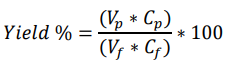Depth filtration for clarification of recombinant adeno-associated virus (rAAV)
Depth and membrane filters can offer a robust, cost-effective solution for clarification of recombinant adeno-associated virus over a wide range of scales from process development to commercial manufacturing. The work shown here provides guidance for how to select the optimal depth filter grade(s) to clarify AAV from either adherent or suspension bioreactors. Performance data is provided for depth filters and sterile filters including filter capacities, turbidity reduction, and AAV yields.
Depth filtration for clarification of rAAV in this study gave:
- Yields > 95% using an adherent cell culture platform.
- Yields > 90% for rAAV using a suspension cell culture platform.
Read more about clarification using depth filtration here.
Introduction
Recombinant adeno-associated virus (rAAV) is non-enveloped virus ~ 20 nm in diameter. It can package ~ 4.7 kilobases of DNA and shows relatively low immune response compared to retroviruses and adenoviruses. Furthermore, it is relatively stable under standard bioprocessing conditions.
Recombinant AAV is produced in host cells which can be grown either on substrates in adherent bioreactors, or in suspension in stirred-tank bioreactors. In typical rAAV production the product can be found both intra- and extracellularly, and many processes therefore include a cell lysis step to maximize product recovery. Similar to recombinant protein processing, the next step is clarifying the product from the complex mixture of impurities which includes cell debris, host-cell proteins, and DNA.
Depth filtration is a well-established technology for clarifying a product from a complex cell culture stream. Depth filters are typically made up of a mixture of cellulose, inorganic filter aids, and resin. They provide the ‘depth’ of filter media and wide pore size distribution necessary to retain the amount and wide size range of impurities present in the bioreactor. Depth filters are often followed by membrane filters to further remove fine particles as well as any bioburden in the process stream. Depth and membrane filters offer a robust, cost-effective solution for clarification over a wide range of scales.
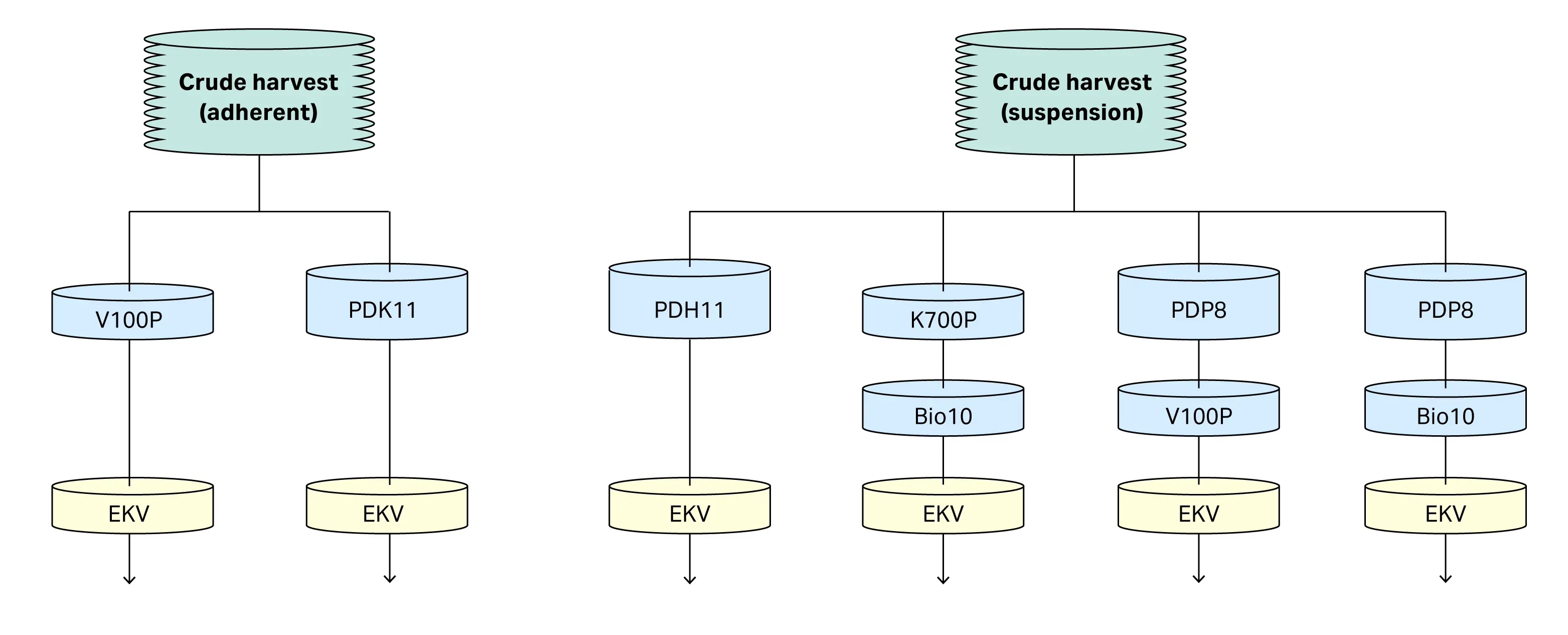
In this application note we evaluate combinations of depth and membrane filters for clarification of rAAV (see Fig 1). The impact of feed stream characteristics (rAAV serotype, adherent vs suspension, turbidity), filter characteristics (chemistry, pore size) and scale are evaluated for impact on clarification performance.
