Compare the benefits and limitations of using X-ray film and digital CCD imaging for Western blot analysis. Which detection method provides better sensitivity, linear dynamic range, and versatility for immunoblotting?
What is Western blotting?
Western blotting (also known as immunoblotting and protein blotting) is an established and widely published form of protein detection and analysis. Our 7-step guide to Western blotting covers the workflow used in most life science applications.
A typical Western blotting protocol involves separating denatured proteins by polyacrylamide gel electrophoresis (PAGE), immobilizing them on a membrane, and then targeting them for detection with specific antibodies against your protein of interest.
Chemiluminescence detection is the most common approach for visualizing protein bands on a Western blot and requires exposing the membrane to X-ray film or a digital charge-coupled device (CCD) imager. Both blot visualization methods are commonly used. In addition to chemiluminescence detection, many CCD imagers can now also be used for fluorescence detection.
How Western blotting detection works
The chemiluminescence approach to detection commonly used by labs is based on an enzymatic reaction between hydrogen peroxide and luminol. The reaction is catalyzed by antibody-conjugated horseradish peroxidase (HRP) and results in light emission.
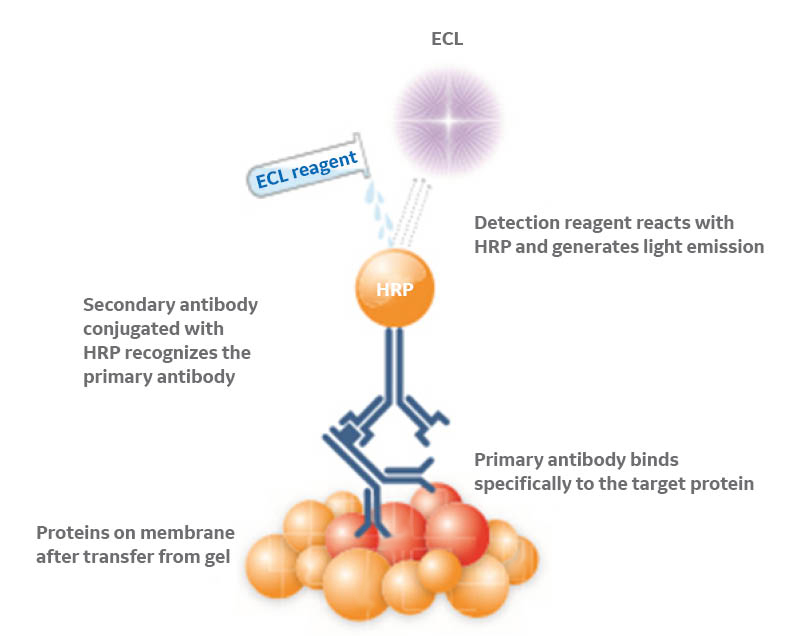
Traditional chemiluminescence detection emits light at a relatively low intensity and for a short period of time. Enhanced chemiluminescence (ECL) products were developed to amplify the signal to levels that enable detection of picograms of protein by film or CCD imaging (Fig 1).
Fig 1. Representation of ECL-based protein detection in Western blots. An HRP-conjugated secondary antibody recognizes the primary antibody directed against the protein of interest and catalyzes the reaction between the ECL substrates, generating light emission.
Using X-ray film for chemiluminescence detection
For many years, X-ray films were the preferred and default choice for chemiluminescence detection. Exposing blots to film typically involves placing a film directly on the membrane inside a darkroom and leaving the film and membrane sealed inside the cassette for the intended exposure time.
The proximity to the blot helps the film provide high sensitivity to detect weak protein bands. The film and blot can be left for several hours or overnight in the cassette, if necessary, for convenience and without occupying equipment.
Film does, however, present a challenge when you have both high- and low- intensity protein bands on your blot. The sensitivity of film means it is also easily saturated and has a limited dynamic range. This limitation can prevent you from visualizing both strong and weak bands in the same exposure.
To address this shortcoming, you might find yourself performing multiple exposures in an attempt to get all the bands of interest within the film’s limited dynamic range. This method could work fine if you are just looking for the presence or absence of a band, but it doesn’t enable you to directly compare or easily quantitate.
Quantitation with film also requires scanning with a densitometer or taking a digital photo for analysis, meaning additional steps if using film exposure for publication.
