliver the optimal pack-out process prior to shipment.
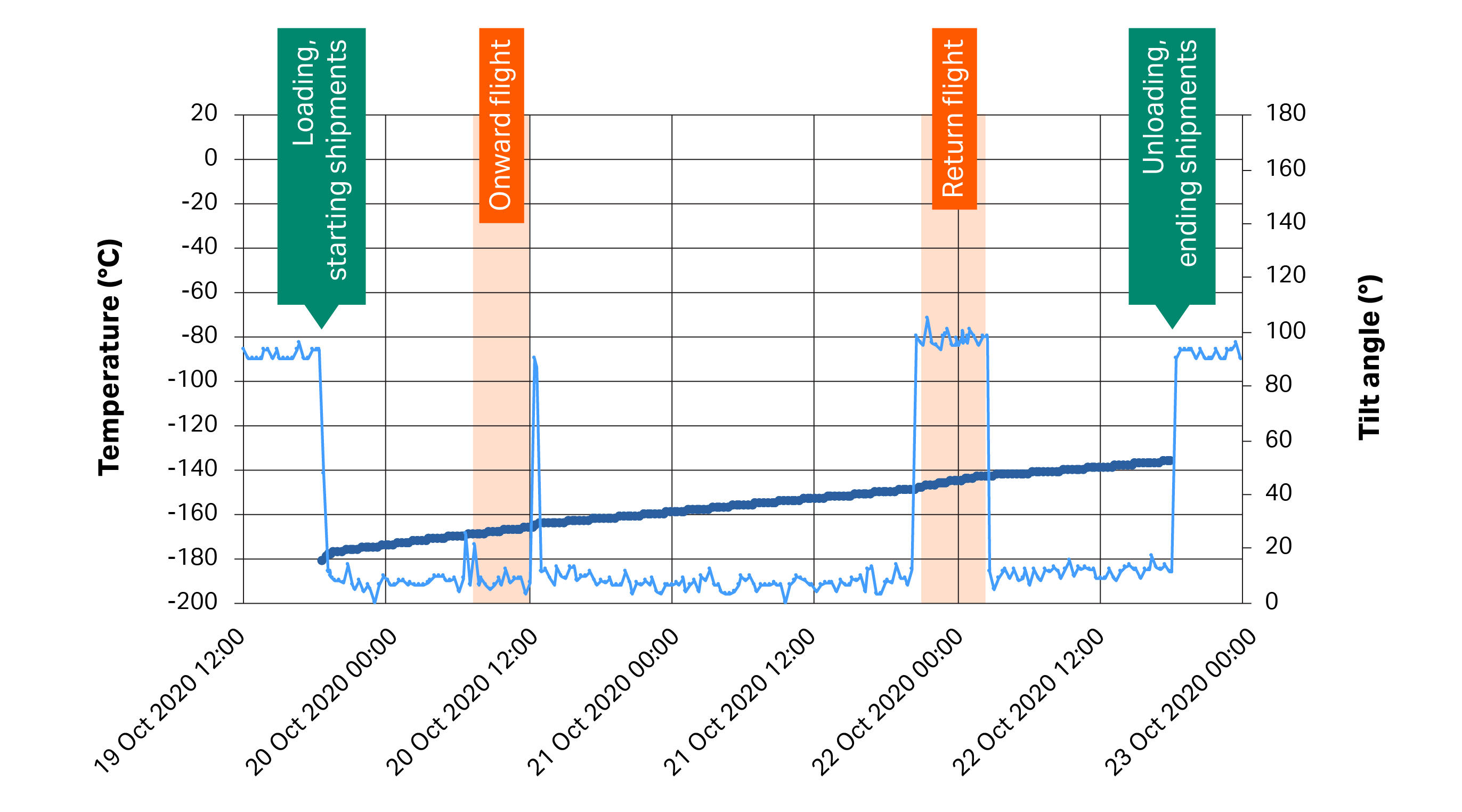
During transit, the VIA Capsule™ shipper maintained temperatures well below -120°C, despite having been heavily mishandled. Tilt data from Trial 1 shows that the unit was tilted on several occasions and kept on its side throughout one of the flights when returning from the East Coast (tilt values of approximately 90°, see Fig 2). However, this positioning had little effect on stand-by time, with a warming rate of 0.9°C/h during this 6.5 h period, compared to an overall warming rate of 0.6°C/h throughout the entire four-day transit period.