Fig 1. Combinations of Seitz™ depth and membrane filters, followed by Supor™ EKV sterilizing grade filters used in this study.
MATERIALS AND METHODS
Crude harvest supply via adherent and suspension cell culture
Adherent cell cultures
All rAAV5 used in this study was supplied through transient transfection of HEK293T cells. Adherent cultures were either produced with Corning CellSTACK culture chambers or in iCELLis™ Nano bioreactor systems. At the end of production, the culture supernatant was removed, the cells were lysed using a detergent, and the lysate collected from the bioreactor. The supernatant, wash, and lysate were combined and treated with an endonuclease prior to clarification. rAAV5 concentration in the crude harvest averaged 7.4 ± 2.0 × 109 gene copies (gc)/mL.
Suspension cell cultures
Suspension cultures producing rAAV5 were grown in benchtop stirred-tank bioreactors. At the end of production, the cells were lysed using a detergent and the pool was endonuclease treated prior to clarification. rAAV5 concentration in the crude harvest averaged 2.7 ± 0.64 × 1010 gc/mL.
rAAV8 and rAAV9 cell cultures
Cells producing rAAV8 and rAAV9 were procured from Vector BioLabs. Treatment of the cells was designed to mimic that of the rAAV5 harvest from the iCELLis™ Nano bioreactor systems. Cells were lysed with a detergent buffer, diluted into culture medium, and endonuclease treated prior to clarification.
Seitz™ depth filters used for rAAV clarification
The depth filters used in this work include several sheets from Seitz™ P-series and Seitz™ BIO-series filter lines.
Table 1. Depth filters used in this rAAV clarification study
| Depth Filters | Materials | Media |
| Seitz™ P-series | Cellulose, inorganic filter aids, binders | Single-layer media |
| Seitz™ BIO-series | Cellulose, binders | Single-layer media |
Seitz™ P-series filter sheets are made of a combination of cellulose, inorganic filter aids, and a binding resin. Note that the V100P is a sheet designed specifically for virus work that is low-charge and free of diatomaceous earth. These filters are available in single-layer format, or in dual-layer configurations in our HP series. The BIO-series filter line is made of cellulose and resin, and is ideal for low binding applications.
All Seitz™ depth filters were tested in Supracap™ 50 or Supracap™ 100 capsule formats. Supor™ EKV membrane was used as a sterilizing-grade filter after depth filtration. It is a dual-layer PES filter with an asymmetric pre-filtration upstream layer and a downstream symmetric 0.2 µm layer. All sterile filters were tested in Mini Kleenpak™ syringe filter, Mini Kleenpak™ 20 capsule, or Mini Kleenpak™ capsule formats.
Discover detailed information and characteristics of the Seitz™ depth filter sheets HERE.
