Avoiding pitfalls in HPLC sample prep
HPLC pain points—Part I
High-performance liquid chromatography (HPLC) systems are widely used in analytical laboratories. Ensuring that systems are in good working order minimizes equipment downtime and increases lab efficiency.
Problems in an HPLC system can arise from several sources. The source of a given problem can often be identified by looking at the system itself or by checking downstream, by looking at the chromatogram.
Recognizing visible signs of in-system issues can identify potential problems and determine solutions that extend the usable lifetime of a column. Maximizing column lifetime has benefits in data quality, cost, and in reducing downtime.
What problems can occur in an HPLC system?
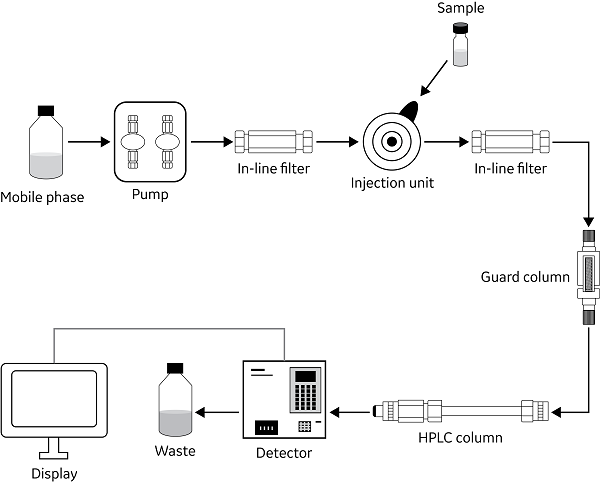
An HPLC system consists of multiple components, including an injector, flow path, pump, column, and detector unit.
A mobile phase (e.g., buffer) enters the system and passes through a pump that pressurizes the system. This mobile phase routes through to the injector to pick up the sample and carry it to the column. Both the sample and mobile phase are applied to the column for analyte separation before passing through the detector.
Problems occurring at any point in this path might influence the success of a run or affect the column quality and lifetime. Visible signs of issues from the HPLC system include:
- Increased back pressure
- Leakages and loss of pressure
- Inability to maintain a consistent flow rate
What causes these problems?
Particle contamination
Particles can make their way into an HPLC system via the sample or the mobile phase. These physical contaminants might be dust from the environment or other solids, such as precipitated protein and undissolved buffer components.
The result of physical contamination over time is two-fold:
- Blockages in the system, most likely at the HPLC column frit or the column itself.
- Wear and tear to the system components, including scratches, adding further particle contamination.
Increased back pressure is a clear indicator that part of the system is obstructed. The pump will try to maintain a fixed, accurate flow rate despite the blockage. But the instrument might eventually shut down to prevent damage.
Blockage of the column or frit also affects the uniformity of sample loading onto the column. These effects are visible from the chromatogram and explained in more detail in part 2 of the HPLC troubleshooting guide.
System components likely to suffer damage from particle contamination include injector valves, pump components, connectors, seals, and reciprocating parts. Depending on the location, damage might be visible as increased back pressure, leakages, or an inconsistent flow rate.
In addition to affecting quality of results, particle damage might require additional servicing/maintenance and column replacement. These delays and system downtime reduce lab efficiency.
Chemical contamination
Physical contamination of the HPLC system can build up over time to cause damage, resulting in the eventual need to replace columns and parts. However, chemical contamination has more immediate effects.
Some chemicals might bind irreversibly to the column, causing an increase in back pressure. Chemicals might also result in solubilization of the column resin, affecting separation and data quality. In both situations, the column will need replacing, which brings additional costs and equipment downtime.
Depending on the source of contamination, physical damage to the HPLC system and its components is another concern for users. This damage is also associated with equipment downtime and increased costs for replacement and servicing.
Dissolved gas
Although dissolved gas can be present in both the sample and the mobile phase, gas in the mobile phase generally causes many issues in an HPLC system.
If dissolved gas reaches the detector, bubbles can form as it comes out of solution, presenting as a drop in pressure. The interference from gas bubbles will be visible from the chromatogram. Part 2 of the HPLC troubleshooting guide covers these effects in more detail.
What solutions are available to minimize in-system problems?
