Adeno-associated virus (AAV) is an increasingly common vector for introducing nucleic acid into living cells, and scientists mostly apply it for gene therapy.
Standard titer analysis uses ELISA, an assay that requires a lot of manual interaction and can come with challenges in precision and reproducibility.
In this application note we describe the performance of two assays for titer analysis of adeno-associated virus (AAV), serotype 2 and serotype 5, based on surface plasmon resonance (SPR). Both assays provide repeatable results and correlate well with established total AAV capsid titer assays using ELISA. Compared to ELISA, Biacore™ assays have a higher degree of automation and precision, and they’re compatible with process samples from harvest to final purified bulk. Their performance and ease-of-use make them very suitable as process development tools.
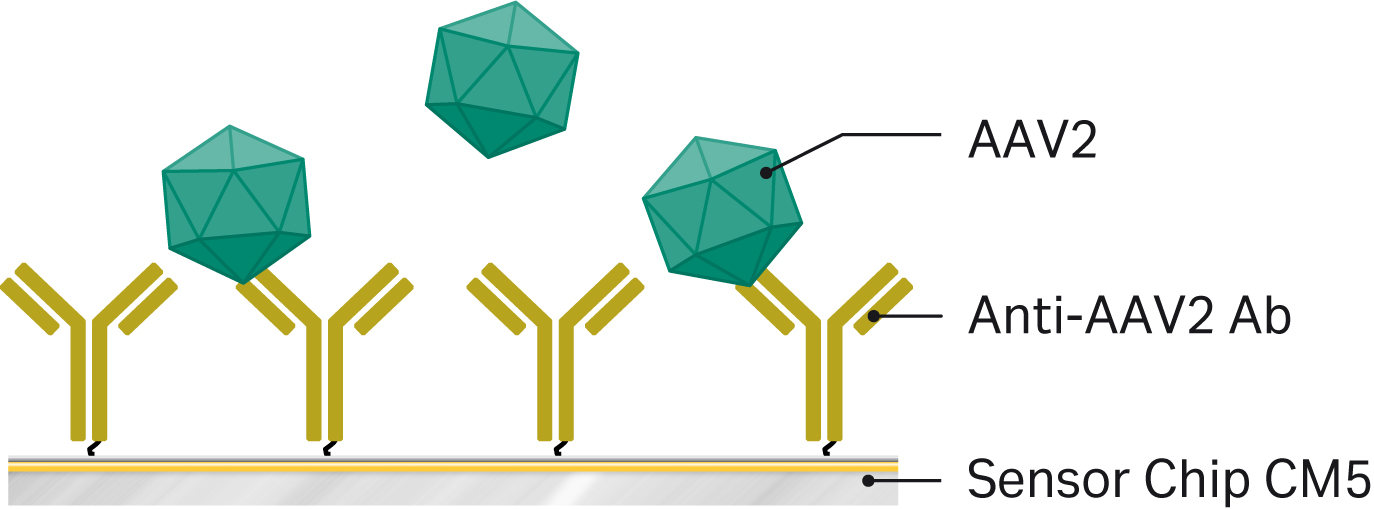
We assessed titer of AAV serotype 2 (AAV2) by allowing virus particles to bind to an AAV2-specific antibody covalently coupled to a Biacore™ sensor chip.
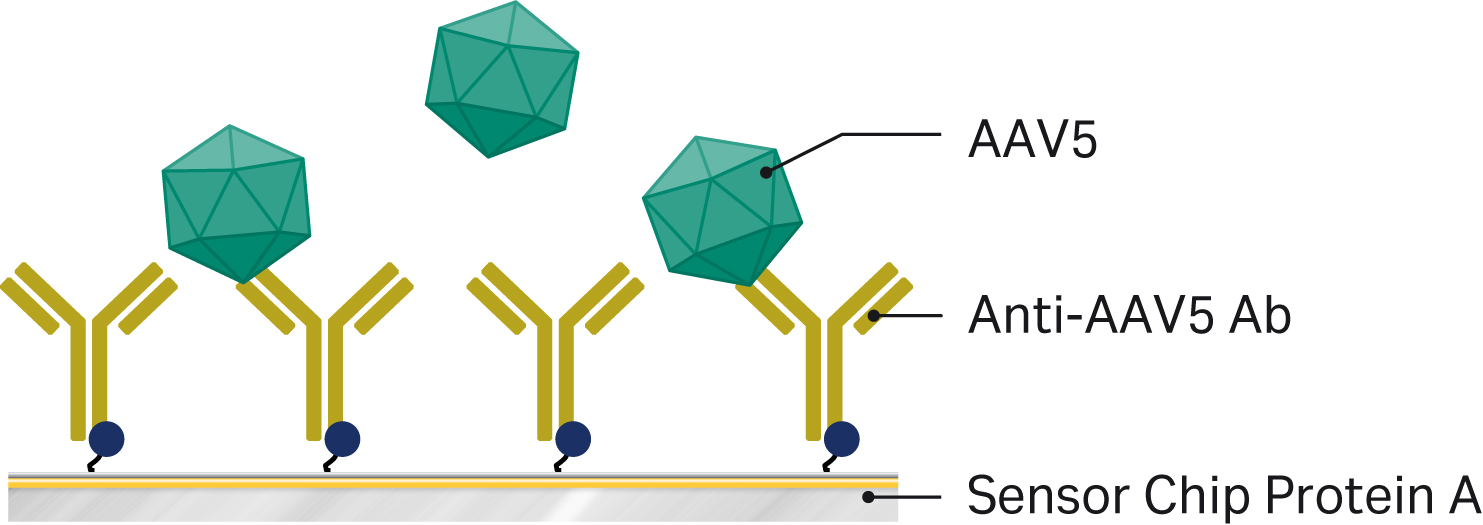
We assessed titer of AAV serotype 5 (AAV5) by allowing virus particles to bind to an AAV5-specific antibody captured on a pre-functionalized Biacore™ sensor chip. We developed both assays using a Biacore™ T200 SPR system, but you can apply them on any Biacore™ SPR system that supports concentration analysis.