All filtration work described in this study was run at constant flux on PendoTECH NFF control systems with peristaltic pumps on the feed lines. Pressures and filtrate volumes were recorded over time. In all trials, filters were equilibrated using a 1× phosphate buffered saline (PBS) solution prior to loading (we recommend flushing 100 L/m2 at 300 L/m2/h (LMH) for the filter grades used here to ensure full wetting of the pores). A post-use buffer chase of 1.5 × hold-up volumes was also employed to maximize virus recovery.
Experiments used to determine filter capacity were run in constant flow format to a terminal pressure of 0.7 bar (10 psi / 0.07 MPa). Adherent rAAV capacity trials were run at 200 LMH on the depth filters and 1000 LMH on the sterile filters. Suspension rAAV trials were run at 75 LMH on the depth filters and 500 LMH on the sterile filters. It is worth noting that depth filter flux rates at clarification can range from ~ 50 to 200 LMH and are often in the ~ 75 to 100 LMH range. Operating flux can impact depth filter performance (including capacity); however, this was outside the scope of this study.
Analytics
Pool turbidities were measured offline on a Hach 2100Q portable turbidimeter. rAAV concentrations were measured by a digital droplet polymerase chain reaction (ddPCR) method. Step yields were calculated using the equation below where Vf and Vp refer to feed and filtrate pool volumes and Cf and Cp refer to feed and filtrate pool concentrations respectively.
Fig 2. Retention ratings of filters evaluated in this work (the overlap in color between sheets three and two indicates the overlap in retention ratings between the sheets.
RESULTS AND DISCUSSIONS
Depth filter selection based on yield and turbidity