Fig 2. Data for chamber temperature (thick blue lines) and tilt (thin blue lines) of the VIA Capsule™ system during Trial 1 as shipped from the West Coast to the East Coast and back. Key events, including loading of the shipper with a cryopreserved sample, its unloading, and flights, are highlighted in orange text for reference.
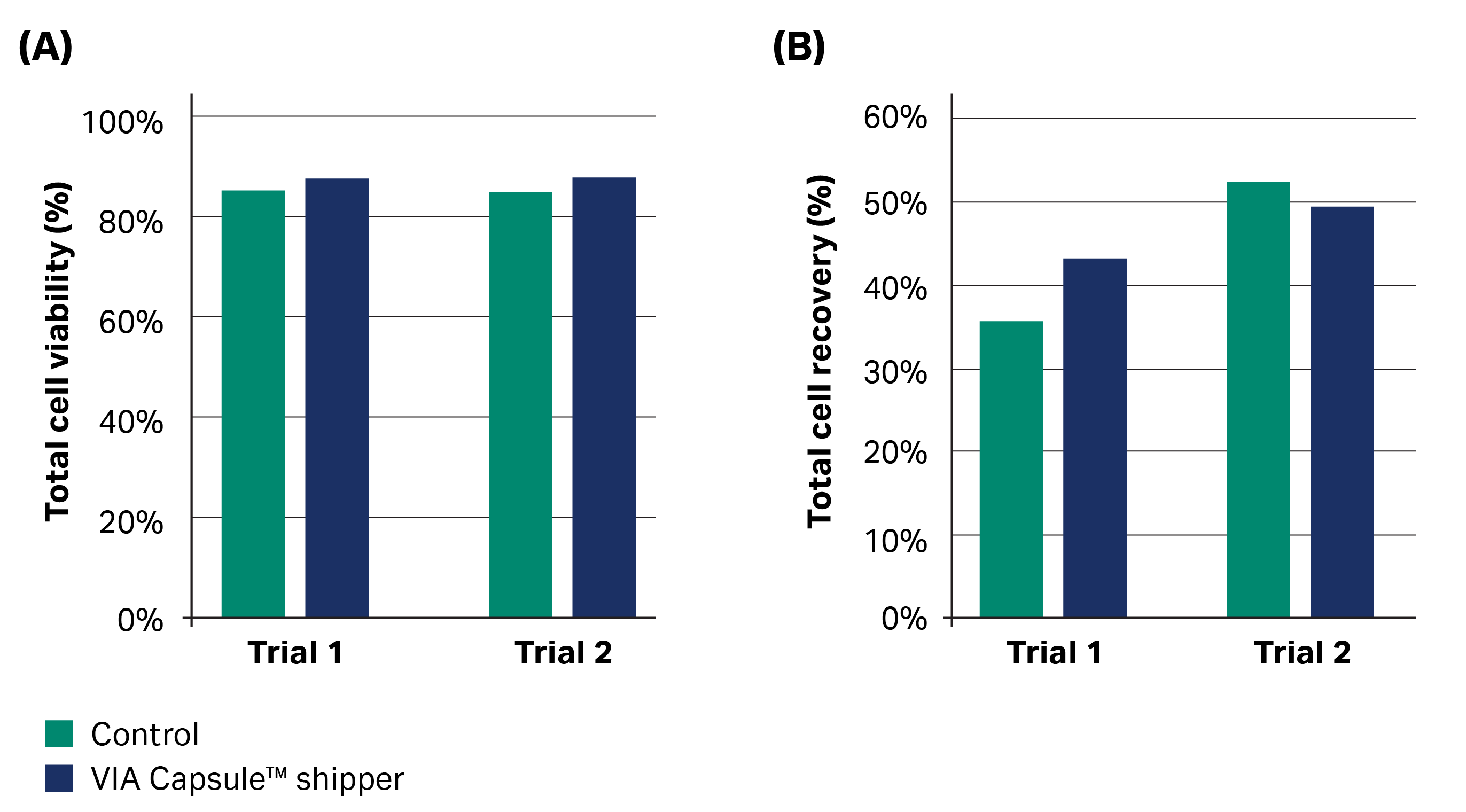
There were no clear differences in viability or recovery between the shipped samples and those maintained in LN2 storage, with 85% to 88% viability immediately upon thaw for all samples (n = 2) (Fig 3A, acceptance criteria 80%). Following Trial 1, recovery of total cells was 36% for the control and 43% for the VIA Capsule™ condition (Fig 3B). After Trial 2, recovery of total cells was 52% for the control sample and 49% for the VIA Capsule™ condition (Fig 3B). Cell recovery values were lower than the 70% acceptance criteria; however, this was true for the transported samples and controls. The poor cell recoveries were therefore likely due to an issue occurring before or at cryopreservation and unrelated to the shipments.