Introduction
Virus particles can introduce foreign nucleic acid into living cells, making them a suitable vector for delivering genetic material. Viral vectors show high potential for use in vaccine and therapeutic applications like gene therapy, cell therapy, oncolytic cancer immunotherapies, and tumor vaccines. Common viral vector systems include adenovirus, adeno-associated virus, and lentivirus.
Adeno-associated virus (AAV) is an increasingly common vector in gene therapy. Scientists identified AAV as a separate virus type in the 1960s — it was previously thought to be a contaminant in adenovirus preparations. This virus type is not known to cause disease, only mild infections. AAV is a small virus which limits its capacity as a vector to smaller nucleotide sequences (< 4.4 Kb). There is, however, ongoing research that aims to increase the packing capacity of AAV [ref. 1].
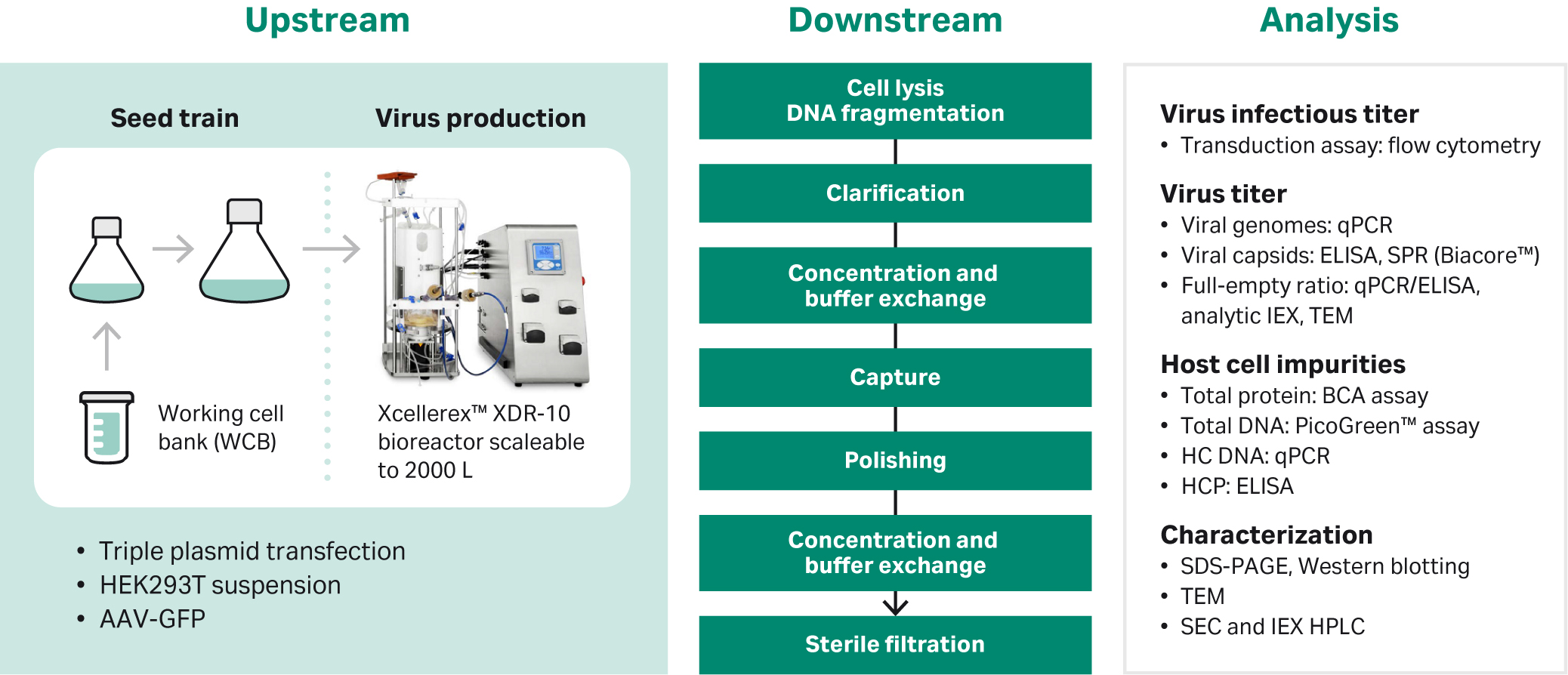
This application note covers the analysis portion of a full adeno-associated virus production process that we developed from upstream production to a final purified product for AAV serotype 5. Refer to Figure 1 below for a description of the production and purification scheme. For more detailed information, see references 2, 3, 4, and 5.
Fig 1. Production and purification scheme of AAV5.
During process development, final production, and purification, we monitor a range of critical quality attributes including infectious titer, viral genome titer, viral total particle, host cell protein, and DNA levels. Many of the techniques used for this are labor-intensive with low precision and a low degree of automation. In addition, the analyses are often time-consuming and costly.
