Introduction
Manufacturing of biotherapeutics requires large volumes of process liquids. A 2000 L production batch of a monoclonal antibody (mAb) at a titer of about 5 g/L is estimated to use 16 000 to 22 000 L of water for injection (WFI). Such volumes, coupled with the large number of different types of buffers and process liquids that are used in bioproduction, can exert pressure on in-house buffer preparation operations and lead to capacity constraints. As many as one in five biomanufacturers report seeing constraints on their purification steps due to buffer preparation, storage, and transfer and are looking for ways to add more capacity to their buffer preparations without major capital investments (1).
Outsourcing is one attractive option to simplify buffer management, and it has been reported that interest in outsourcing of buffer preparation has doubled in the past 5 years, from around 6% to 13% (1). This increase in interest is, likely driven by the need for a wider range of buffers to meet the growing diversity in the drug pipeline. Outsourcing can be a good solution especially for biomanufacturers operating at smaller scales. In addition to enabling a smaller facility footprint, the ability of a supplier to handle quality assurance (QA) and quality control (QC) can further lessen the burden on the manufacturer.
Another way to overcome buffer challenges is the use of technologies such as inline dilution (ILD) where buffers are prepared in line from buffer concentrates, or inline conditioning (IC), where they’re prepared from highly concentrated, low-volume, single-component stock solutions. This way, buffer preparation can be directly integrated with the chromatography or filtration step, eliminating the need for intermediate storage in buffer bags or holding tanks. In addition to reducing facility footprint, automating the buffer preparation process can also ensure the quality of the final formulation. About 20% of biomanufacturers and about 30% of CDMOs report they are looking to buy inline dilution or inline conditioning skids as they look to streamline processes, increase productivity, and utilize facilities more efficiently (1). However, more western European companies are considering adding inline buffer dilution systems (35%) than U.S.-based companies (13.7%) indicating there are some regional drivers at play (1).
Buffer preparation drives cost
Buffer preparation is a highly manual and resource intensive activity, and the large holding tanks require significant floor space that could be used for other core operations. By efficient use of existing stainless-steel facilities, cost of in-house buffer preparation and other process liquids is relatively low compared with preparation using single-use equipment or outsourcing. Challenges, however, arise when buffer capacity has reached its maximum and needs to be expanded or when the capacity is not fully utilized. In addition, biomanufacturers must consider how current capacity aligns with their product pipeline for drug candidates that may or may not achieve commercial approval. Hence, it can be beneficial to postpone investments in in-house facilities and instead outsource buffer production until outcome of clinical trials have been evaluated (Fig 1).
Fig 1. Outsourcing or in-house preparation of buffers and other process liquids can be more or less beneficial at different drug development stages. At larger scales, inline buffer preparation can be a viable alternative to meet challenges in buffer production.
Add buffer capacity with outsourced buffers
A quick and simple way to gain extra or new capacity is to outsource buffer production to an expert supplier with a proven track record of delivering large volumes of bulk fluids or preblended powder. While using preblended powder might save time and cost for raw material validation and characterization, it does not reduce the need for in-house hydration. As a result, outsourcing the preparation of sterile liquids or buffer concentrates is more common.
Advantages of outsourcing buffer preparation
Reduce capital investment
- Access initial capacity at a fraction of the cost.
- Reach market faster and at lower risk.
Reduce cost
- Improve efficiency and reduce QC testing with larger batch volumes.
- Reduce cycle times with just-in-time hydrated solutions.
- Improve use of qualified resources.
- Relieve capacity constraints.
- Gain immediate capacity and inventory relief.
Secure quality and supply
- Qualify additional supplier.
- Reduce audit burden.
- Reduce raw material risks and batch-to-batch inconsistencies.
While outsourcing buffer production requires some initial efforts for activities such as qualifying the supplier, setting specifications for raw materials and containers, and validating the materials used in single-use bags, the advantages gained by outsourcing include leveraging the supplier’s manufacturing capabilities, supply chain, logistics expertise, and quality assurance (QA) and quality control (QC) teams. Once the initial work on vendor validation has been done, the biomanufacturer continues to benefit from a robust supply chain through reduction in buffer preparation and quality control testing time as well as QA documentation, which can result in a reduced audit burden.
