Given the vast range of paths to higher productivity, and the number of products and vendors available, how can you decide on the best solutions for your circumstances? And how can suppliers help?
Change is one of the few constants in biopharma manufacturing. New therapies and technologies bring exciting opportunities for patients, but they also place pressure on companies to keep up, cut costs, and stay flexible. Demand can change quickly, patents expire, and global events can shift production needs. At the same time, manufacturers must keep improving productivity through smart process design and targeted investment.
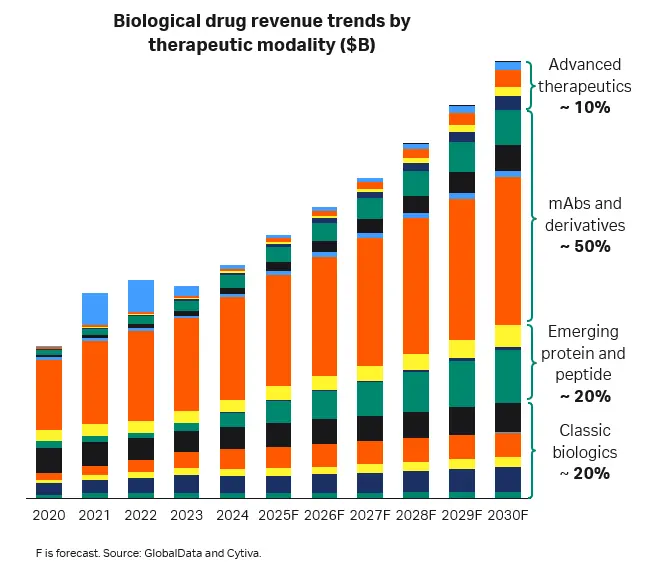
Monoclonal antibodies (mAbs) remain the dominant modality, but the space is evolving. More complex formats—such as fragments, multispecifics, and antibody-drug conjugates—are becoming more common. As mAb patents expire, biosimilar competition grows and companies look for ways to increase capacity, improve efficiency, and spread manufacturing across regions.
Other fast-growing modalities such as mRNA, oligonucleotides, plasmids, and viral vectors are reshaping the landscape. These newer products introduce process challenges and regulatory questions that disrupt timelines, and they sometimes require new technology and process changes. To stay competitive, manufacturers need flexible production lines that can handle a range of products and output levels.
Fig 1. Revenue projections for various therapeutic modalities through 2030.
Finally, sustainability has become a critical issue across nearly every industry – including biologics manufacturing. Many companies want strong sustainability commitments before they agree to work with vendors.
All of this adds up to a field of promising opportunities. To realize them, you need to consider where and how to invest your attention, time, and resources.
Approaches to increase output
Manufacturers often look for higher productivity from a bioprocess — whether they’re responding to rising demand, the need to have a more efficient facility, or their goals to be more sustainable. There are three main approaches:
- Scale-up: increase batch volume – by upsizing, which might lead to expanding or building a facility
- Process intensification: increase output without larger inputs
- Scale-out: run more production lines
Scale-up vs scale-out or intensification
Big can be efficient and necessary – but not always. In manufacturing, scaling up can cut costs, especially for large-volume needs. That can mean larger systems and even new facilities to keep up with demand. Today, many companies are facing the need for fast, flexible capacity amid geopolitical tensions. As a result, many are choosing smaller processes and more distributed manufacturing.
Scale-up is a standard path during clinical development: each phase requires more product. Some companies believe they need a new facility only to learn that intensifying their current process would meet their needs. So, the question isn’t so much “Will I need to scale up” as it is “When do I stop scaling up and start intensifying or scaling out?”
Planning ahead
Whether you’re scaling up for the first time or looking to squeeze more value out of an existing process, the core principles for process optimization are the same: start early and keep the end in mind.
Starting early is essential, because process changes become harder over time. Late-stage changes can affect critical quality attributes and may even require repeating costly clinical trials. Some companies choose to launch quickly with a nonoptimized process and improve it later. To avoid this tradeoff, consider whether early process decisions will remain feasible and scalable over the long term.
Designing with the constraints of production environments in mind during development helps you control costs and complexity. For example, reducing the number of buffers can simplify logistics and reduce costs. Manufacturers should take a holistic view of their processes and ensure that process development, pilot plant, manufacturing, QC, and QA teams work together as one unit to build an efficient, optimized process.