Simplified, high-precision analytics like SPR can enhance today’s workflows with a high degree of automation. Here we use SPR for total titer analysis of AAV2 and AAV5. Both assays are based on binding of virus particles to an antibody specific to intact virus particles.
Materials and methods
Titer analysis of AAV2
We developed the assay for total AAV2 virus particle titer using Biacore™ T200. However, you can apply the assay to any Biacore™ system with software that supports concentration analysis. The assay principle is shown in Figure 2.
Fig 2. Assay principle AAV2 titer analysis using Biacore™ systems. An anti-AAV2 antibody is amine coupled in one flow cell on Sensor Chip CM5 in Biacore™ T200. AAV2 samples diluted 5-1000 times in running buffer supplemented with extra NaCl are injected over the surface and virus titer is evaluated against a calibration curve.
We immobilized anti-AAV2 antibody from Progen (Cat. No. 610298) to Sensor Chip CM5 using standard amine coupling and HBS-EP+ as running buffer. Then we diluted antibody to 20 µg/mL in 10 mM acetate pH 5.0 and injected it over 7 min, resulting in immobilization levels between 6000 and 10000 RU. We performed immobilization at 25˚C in a single flow cell.
For the titer analysis, we used HBS-EP+ buffer supplemented with extra NaCl to a final salt concentration of 0.3 M as running and sample dilution buffer. The extra NaCl eliminates nonspecific binding from crude samples. Samples are generally diluted between 5 and 1000 times depending on expected titer, and we included two different dilutions of each process sample in each experiment. We injected the samples using a contact time of 400 s at 10 µL/min, and performed regeneration using a 60 s injection of 3 M MgCl2. Then we performed analysis at 25°C with sample compartment temperature set to 10°C to preserve sample integrity. Titer analysis was performed without a reference surface as recommended for Biacore™ concentration analysis.
We constructed the calibration curve using an AAV2 ATCC standard (ATCC AAV2 reference standard VR-1616) and range from 3.6 × 108 to 9.2 × 1010 VP/mL in a 2-fold dilution series. A first calibration curve is included before the samples and a second calibration curve is included after the samples. The assay includes 5-10 start-up cycles with buffer and regeneration.
The anti-AAV2 antibody used in the titer assay is specific to intact virus particles only. That means that you can use the assay to monitor AAV2 titer in crude cell culture samples without interference from free virus and host cell proteins. According to the vendor’s specifications, this antibody is specific to AAV3, meaning that you can also apply the assay to this serotype after optimizing standards, regeneration, contact times, calibration range, and other parameters.
Titer analysis of AAV5
We also used Biacore™ T200 for analysis of AAV5 virus titer. This assay is run using capture to Sensor Chip Protein A and doesn’t require separate immobilization. We used a capture format instead of covalent coupling because of the binding antibody for this assay. Our early tests showed that the antibody was sensitive to acidic pH, which ruled out amine coupling because this procedure requires dilution in acidic buffer. Capture occurs at neutral pH, which is much gentler for the antibody. The assay principle is shown in Figure 3.
Fig 3. Assay principle AAV5 titer analysis using Biacore™ systems. An anti-AAV5 antibody is captured in one flow cell on Sensor Chip Protein A in Biacore™ T200. AAV5 samples diluted 5-1000 times in running buffer supplemented with extra salt are then injected over the surface and virus titer is evaluated against a calibration curve.
We diluted anti-AAV5 antibody from Progen (Cat. No. 610148) to 16 µg/mL in running buffer and captured it on the sensor chip in a single flow cell before injecting AAV5 virus samples. We performed capture using a contact time of 240 s and flow rate 5 µL/min.
We injected samples using a contact time of 120 s and flow rate 10 µL/min. Regeneration with 30 s 10 mM glycine-HCl pH 1.5 removes captured antibody and virus from the sensor surface. New antibody is captured in every cycle. To preserve samples, we set the sample compartment temperature to 10°C and the analysis temperature to 25°C. We used HBS-EP+ supplemented with extra NaCl to a final salt concentration of 500 mM as a running and sample dilution buffer. Samples are diluted between 10 and 1000 times depending on expected titer, and two different dilutions of each sample are typically run. We constructed the calibration curve in the range of 1.7 × 1010 to 5.8 × 1011 VP/mL using Vigene Bio pAV-CMV-GFP (Full) AAV5 (art. No. CV10005) as standard. We ran the assay with 5-10 start-up cycles with buffer and regeneration, running a first calibration curve before the samples and a second calibration curve after the samples.
The anti-AAV5 antibody is specific to intact virus particles, making this assay suitable for analyzing cell culture samples because there is no interference from free virus and host cell protein.
AAV upstream and downstream samples
We took samples from different steps in upstream and downstream workflows with large variation in samples matrices and expected titers. See Figure 1 for more details.
