Considerations for chromatography steps in viral clearance studies
Reduce risk of failure in your virus clearance study by choosing robust scale-down chromatography tools
Tina Pitarresi and Linnea Troeng
Cytiva Bio-Sciences AB, Björkgatan 30, 751 84 Uppsala, Sweden
Introduction
Effective viral clearance studies remain one of the challenges facing manufacturers of biologics. These studies are an essential part of process validation and are critical to ensure drug safety. Viral clearance studies are usually performed using a scale-down model mimicking the large-scale process step. For chromatography steps, using a scale-down model limits the amount of sample and resin used, lowering the cost for the study. As viral clearance studies are often cost and time consuming, it is essential to minimize risk of failure.
Seven considerations for performing viral clearance studies on chromatography steps
There is a lot to keep in mind before designing your study. Here are seven things to consider for your viral clearance study column:
|
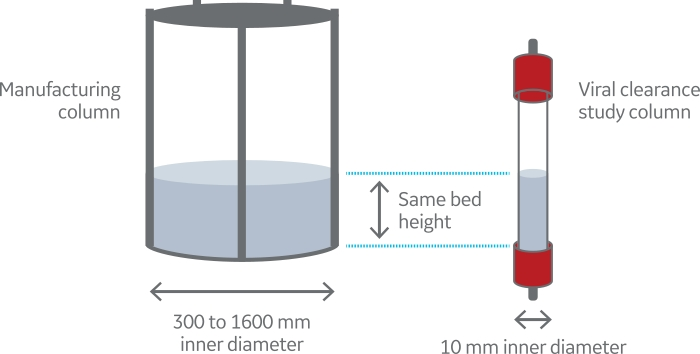
Fig 1. Schematic description of scale-down chromatography mode |
High-quality column packing reduces risk of failure for robust viral clearance studies
Several chromatography columns are required for each viral clearance study. So, it is essential to carefully consider which columns to use.
Remember that the column should:
- be easy to pack or be prepacked
- provide reproducible results between runs
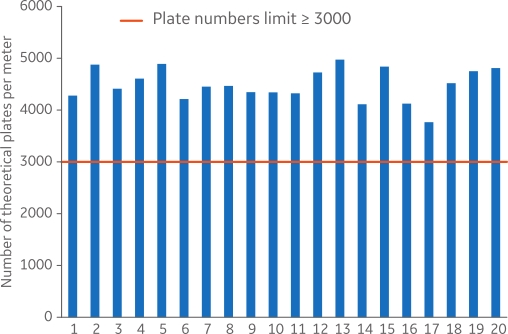
Figure 2 shows efficiency analysis for 20 different MabSelect SuRe Validation columns featuring high-quality packing with theoretical plate numbers per meter above the limit (3000) and consistency between the packed columns.
Fig 2. Efficiency analysis of 20 MabSelect SuRe Validation column
Case study: a successful viral clearance study for a protein A chromatography step
|
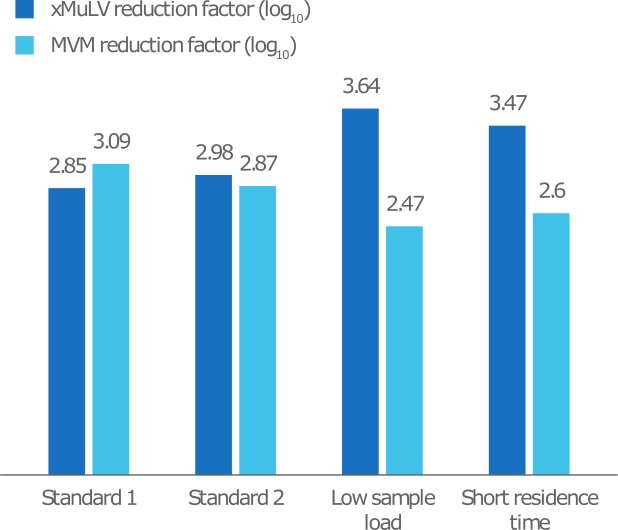
In this study, the virus reduction of an affinity capture step was evaluated using HiScale 10/40 columns packed with MabSelect PrismA resin. The study was performed by a third-party viral testing laboratory in collaboration with a biopharmaceutical company. The model viruses used were Xenotropic murine leukemia virus (xMuLV) and Minute virus of mice (MVM). Design of experiments to challenge viral clearance robustness Results and discussion |
Fig 3. Summary of reduction factor results in relation to sample load and |
Table 1. Details of the different scenarios studied
| Scenario | Sample load concentration (mg/mL) | RT in chromatography process (min) | RT in loading step (min) |
|---|---|---|---|
| Standard 1 | 65 | 6 | 12 |
| Standard 2 | 65 | 6 | 12 |
| Low sample load | 20 | 6 | 12 |
| Short residence time | 65 | 4 | 4 |
View the full case study: virus reduction of an affinity capture step.
When to use prepacked columns or pack your own?
| Add flexibility with empty HiScale 10/40 columns |
Save time with prepacked Validation columns |
|
|
|
Summary
- Scale-down columns are commonly used for resin viral clearance studies.
- Both the empty HiScale 10/40 columns and the prepacked Validation columns are well-suited for scale-down studies.