Intensify processes with inline buffer preparation
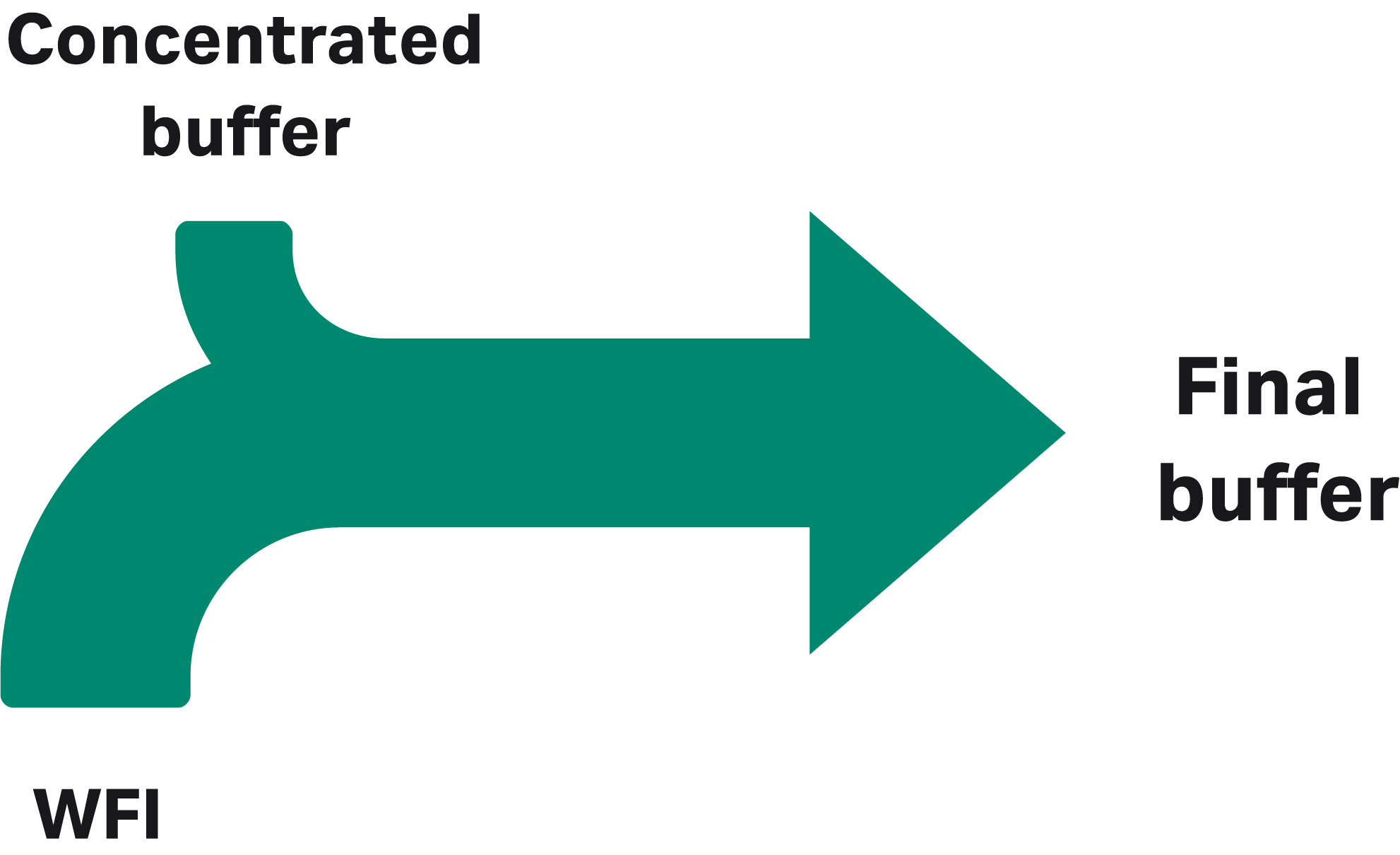
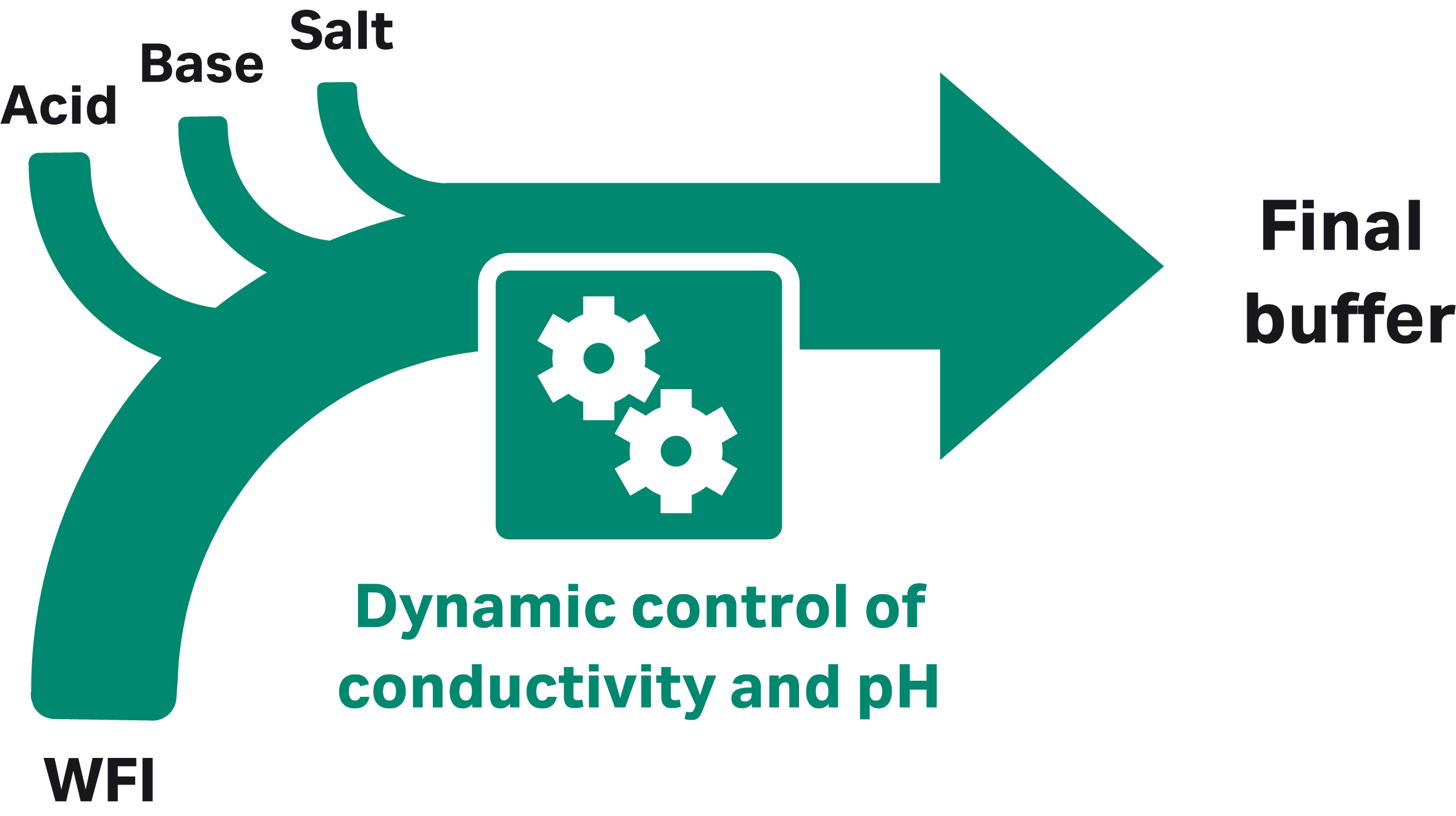
Manufacturers who want to keep buffer production in house while reducing the amount of space and labor devoted to buffer preparation and handling can consider implementing ILD or IC (Fig 2). With ILD, volumes are reduced by using buffer concentrates that are diluted in line with water for injection (WFI) using a system such as the ÄKTA process™ chromatography system with an additional pump to allow buffer dilution along with gradient formation (2, Fig 3). One concentrate is required for each individual buffer used in a specific process. Using IC, buffers are prepared in line from concentrated, single-component stock solutions of acid, base, salt, and WFI. With both ILD and IC, significant reductions in floor space and tank volumes can be achieved.
Concentrated, well-defined buffer as input + Significant reductions in floor space and tank volumes.
+ Increased automation.
- pH shifts due to dilution have to be handled.
- Sensitive to least soluble component.
- Affected by common ion effect (CIE).
Stock solutions of buffer components as input + Significant reductions in floor space and tank volumes.
+ Reduced operator handling, and increased automation.
+ Can condition many buffers from same stock solutions (steps, gradients, etc.).
+ Dynamic control: use combinations of various types of feedback control.
+ The least soluble component affects only itself and not affected by CIE.