Results and discussion
AAV2 titer analysis
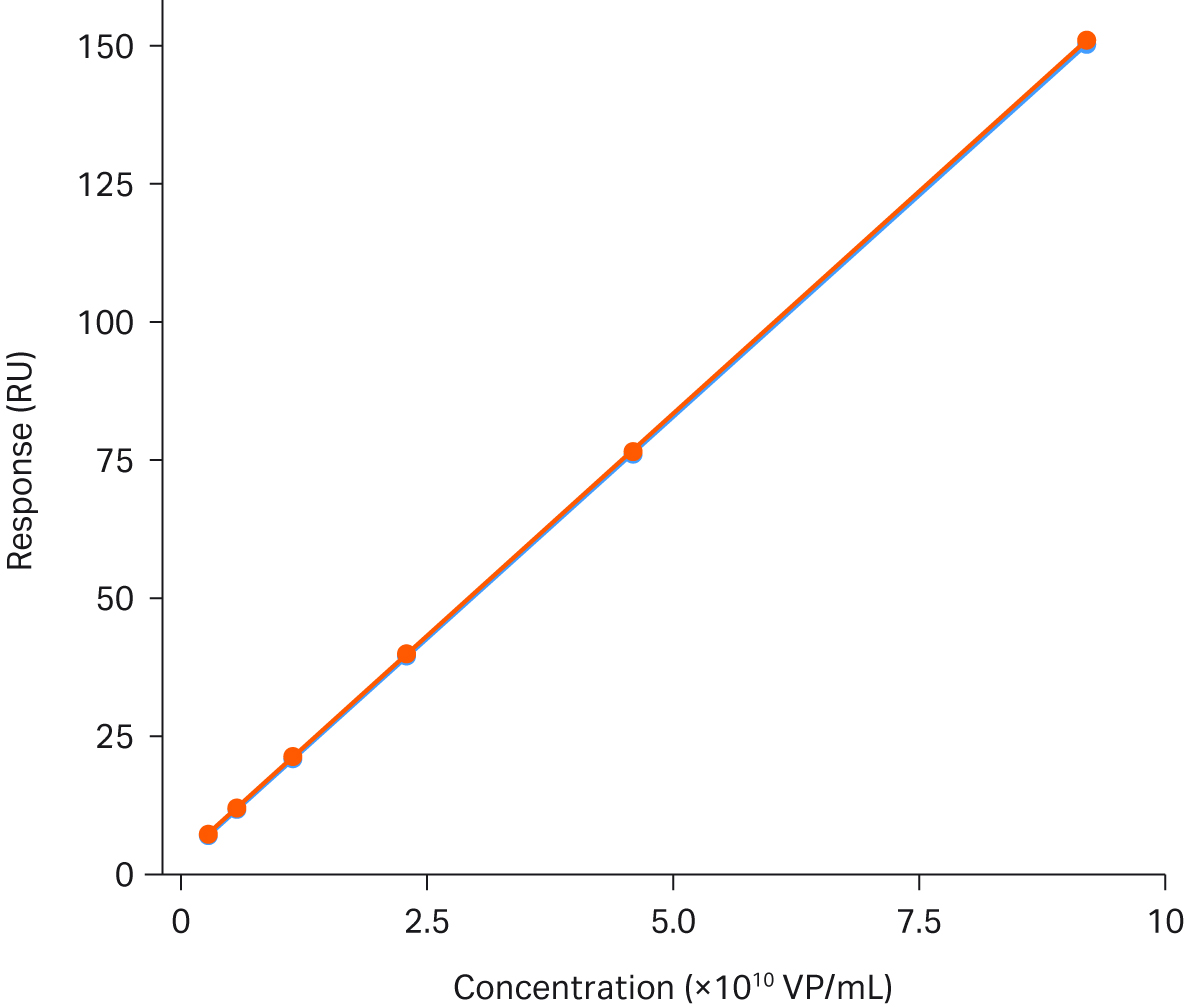
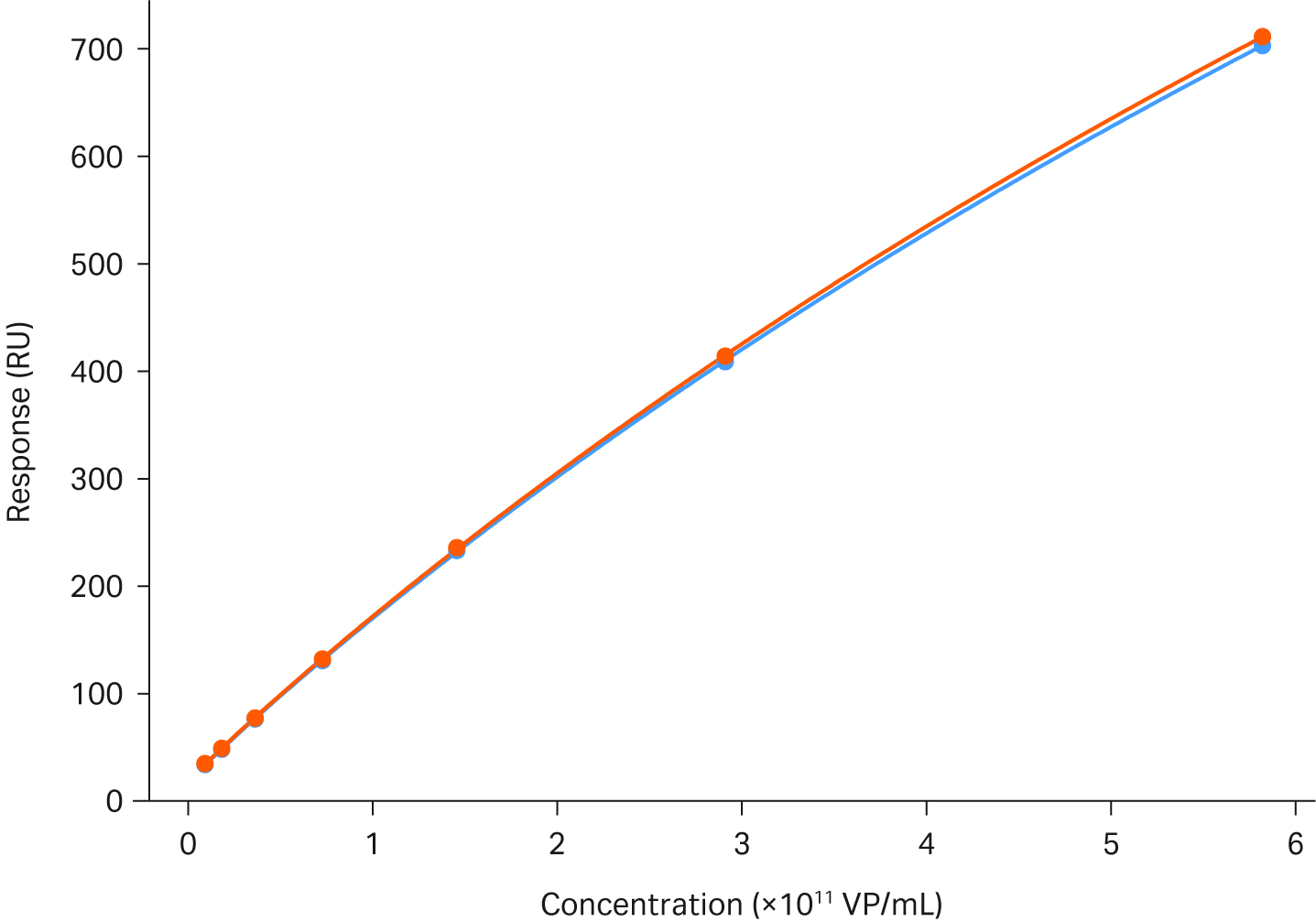
Assay stabilityFigure 4 describes the stability of the AAV2 titer assay. The figure shows a perfect overlay of two calibration curves with 66 samples run between them.
Fig 4. Titer analysis of AAV2 using Biacore™ T200. The figure shows an overlay of two calibration curves run with 66 samples between them. The range of the calibration curve is 3.6 × 108 to 9.2 × 1010 VP/mL.
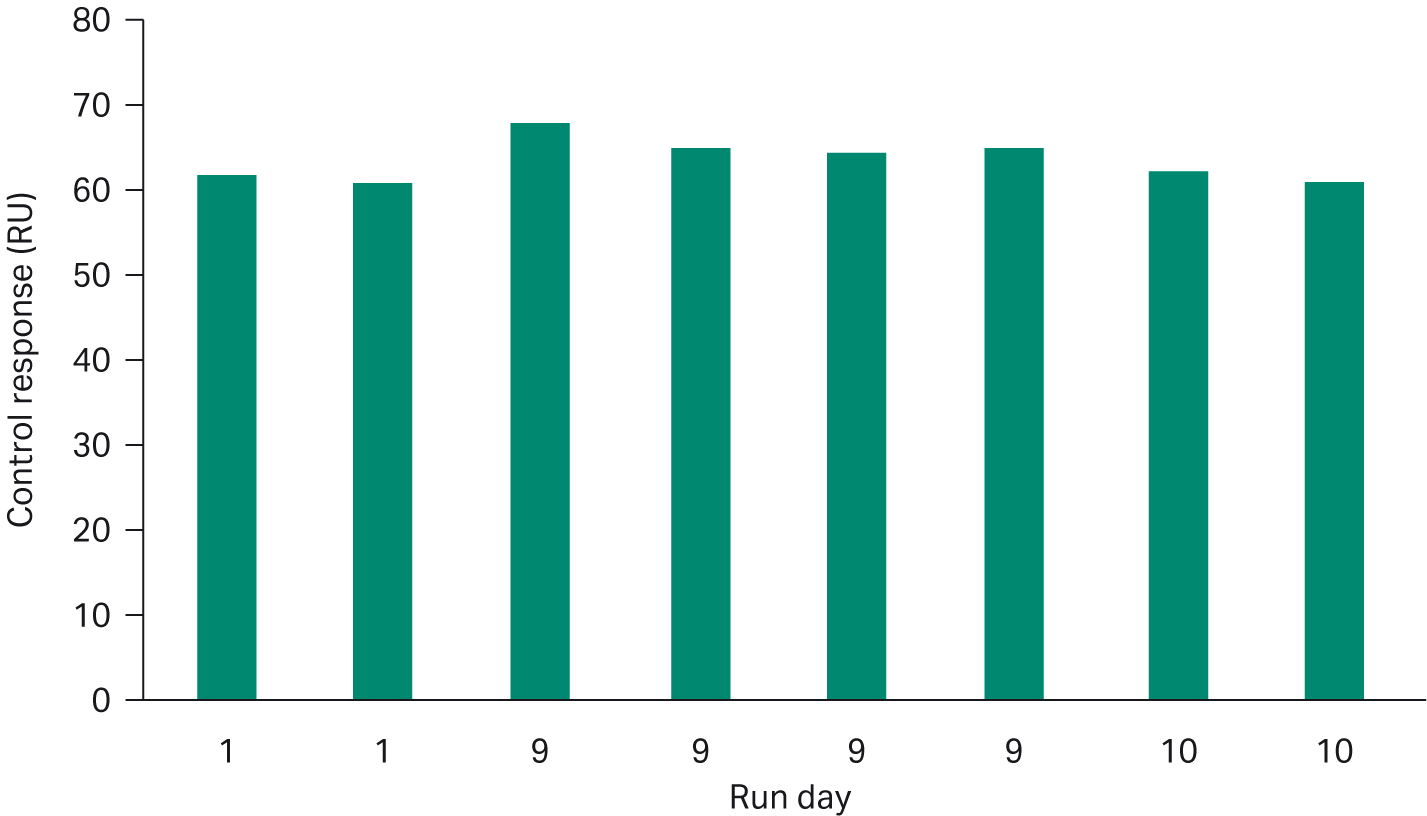
When developing the assay, we found that the immobilized chip is stable enough to use in multiple runs for at least 10 consecutive days under recommended run conditions. The high stability also supports a master calibration curve — a calibration curve run on day 1 which is then used to evaluate samples in subsequent runs. We monitored the performance of the immobilized chip by including an in-house positive control sample in each run. Figure 5 shows the response levels for the positive control from three separate runs performed over ten days on the same immobilized sensor surface. The relative standard deviation of the control response level is less than 4%. A master calibration curve helps save expensive standard and reduce time to results.