When planning large-scale operations, you’ll need to choose between stainless steel facilities and single-use systems. Stainless steel is commonly used at very high volumes, but single-use can also scale – for example, by running multiple 2000 L bioreactors in parallel for mAb production. Whichever path you choose, vendor reliability, supply chain strength, and support across your workflow are critical to successful scale-up.
Why consider intensification?
Process intensification describes any process change that improves productivity or reduces the cost of goods sold (COGS) – usually by producing higher yields from similar or smaller footprints.
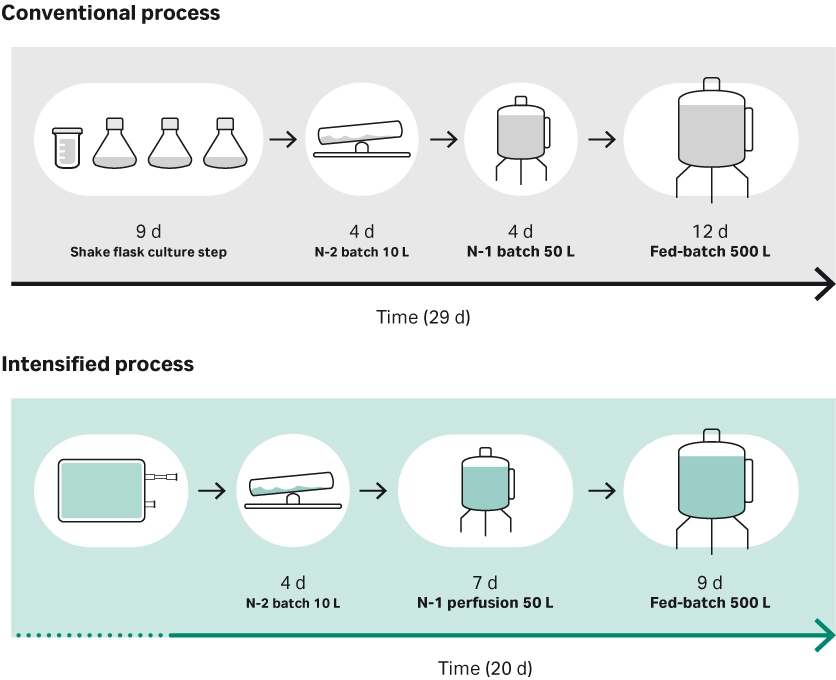
Upstream, using perfusion to grow cells to higher density is a common strategy. There are several ways to use perfusion at the N-2 or N-1 (seed train) stage to shorten time in the production (N) bioreactor or increase output; one example, which also includes high-density cryobags generated using perfusion, is shown in Figure 2 (1). In the production bioreactor, dynamic perfusion can lower the media volume by 40% compared to steady-state perfusion, while keeping productivity high (2).
Fig 2. Overview of conventional versus an intensified high-density seed process evaluated in a study showing that high-density cryobags can be used to seed a bioreactor at a 500-fold higher seed density, reducing the total process time by up to 9 days (2).
Downstream intensification options include:
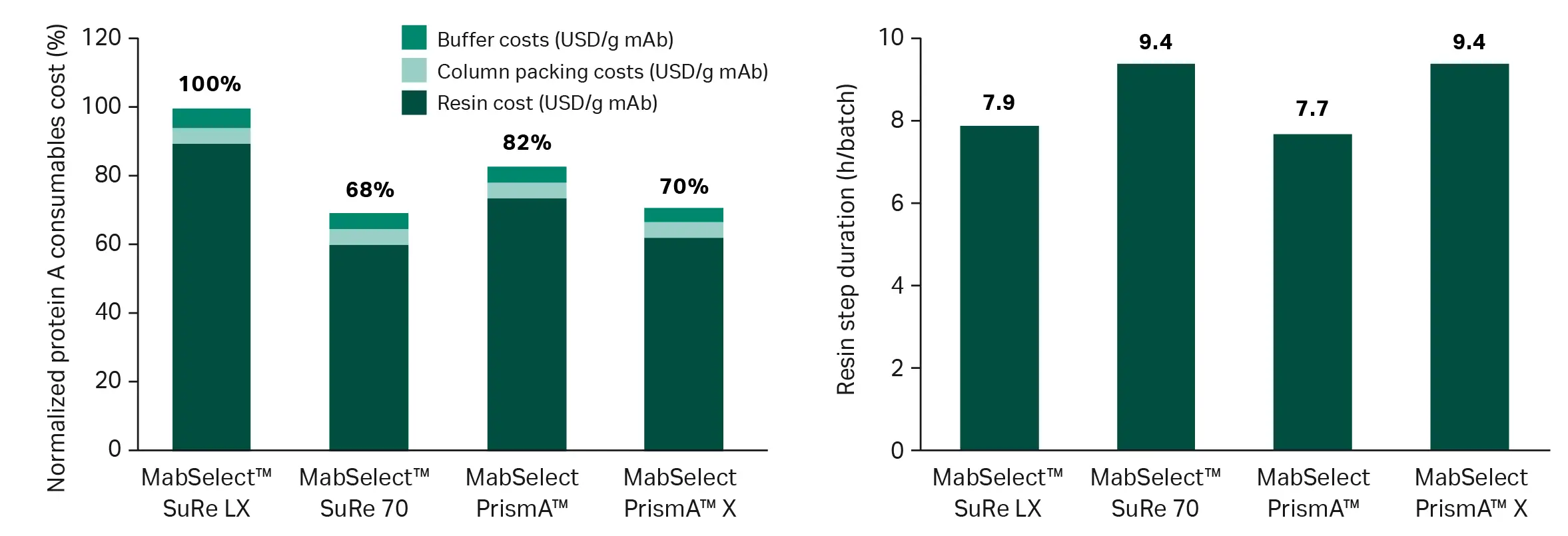
- using higher capacity resins (Fig 3) (3) or filters to reduce consumable and buffer use per unit of product.
- using prepacked chromatography columns to avoid the need for packing and cleaning.
- running a continuous chromatography process. Multicolumn chromatography using periodic countercurrent chromatography (PCC) is one way to purify target proteins in continuous downstream processes.
- connecting chromatography unit operations with filtration to reduce process time – for example, by connecting polishing and concentration under one automation method (4).