Fig 2. Two ways of addressing buffer challenges: (A) inline dilution and (B) inline conditioning.
Fig 3. ÄKTA process™ chromatography system with a third pump supports inline dilution.
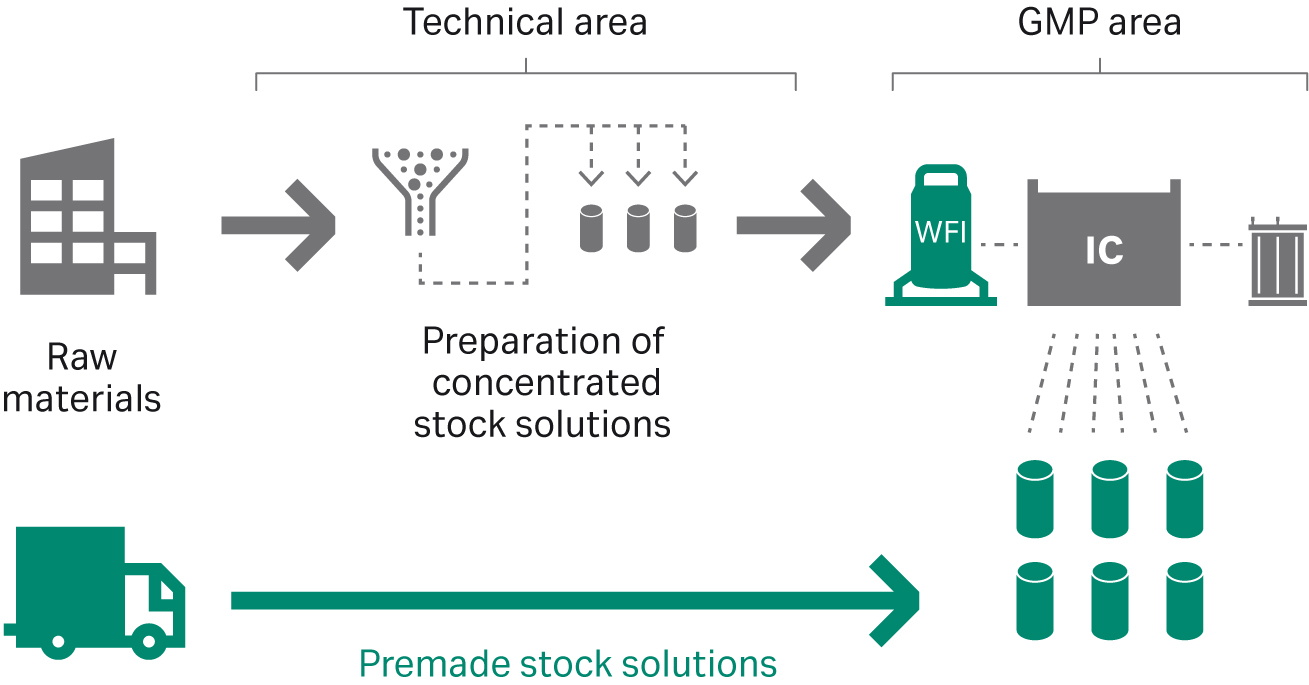
However, a significant space reduction comes with IC. Single-component stock solutions can be much more concentrated than a multi-component buffer concentrate, of which the least soluble ion will limit its maximum concentration. The use of single-component stock solutions as inputs provides additional benefits in that they are more straightforward to prepare than buffers or buffer concentrates, as they require dissolving of one single component only and no need for titration and pH adjustment. As an alternative to manually preparing buffer concentrates or single-component stock solutions in-house, production of the input solutions can also be outsourced (Fig 4).
Fig 4. Outsourcing preparation of stock solutions enables great time savings and enhances resource utilization.
For standard 1× buffers, there is a practical batch size limitation, as some of the largest available single-use bags are around 3000 L, a volume generally not sufficient for biomanufacturing applications. From this perspective, technologies such as ILD and IC, not only decrease the facility footprint, but also facilitate the transition to single-use solutions due to the reduction in input volumes from the use of concentrated buffers or stock solutions. Implementing technologies such as ILD and IC help streamline the entire buffer preparation process and reduce manual handling by automating several steps, not only reducing the risk of human errors, but also making it possible to reassign personnel to other tasks that provide more value.