Fig 5. Response of positive control from three separate analysis occasions using the same immobilized sensor chip over ten days. Relative standard deviation for replicate measurements of the control is less than 4%.
Stability of standard
The AAV2 ATCC standard is delivered on dry ice in 500 µL frozen aliquots and stored at -80˚C upon reception. At first use, we thaw the ATCC vial and dispense the volume in 15 µL aliquots in Protein LoBind Eppendorf vials. We then store the aliquots at -80˚C. We thawed fresh 15 µL aliquot for each analysis occasion.
Despite careful handling, the ATCC aliquots gradually lost activity over time, observed as a reduction in response range for the calibration curve. We didn’t see a difference in response level for the positive in-house control during the corresponding time period. Reduced activity and lower response level of the standard resulted in overestimation of virus titer for the samples. We compared the aliquoted lot of ATCC standard with a freshly thawed lot of standard in an assay run. The response level of the new lot came out twice as high as the old lot, confirming loss of activity for the old lot.
The stability issue was likely caused by improper handling with accidental freezing and thawing in connection to taking out standard from the freezer for analysis. More rigid handling with minimized exposure to room temperature significantly increased the longevity of the standard. For high performance analysis, it’s essential to properly handle the ATCC standard at all times.
LOD and LOQ for AAV2 titer assayWe calculated limit of detection (LOD) and limit of quantitation (LOQ) from the background signal from negative harvest samples. We calculated average response and standard deviation from ten replicate measurements of a negative sample in three consecutive runs. Then we calculated LOD (RU) and LOQ (RU) as Average+3*Stdev and Average+10*Stdev respectively. By reading off the standard curve, we estimated LOD and LOQ to be approximately 2 × 109 VP/mL and 5 × 109 VP/mL
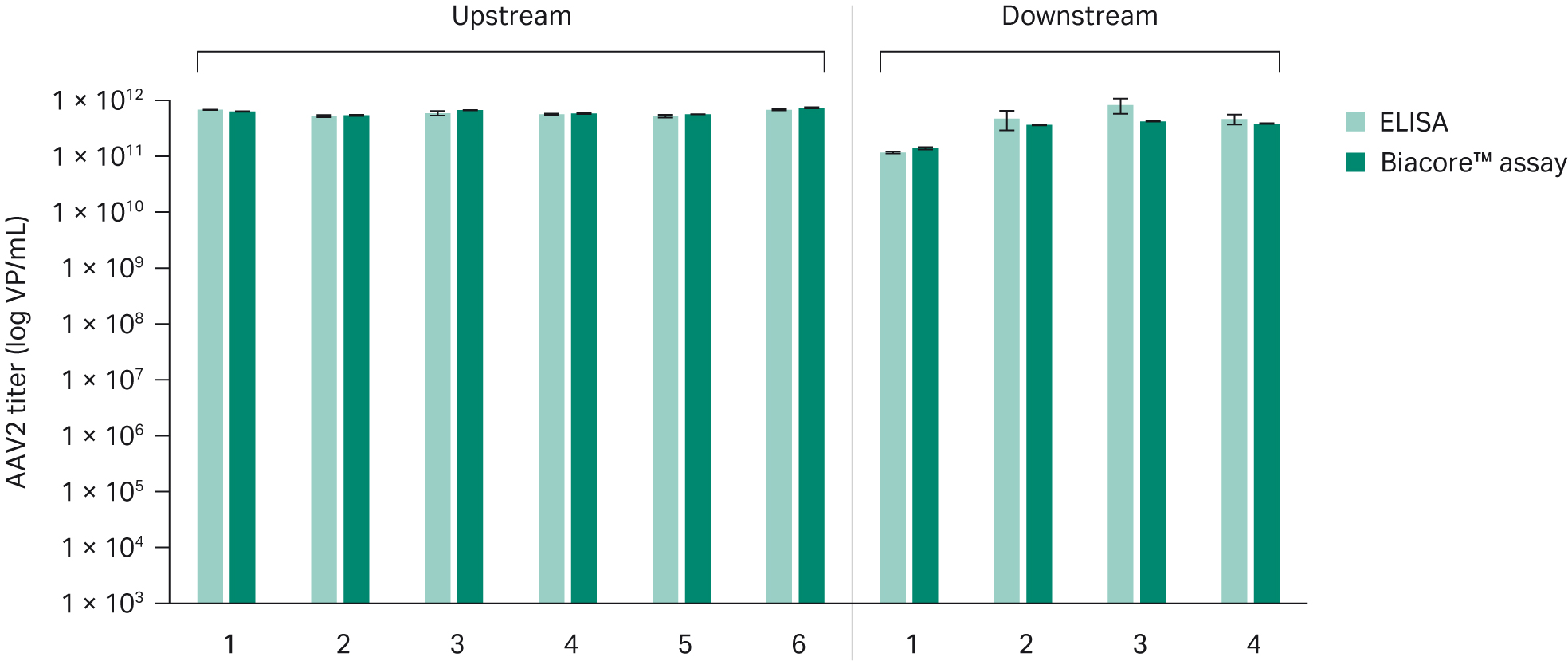
Comparison with ELISAWe compared the Biacore™ AAV2 titer assay with ELISA (Progen AAV2 Titration ELISA, art no. PRATV). We performed analysis on both upstream harvest samples and downstream process samples. The results showed good correlation between Biacore™ assay and ELISA. The intra-assay precision of the Biacore™ assay is 3%, significantly lower than the ≥ 20 % precision of the ELISA.