Initial screening work was undertaken with both adherent and suspension rAAV5 crude harvest feed streams to identify an appropriate filter train for clarification.
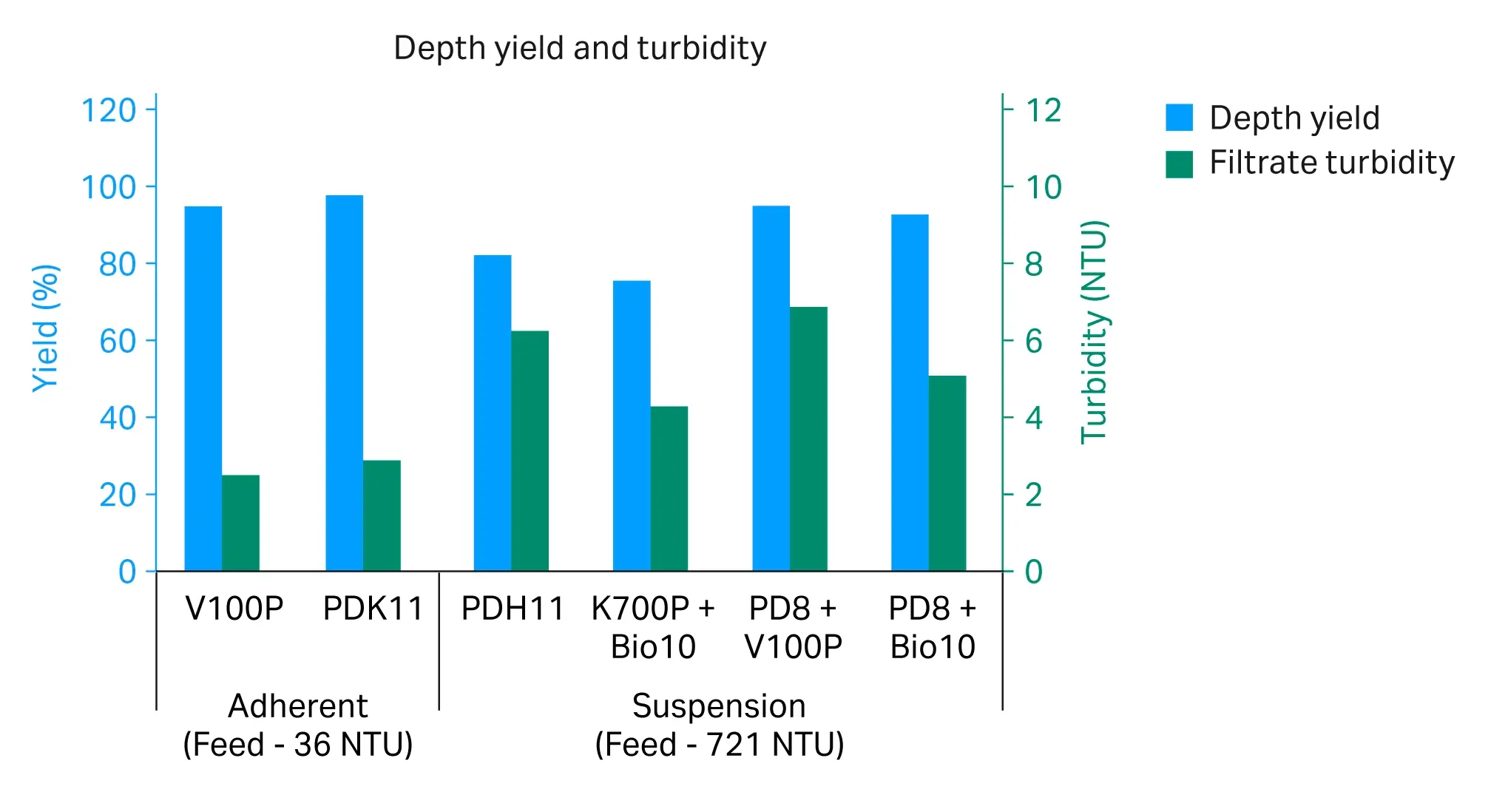
Select combinations of single- and dual-layer filter capsules were tested; retention ratings are shown in Figure 2. Filters were evaluated with the same feedstock run under the same flux conditions in constant flow studies to a terminal pressure of 0.7 bar (10 psi / 0.07 MPa). The turbidity of the adherent rAAV5 crude harvest was 36 nephelometric turbidity units (NTU).
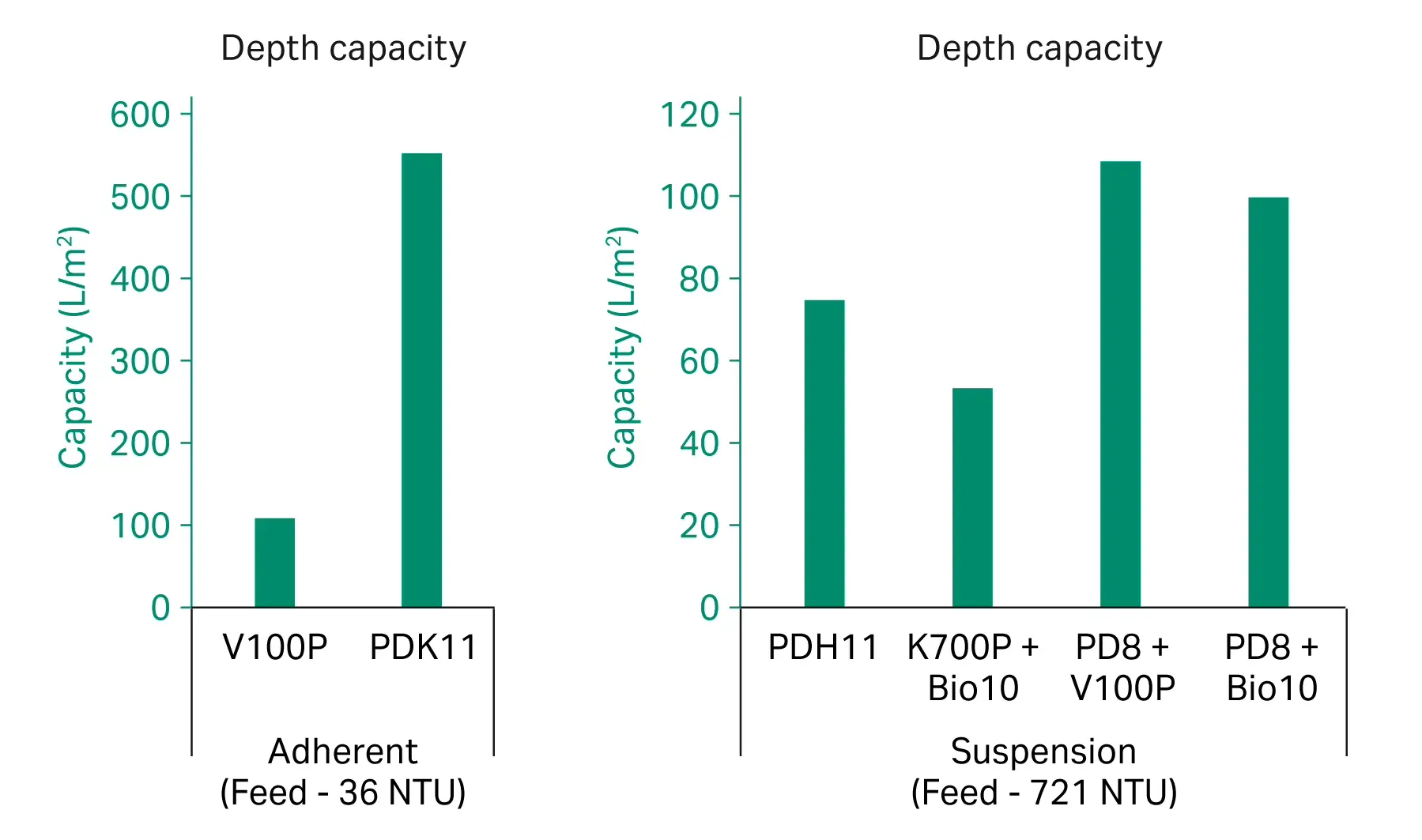
For clarification, both V100P and PDK11 filters demonstrated high yields (≥ 95%) and strong turbidity reduction (< 3 NTU in the filtered pool) as shown in figure 3. Here we also saw a significant capacity benefit from the dual-layer PDK11 (Fig 3), reaching >500 L/m2 at 0.7 bar (10 psi / 0.07 MPa). However, results also show that for a cleaner feedstream the single-layer V100P may also be a good option.
Both depth-filtered pools were taken offline and used to measure capacity on Supor™ EKV sterilizing-grade filters with both showing capacities > 1700 L/m2 and rAAV5 yields > 99%. The turbidity of the suspension rAAV5 crude harvest was 721 NTU.
For clarification, the two-stage filter combinations of PDP8 with either V100P or BIO 10 provided the highest capacity (≥ 100 L/m2) as shown in Figure 4, and rAAV5 yield of ≥ 93% as shown in Figure 3. Using the tighter Bio10 as a stage 2 filter provided slightly better turbidity reduction (5 NTU in the filtered pool) as seen in Figure 3, but also slightly lower capacity.