Integrating buffer preparation with unit operations
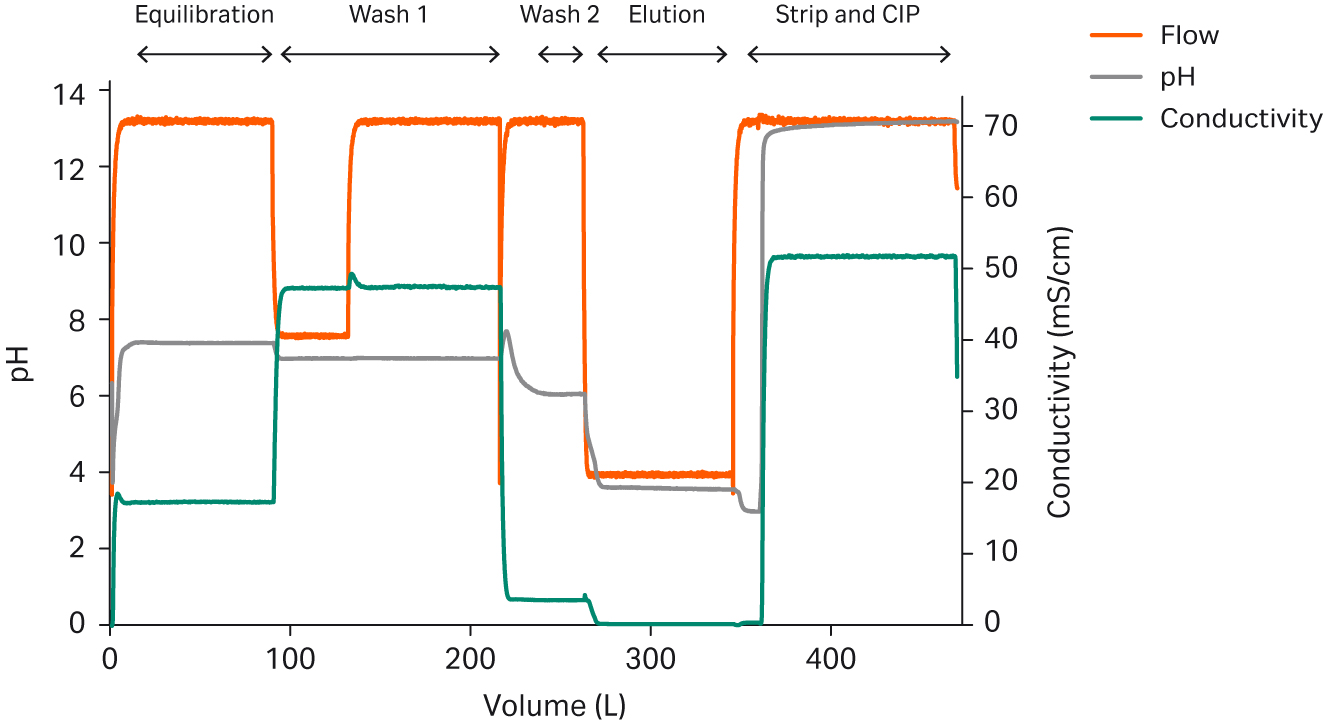
IC systems and more advanced ILD systems have the functionality required to operate as a chromatography unit, allowing direct connection to a chromatography column to make it possible to deliver the buffers directly to the process without the need for storage in bags or tank (3). Furthermore, with IC, the system will prepare the exact amounts of the relevant buffers in line from the stock solutions. Waste will only be generated during the switch between buffers until the set pH is reached and stable (Fig 5).
Fig 5. IC used in preparation of buffers as well as strip and clean-in-place (CIP) solutions required for a mAb capture step (4). The arrows indicate preparation of formulations within specifications.
Fig 6. Highly concentrated, low-volume stock solutions required for a three-step mAb purification process from 2000 L culture feed with a mAb titer of 3 g/L. Different combination of stock solutions and WFI will generate the different buffers in an automated manner. A total volume of 1428 L of the eight stock solutions is required to prepare 11 different buffers for a total volume of 6664 L.
Moving inline conditioning into the technical area
As inline conditioning uses single-component stock solutions as inputs, this technology can also be transferred into the technical area and serve as a stand-alone buffer production unit. The system has dedicated pump lines for acids, bases, and salts, with several inlet ports to enable formulation of different buffer families in one production run, providing a high degree of flexibility (Fig 6).
Ensuring quality rigor
Inline conditioning control functions
Because buffers are used to maintain purification conditions as well as stabilize the bioproduct and preserve its functional characteristics, correct buffer formulation is crucial for success in bioproduction. Usually, a buffer is defined by a set of parameters that typically are critical process parameters (CPPs) of which pH and conductivity are the most obvious.