Fig 6. Comparison between ELISA and Biacore™ assay determination of AAV2 titer in upstream and downstream samples.
AAV2 titer analysis using Biacore™ 8K
We set up the assay on a Biacore™ 8K system which significantly increases sample throughput.
Biacore™ 8K is equipped with eight channels with two serial flow cells and eight injection needles that operate in parallel. This parallel configuration enables the use of parallel concentration analysis. In a parallel concentration experiment, the calibration curve is run across the channels with one calibration point per channel. This means scientists only need one cycle to run the calibration curve, unlike the serial setup where the calibration points are run in separate cycles. The parallel assay uses the same range for the calibration curve and the same contact times. A normalization cycle is added to the run to compensate for minor differences between channels. For practical reasons, the normalization solution can be one of the calibration points. With the parallel calibration curve and eight samples run in parallel, the system significantly reduces total run time. For a full 96-well plate of samples, the system reduces run time to less than 3 hours, compared to 19 hours on a single-needle system.
Fig 7. Biacore™ 8K lets you assay eight samples in parallel, significantly reducing run time.
AAV5 titer analysis
Assay stabilityThe AAV5 titer assay is set up and run as a capture assay, which means that capture of anti-AAV5 antibody is performed in every cycle. This way, the surface is considered new in every cycle, which prevents the use of a master calibration curve as for the AAV2 titer. However, the sensor chip used for capture of the anti-AAV5 antibody, Sensor Chip Protein A, is very stable and can be reused for multiple experiments without changing the surface’s performance. A calibration curve using the Vigene full standard is shown in Figure 8.
Fig 8. Titer analysis of AAV5 using Biacore™ T200. The figure shows an overlay of two calibration curves run with 16 samples between them. The range of the calibration curve is 9.1 × 109 to 5.8 × 1011 VP/mL.
Stability of standard
We used Vigene Full Standard as standard for the AAV5 assay. We prepared 25 µL aliquots of the standard and stored them at -80˚C. New aliquots were taken from the freezer for every new analysis. During the project, we did not experience any issues with stability and declining activity of this standard. The Vigene standard appeared less temperature-sensitive than the AAV2 ATCC standard.
LOD and LOQ for AAV5 titer assayWe calculated average response and standard deviation of a negative sample from ten replicate measurements in three consecutive runs. Then we calculated LOD (RU) and LOQ (RU) as Average+3*Stdev and Average+10*Stdev respectively. We estimated LOD and LOQ to be 9.8 × 109 VP/mL and 1.7 × 1010 VP/mL respectively.