Depth-filtered pools were taken offline and used to measure capacity on Supor™ EKV sterilizing-grade filters. Unfortunately, there was not enough feed material to reach the pressure limit on the sterile filters, but both showed > 700 L/m2 throughput with pressure drops of ≤ 0.3 bar (5 psi / 0.03 MPa). The data shows both filter trains would provide a strong clarification solution for this feed. The best option for suspension rAAV will likely depend on small differences in process or feedstream as well as any manufacturing constraints.
Fig 3. Depth filter yields and pool turbidities from select filters run with adherent and suspension rAAV5 crude harvests.
Fig 4. Depth filter capacities from select filters run with adherent and suspension rAAV5 crude harvests.
Turbidity as a measure of process robustness in rAAV clarification
Performance of any clarification step can be influenced by many factors, including control over process conditions and variability in the feedstock. The latter can be particularly challenging as it is often difficult to measure and control important feed characteristics such as cell density, cell viability, and particle size distribution and concentration. This highlights the importance of a robust clarification process that can accommodate some of this variability. Turbidity of the crude harvest is often used as a rough measurement to encompass the characteristics described above.
The feed turbidity will impact the capacity on the depth filters, and this should be accounted for when developing a process. To assess process robustness we used an adherent rAAV5 and ran seven replicate trials with feedstocks ranging from 29 to 129 NTU. In this work we consistently reached > 250 L/m2 throughput using a PDK11 depth filter + EKV membrane filter train.