Fig 3. Consumable cost per gram of mAb produced and step duration comparisons for a clinical process scenario. Refer to reference 3 for details.
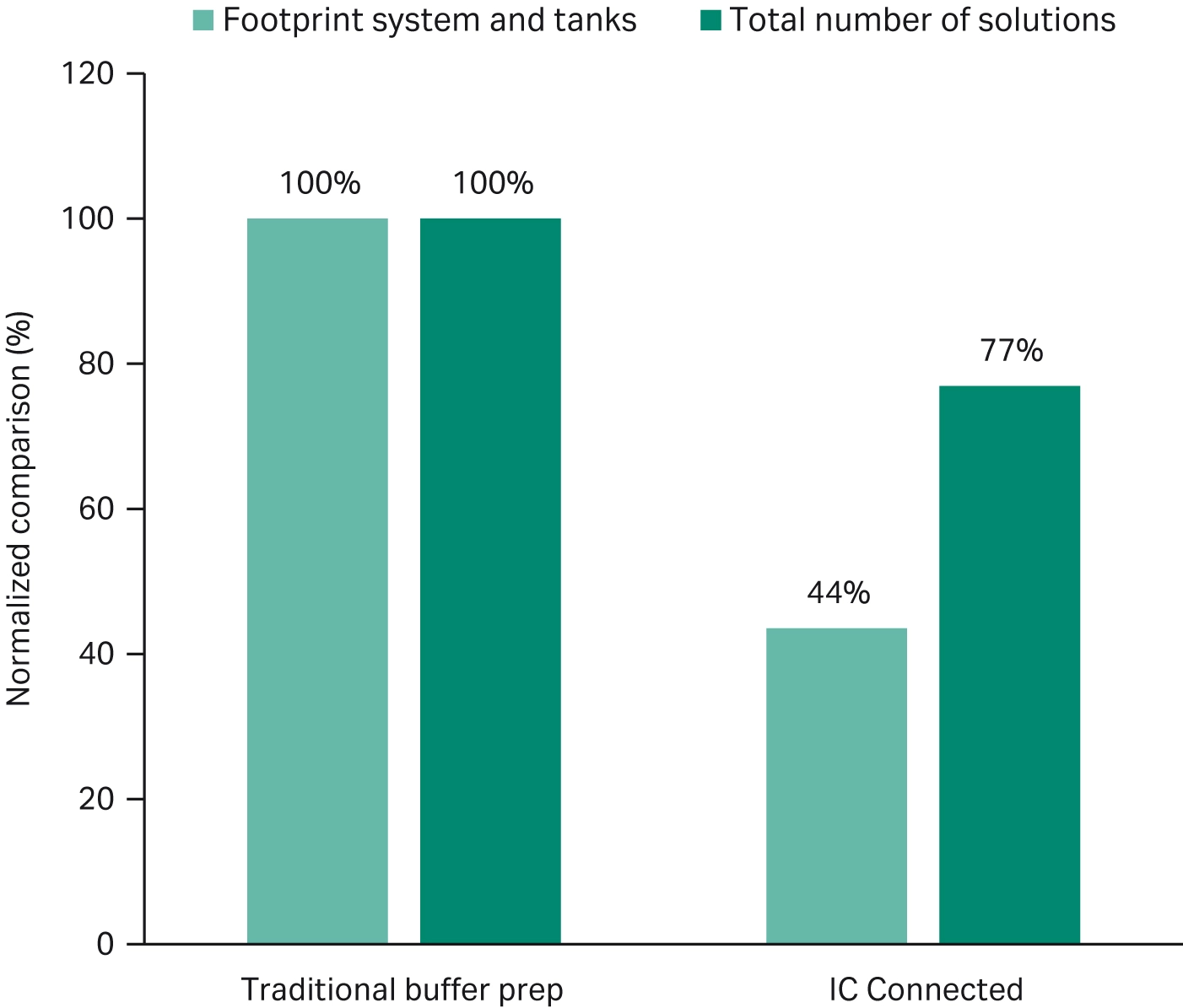
Across unit operations, large-scale buffer management strategies can save time, space and cost. Options include inline dilution, inline conditioning (IC), and ready-made buffers. To increase the level of intensification, inline conditioning systems can be used to deliver buffers directly to different connected downstream systems (Fig 4) (5).
Fig 4. Normalized comparison of footprint and number of solutions needed for a batch size of 10 kg mAb from a 2000 L bioreactor (5).
Switching from batch to continuous manufacturing can boost productivity, and advances in technology are making it easier to adopt. You can also improve efficiency by intensifying individual steps without making the whole process continuous.
Switching to continuous processing early in development reduces risk and regulatory hurdles because you don’t have to prove equivalence between old and new processes. If the switch happens later, good process analytics are essential to ensure that product quality isn’t compromised by process modifications. Better sensors and digital tools are helping, though there’s still room for improvement.
Standardizing processes helps cut costs, reduce process risks, and speed up development. Custom bioreactors and specialized downstream tools are common, but newer standardized solutions are emerging that are simpler, cheaper, faster, more efficient, and easier to control for quality.
Outsourcing as another option
Contract development and manufacturing organizations (CDMOs) can help increase capacity quickly and offer specialized expertise. However, outsourcing might reduce operational control, limit future flexibility, or require sharing sensitive intellectual property. The right CDMO can help you avoid these common challenges.