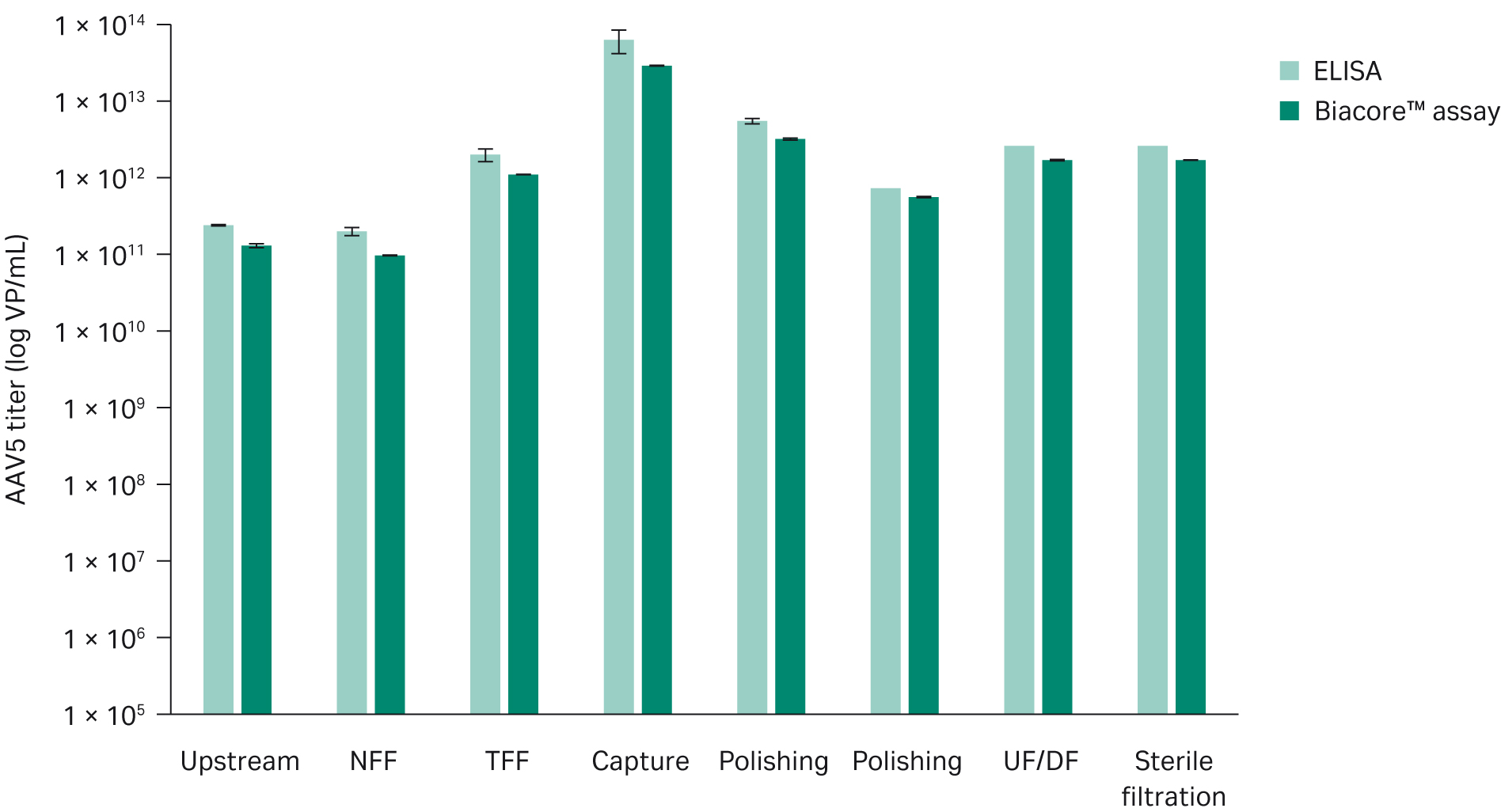
Comparison with ELISAWe performed a Biacore™ analysis of a set of upstream and downstream AAV5 process samples and compared the results with ELISA (Progen AAV5 Titration ELISA, art no. PRAAV5). Our results correlate well with a slightly lower titer for the Biacore™ assay (Figure 9). However, the difference in titer between the assays is small, and is likely due to differences in running conditions because the Biacore™ assay is optimized to reduce nonspecific binding and the ELISA is run with standard buffer conditions. This means there could be nonspecific binding in the ELISA.
For this sample set, the Biacore™ assay determined the average intra assay precision to be 2%, while the ELISA determined it to be 15%.
Fig 9. Comparison between ELISA and Biacore™ assay determination of virus titer in different types of upstream and downstream AAV5 samples. We obtained slightly higher titers with ELISA which might be due to nonspecific binding to the ELISA plates.
Conclusions
Biacore™ AAV titer assays generate robust and repeatable results that correlate well with established methods using ELISA. Compared to ELISA, Biacore™ titer assays have significantly lower assay variation. Biacore™ assays are more automated with minimal hands-on time, easy sample preparation, and automated data evaluation. Another advantage is that you can reuse Biacore™ sensor surfaces multiple times. For titer analysis of AAV2, you can use the same immobilized sensor chip for up to at least 10 days with a master calibration curve, so you can save standard and costs. The sensor surface we used in the AAV5 assay, Sensor Chip Protein A, is also very stable and can be reused for several analyses.
You can apply both assays on any Biacore™ SPR system that supports concentration analysis. Running the assay on Biacore™ 8K or Biacore™ 8K+ systems significantly increases sample throughput and reduces total run time. Biacore™ assays are excellent tools not only for quality control, but also for process optimization.
References
- Choi, JH., Yu, NK., Baek, GC. et al. Optimization of AAV expression cassettes to improve packaging capacity and transgene expression in neurons. Mol Brain 7, 17 (2014). https://doi.org/10.1186/1756-6606-7-17
- Application note: Cell culture process development for AAV vector production in suspension cells
- Application note: Adeno-associated virus production in suspension HEK293 cells with single-use bioreactors
- Application note: AAV5 capture and polishing optimization
- Application note: AAV5 purification including analytics
- Article: Develop and refine your AAV production process
Ordering information
| Product information |
Information |
Product code |
|---|---|---|
| Series S Sensor Chip CM5 Pack of 3 |
Pack of 3 |
BR100530 |
| Series S Sensor Chip Protein A |
Pack of 3 |
29127556 |
| Amine Coupling Kit |
Reagents for immobilization |
BR100050 |
| HBS-EP+ Buffer 10x |
Analysis buffer |
BR100669 |
| Glycine 1.5 | Regeneration solution |
BR100354 |
| Acetate 5.0 |
Coupling buffer |
BR100351 |
| Biacore™ T200 |
SPR system for characterization of molecular interactions |
28975001 |
| Biacore™ 8K |
SPR system for characterization of molecular interactions |
29337763 |
| Biacore™ 8K+ |
SPR system for characterization of molecular interactions |
29283382 |