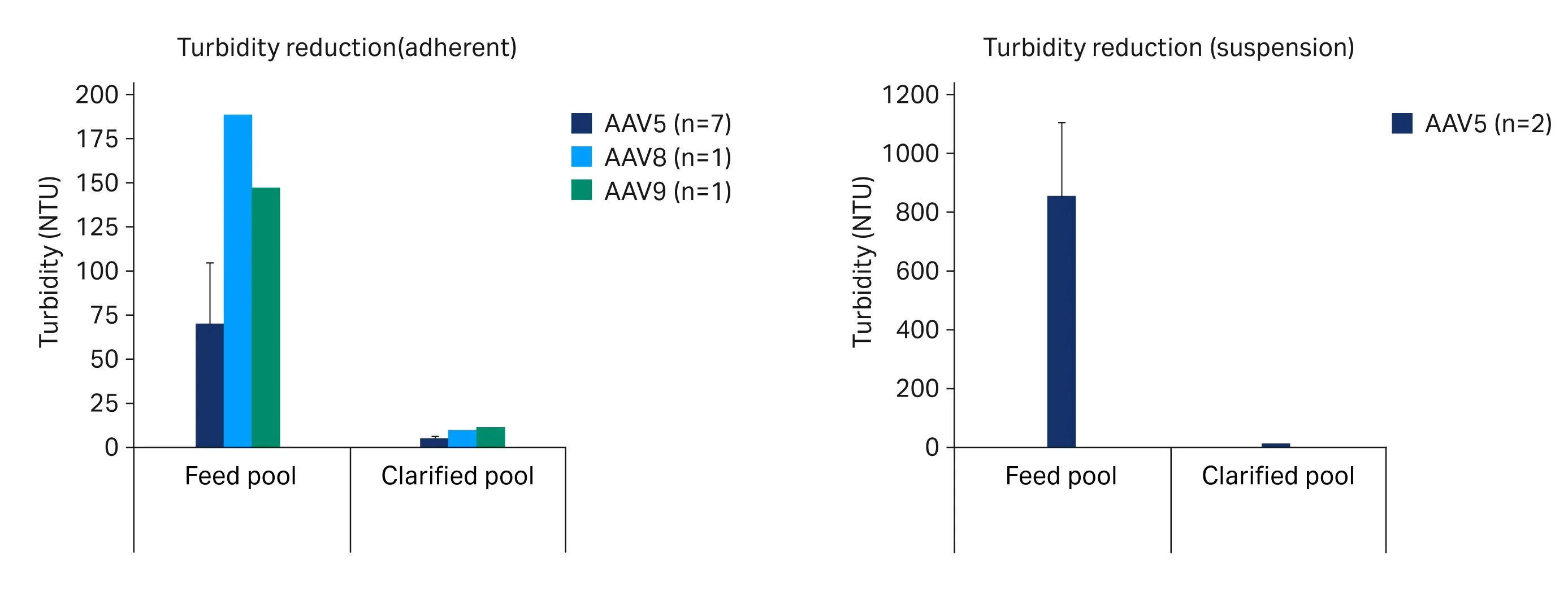
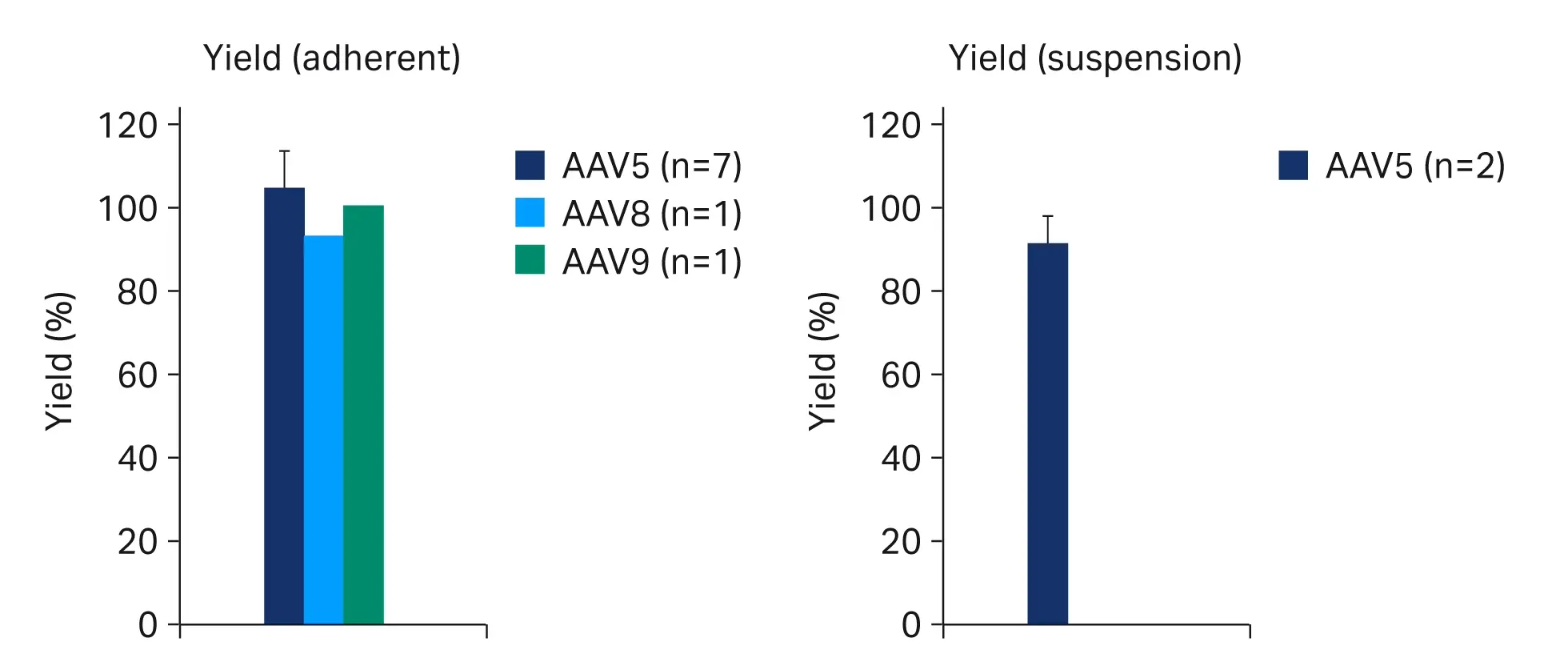
Across the range of feed turbidities we observed strong robustness for turbidity reduction (Fig 5A) and yield (Fig 6A) with pool turbidities at 3.0 ± 1.3 NTU and yields at 104% ± 9.6%. Similar process robustness was seen over two trials with suspension rAAV5 material. In these trials the feed turbidity ranged from 721 to 984 NTU. The pools clarified over a two-stage PDP8/V100P depth filter + EKV membrane filter train had turbidities at 4.0 ± 1.8 NTU (Fig 5B) and yields at 92% ± 6.1% (Fig 6B).
Finally, we evaluated three different rAAV serotypes produced in adherent culture. Here we saw no significant difference in pressure curves on the PDK11 or EKV when run with an rAAV8 or rAAV9 feed when compared to an rAAV5 feed with a similar turbidity (data not shown). We also saw strong robustness to serotype for turbidity reduction (Fig 5A) and yield (Fig 6A) with all clarified pools at or below 10 NTU and yields > 93%.
Fig 5. Turbidity reduction over clarification from adherent (A) and suspension (B) rAAV crude harvests. Where multiple trials were run error bars represent a 95% confidence interval.
Fig 6. Virus yields over clarification from adherent (A) and suspension (B) rAAV crude harvests. Where multiple trials were run error bars represent a 95% confidence interval.
Conclusion
The data here shows that either the V100P or PDK11 depth filters work well for clarifying AAV from adherent processes, providing yields > 95% with strong protection of the sterilizing-grade filter. Low-turbidity feedstreams may achieve adequate capacity from the single-layer V100P whereas high-turbidity feedstreams may require the dual-layer PDK11 to increase capacity..
For the suspension cultures tested, the two-stage depth filter approach of PDP8 followed by either V100P or BIO 10 performed well, providing yields > 90% and strong protection of the sterilizing-grade filter.
This study provides guidance for depth filter selection when developing a clarification process for recombinant AAV. Although the performance data is not directly transferrable to other AAV processes it provides an example approach to clarification process development. Figure 7 shown below gives an indication of the range of depth filter sheets available, taking into account your total cell density and type of cell culture platform.
Fig 7. Guide to the Seitz™ depth filter range.