Particle contamination is a common issue affecting HPLC system components.
Filtration of the sample and mobile phase can help to combat particle contamination by reducing blockages and the need to replace system components, including the column.
Filtering the mobile phase through a 0.45 µm filter can reduce particles entering the system.
There are many considerations for sample preparation and filtration, but it is generally straightforward to remove particulates using a syringe or syringeless filter device. Prefiltration or a multilayer syringe filter can be an effective option for thick or particulate-laden samples. Using a precolumn filter and guard column can also aid the removal of particles to minimize damage and increase lab efficiency.
Inherently, columns and other components of HPLC systems have limited lifetimes.
HPLC troubleshooting part 2: UV analysis
Maintaining a high-performance liquid chromatography (HPLC) system in good condition provides users with consistent data quality and accuracy. A well-functioning system also minimizes downtime, helping to maintain lab efficiency.
As discussed in Part I, problems in HPLC systems might arise from several sources. When using a UV detector, the chromatogram can indicate the presence of a problem and provides clues about where to find the cause.
What problems can show up on a chromatogram?
A UV chromatogram plots absorbance at the selected wavelength over time. As the mobile phase carries the sample through the HPLC column, peaks indicate the relative abundance of compounds eluting from the column.
In the preferred scenario, sharp uniform peaks with clear separation and high signal to noise ratio will allow the identification of each analyte. Sometimes, however, the chromatogram might show distortions, including:
- Shouldered peaks
- Twin peaks or split peaks
- Tailed peaks
- Low signal to noise ratio
These distortions can add complexity to analysis, indicate upstream problems, and affect data accuracy and reliability.
What causes chromatogram distortions?
Particle contamination
Physical contamination of the HPLC system and column can present as peak tailing, splitting, and shouldering.
On an uncontaminated column, sample application is immediate. Uniform application in a short timeframe allows efficient separation, supporting consistent and accurate results.
Contamination of the column or frit can increase the time taken to apply a sample in comparison with a clean column. This increased time can lead to poor resolution of the analytes.
Chemical contamination
Any chemicals or compounds that absorb at the same wavelength as the analyte can distort chromatograms and confuse analysis. If contaminants elute at a similar time as the analyte, the chromatogram might report inaccurate absorbance values or unexpected peaks. Any contaminants in the mobile phase can also cause background noise.
Possible sources of contamination include extractables from filter devices or other system components. Filter materials that release extractables or housings with low solvent resistance have the potential to interfere with the chromatogram.
However, extractables only become a problem if they are detectable and co-elute with the analyte of interest. Other sources of contamination might include residue on glassware from previous experiments.
Dissolved gas
High pressure in the HPLC system keeps gas dissolved. Generally, dissolved gas is only likely to cause problems if it comes out of solution to form bubbles. These bubbles are most likely to arise at the detector where the pressure drops.
Detectors vary in their sensitivity to gas bubbles, but the effect is often evident as baseline noise on the chromatogram, leading to a low signal-to-noise ratio.
A low signal to noise ratio is a common indicator of a high proportion of dissolved gas. This noise can affect the reliability of peak identification, making HPLC analysis particularly difficult when the analyte is limited and has a low absorbance level.
How can problems in HPLC analysis be minimized?
Reducing sources of contamination
Filtering the sample can reduce particle contamination by preventing undissolved particulates from entering the system.
Solvent compatibility and level of extractables are considerations in selecting an appropriate filter device. Running a comparative test with and without a standard in place of the sample can assess the effect of extractables on the chromatograph.
Reversing the solvent flow is also a common technique to clear particulates from the column and frit. However, note that this technique can disrupt packing and affect separation efficiency.
Sufficient HPLC column packing and column equilibration procedures can help make sure sample application is uniform. These actions can also reduce the likelihood of bubbles forming in the column.
Degassing
It is common practice to degas the mobile phase before mixing it with the sample. Degassing with a cellulose filter minimizes bubble formation at the detector.
Some HPLC systems incorporate a degasser, but an alternative option is vacuum filtration of the mobile phase before use. Degassing before each HPLC run also reduces the likelihood that gases dissolved in the mobile phase reservoir between runs will affect results.
The quality and resolution of UV chromatogram data can provide users with feedback to help them achieve and maintain high-quality HPLC data.