Fig 3. Cell viability (A) and recovery (B) upon thawing for control cryobag samples that remained in LN2 storage (green) versus cryobag samples that were transported in the VIA Capsule™ system (blue).
To evaluate any downstream effects of the shipping methods, cells were recovered from cryopreservation overnight in base culture media plus 2% HSA, and analyses were performed again on day 2. Similar trends in cell counts and viability persisted (data not shown), as well as for total viable CD34+ cells (Fig 4). Additionally, there were no differences in relative purity between samples for either trial.
Fig 4. Total cell number (A) and total viable CD34+ cells excluding dead cells and CD34- events (B) present in overnight cultures post-thawing of un-transported cryobag samples that remained in LN2 storage (green) and of cryobag samples that were transported in the VIA Capsule™ system (blue).
CSL Behring’s conclusions
The data gathered from the two evaluation trials suggests no differences in post-thaw cellular parameters between transported and control samples. These results highlight the ability of the VIA Capsule™ system to maintain cryopreserved cellular products within a cryogenic temperature range.
An electronic, mechanically cooled cryo-shipper offers a substantial logistical advantage when a receiving site is unequipped with LN2 capabilities. Provided there is A/C power available for charging the device, the VIA Capsule™ system is able to maintain cryogenic temperatures without teams having to worry about how long it will take to unload the sample or product.
Additional advantages of the VIA Capsule™ shipper include a smaller physical footprint, allowing for quicker and simpler sample handling and loading versus an equivalent dry shipper. Shipments utilizing the VIA Capsule™ system would also not have to rely on third-party dry shipper availability.
With the many logistical benefits of the VIA Capsule™ system, this LN2-free shipper from Cytiva emerges as a viable cryogenic shipping solution for cryopreserved cell products.
Find out more about the VIA Capsule™ system.
References
- Pomeroy, K.O., Reed, M.L., LoManto, B. et al. Cryostorage tank failures: temperature and volume loss over time after induced failure by removal of insulative vacuum. J Assist Reprod Genet. 2019;(36), 2271–2278.
- Meneghel J, Kilbride P, Morris JG, et al. Physical events occurring during the cryopreservation of immortalized human T cells. PLoS One. 2019;14(5):e0217304. 2019 May 23. doi:10.1371/journal.pone.0217304.
- Meneghel J, Kilbride P, Morris GJ. Cryopreservation as a Key Element in the Successful Delivery of Cell-Based Therapies-A Review. Front Med (Lausanne). 2020;7:592242. 2020 Nov 26. doi:10.3389/fmed.2020.592242.
- JIGSAW Market Research data (June 2020). Responses of 25 cell therapy manufacturer respondents.
Ordering Information
| Product | Description | Product code |
| Main instrument | VIA Capsule™ Cryocooler | 29435429 |
| Main instrument | VIA Capsule™ Shipper | 29435428 |
| Accessory | Sendum Tracking System | 29435205 |
| Accessory | Thermal Core Bag | 29483894 |
| Accessory | Thermal Core SBS | 29484272 |
| Accessory | Thermal Core Removal Tool | 29484296 |
| Accessory | Thermal Core Screw Driver Assembly | 29492489 |
| Accessory | VIA Capsule™ Shipper Cap | 29463250 |
| Consumable | Sample Loader SBS | 29435432 |
| Consumable | Sample Loader Cryobag | 29435430 |
| Consumable | Transport Box Shipper | 29441698 |
| Consumable | Transport Box Cryocooler | 29462958 |
Acknowledgements
This work was led by CSL Behring and Cytiva in collaboration with World Courier; data, evidence, and conclusions provided with kind permission from CSL Behring.