Using digital CCD imaging for chemiluminescence detection
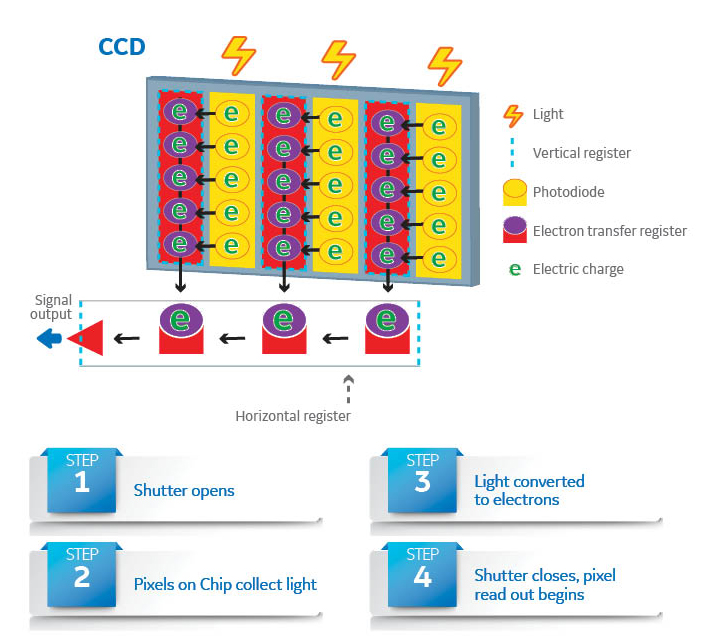
Much like the trend observed in traditional photography over recent years, digital CCD imagers are replacing film. These imaging systems use a light-sensitive CCD chip that converts photons into digital signals (Fig 2) and immediately displays the result of the exposure on a screen.
Fig 2. Components of a CCD imaging chip, which forms a digital image by converting photons hitting sensor pixels to an electrical signal.
Specially developed CCD imagers for laboratory use can be just as sensitive as film for Western blot analysis. With no need for a darkroom or development steps, CCD imagers make it simple to check, repeat, and adjust exposures.
CCD imagers also tend to have a high linear dynamic range, a key advantage over film. It is often possible to detect both high- and low-intensity bands in the same exposure without risking saturation, either through trial-and-error or with an automatic exposure mode provided by the system software.
Digital exposures also make comparing and quantitating Western blots more straightforward. Vendors often provide tools in their software to support quantitation.
A challenge that you might face using a CCD imager, however, involves the CCD chip’s sensitivity to light, heat, and high-energy radiation. These factors can affect performance and image quality. Cooled CCD chips help combat this, aiming to improve both sensitivity and linearity.
Sophisticated imaging algorithms in CCD imaging systems can also help minimize random noise and maximize the signal-to-noise ratio, making it possible to visualize and quantitate even the faintest protein bands.
Cost and convenience of film and CCD imaging for lab users
In terms of cost, convenience, and environmental impact, film presents more immediate and visible challenges than CCD imagers.
Using many films over time can be more expensive than a single CCD imaging system. In addition to the cost of reagents and films, you should also consider the demands and risks of maintaining a darkroom. The film processing chemicals contain components that are potentially hazardous to both human health and the environment, so disposal requires special care.
Although the initial cost can seem high by comparison, CCD imagers are versatile systems. CCD systems are capable of imaging a variety of samples with different modes of illumination, including infrared wavelengths. This versatility makes CCD imagers useful across a range of laboratory imaging applications, including fluorescent Western blots.
Fluorescent Western blot detection by CCD imaging
Using fluorophores on a secondary antibody is less common than the chemiluminescence approach, but fluorescence detection does provide similar sensitivity to chemiluminescence, signal stability, and broad linear dynamic range.
Whereas laser scanners, with a considerable upfront cost, used to be the only option for reliable imaging of fluorescent Western blots, modern CCD imagers provide similar capabilities without the same expense.
The final exposure on film versus digital CCD imagers
Although there are several differences between X-ray film and digital CCD imaging, both imaging options have a place in the lab. Table 1 summarizes some of their key differences.
Table 1. Film versus digital CCD imaging, in summary
| X-ray film-based detection | CCD imager-based detection | |
|---|---|---|
| Linear dynamic range | Limited: High sensitivity due to proximity of film to blot, but saturates protein bands of medium to high intensity easily | High (up to 4.8 orders of magnitude): Can detect proteins with both high and low intensity without risking signal saturation |
| Ease of exposure |
|
|
| Suitability for quantitation | Not suitable for quantitation | Accurate and reliable quantitation |
| Ease of use | Film requires development in darkroom before digitizing |
|
| Environmental concerns | Film development reagents are harmful to the environment and require careful disposal | Eco-friendly, no need for additional reagents |
| Cost | Lower up-front cost, high consumable and maintenance costs | Higher up-front cost, no consumables and low maintenance costs |
We aim to streamline and improve imaging quality in Western blotting workflows. Read more about Western blotting detection in our detailed imaging principles and methods handbook. For support with any aspect of your Western blotting or imaging workflows, contact our Scientific Support team.