Any change—intensifying steps, outsourcing, or building new facilities—adds cost through validation, tech transfer, and regulatory checks. Intensification may even increase QC workload. This is why it’s essential to have an integrated ecosystem with strong digital tools and aligned teams.
Technology is reshaping timelines
Standardized equipment, automation, and digital tools are shortening process development timelines. Companies can now move to phase I in as little as six months. This creates pressure on manufacturers and CDMOs to stay competitive while also training and retaining skilled staff.
Overall, the industry is shifting quickly, and companies must adapt. The choices are wide—cell lines, media, bioreactors, operating modes, capture resin choice, filters, and other downstream setups—and access to some advanced technology can be limited. But with careful planning and the right collaborators, manufacturers can meet demand more efficiently.
References
- Intensifying your fed-batch process using cryobags. Cytiva. https://www.cytivalifesciences.com/insights/intensifying-your-fed-batch-process-using-cryobags. Updated July 28, 2025. Accessed January 28, 2026.
- Upstream process intensification with dynamic perfusion. Cytiva. https://www.cytivalifesciences.com/insights/upstream-process-intensification-with-dynamic-perfusion Updated November 19, 2025. Accessed January 28, 2026.
- Balancing protein A resin cost, performance, and productivity. Cytiva. https://www.cytivalifesciences.com/insights/protein-a-resin-cost. Updated April 27, 2025. Accessed January 28, 2026.
- Connected polishing and concentration under one automation method. Cytiva. https://www.cytivalifesciences.com/insights/connected-polishing-and-concentration-under-one-method. Updated October 17, 2025. Accessed January 28, 2026.
- Connected inline conditioning for better process efficiency. Cytiva. https://www.cytivalifesciences.com/insights/connected-inline-conditioning-solution-for-bioprocessing Updated February 16, 2025. Accessed January 28, 2026.
CY57743-16Feb26-AR