However, there might be additional parameters that are important for the process, such as temperature, buffer concentration, and concentration of additives. If the buffer is a mixture of components, the concentration of each component can be critical. If a post-adjustment approach is used, having full control of these additional parameters can be challenging.
With IC, it is possible to select the feedback mode that best controls critical process parameters (Table 1). Feedback control will also ensure that the mass balance is kept. There are three modes of feedback control that can be used with the dynamic control functionality of the system: recipe and flow; pH and flow; and pH and conductivity.
Recipe and flow feedback
A known buffer formulation is entered in the system control software. The software adjusts the flow rates of the specified stock solutions to achieve desired formulation. This control mode is useful when the temperature is constant, and the stock solutions are accurate.
pH and flow feedback
The user enters target pH, and the software adjusts the flow rates of the acid and base stock solutions to achieve desired pH in the final formulation.
pH and conductivity feedback
The user enters the target pH and conductivity, and the dynamic control functionality of the software uses feedback from flow, conductivity, and pH sensors to adjust flow rates of the stock solutions to achieve desired conductivity and pH. In this control mode, both the temperature and the concentrations of the stock solution can vary without affecting accuracy of the final buffer formulation.
Table 1. Control modes used with IC
| Control mode | Benefits | Drawbacks |
| Recipe and flow | Robust if temperature is constant. Can use recipe generated from pH and flow feedback. |
Can lead to variations in pH and conductivity if temperature varies. Accurate stock solutions are needed. |
| pH and flow | Correct pH even if temperature varies. Generates recipe to be used in flow feedback. |
Sensitive to bias in pH meter. Require accurate stock solutions. |
| pH and conductivity | Correct pH even if temperature varies. Correct conductivity even if temperature varies. Does not need accurate stock solutions. |
Sensitive to bias in pH meter. Sensitive to bias in conductivity meter. |
Considerations in supplier selection
Logistics is an important aspect when purchasing process fluids for use in biomanufacturing, and care should be taken when selecting a supplier. Preferably, the supplier has local manufacturing facilities or warehouses to reduce cost and time for shipping.
The shipping container is also of importance. The primary container needs to maintain its integrity during shipment and storage. If single-use containers are used, the plastic film should be flexible, yet strong, durable, and resistant to puncture and abrasion. Any seals must be strong enough to avoid any breaks or leakages. In addition, the film must also help maintain important quality attributes of the buffer during storage. Therefore, the film should be biocompatible and have a low profile of extractables and leachables to avoid any harmful substances from the plastic contaminating the liquid. The film also needs to possess excellent moisture and gas-barrier properties to enable maintenance of pH and osmolality during long-term storage. Thus, qualifying the film is as important as qualifying the buffer. Different vendors use different films with different properties. If plastic bags are used in other applications, such as in single-use bioreactors or for hydration and storage of liquid cell culture media, it can be time-saving to use the same film across all applications, thereby avoiding the qualification of several films for the different applications.
Regulatory requirements and security of supply
Regulatory pressure and supply chain management add costs and resources to in-house buffer preparation. Concerns include raw material characterization and qualification, documentation, validation, quality control, and security of supply. Any raw materials and their suppliers need to be prequalified and validated, and the final formulation must be quality control tested. If buffer preparation is conducted at a separate location from the actual manufacturing site, there will be one batch record for buffer preparation and testing, and yet another for the process step. The documentation can therefore be scattered, lowering traceability.
Environmental health and safety burdens as well as bioburden control can also contribute to increased in-house manufacturing costs. Some buffer components required for downstream purification, such as caustics, are hazardous and need to be handled with care. Buffer preparation in open tanks increases risk of contamination and raises safety concerns. Obtaining ready-made buffers in closed single-use containers reduces such risks.
Outsourcing buffers can alleviate many of these concerns, and the technical expertise of the supplier should not be undervalued. Letting an expert supplier deal with buffer preparation as well as quality assurance, quality control, and their associated documentation can be a cost-efficient alternative for many biomanufacturers. Responsiveness of the supplier is also of utmost importance, as supply assurance requires good communication between the supplier and the customer.
However, outsourcing versus in-house buffer preparation is decision based on the situation. In fact, most biomanufacturers that purchase finished fluids also have internal buffer capability, therefore, utilize different strategies for outsourcing. Some biomanufacturers prefer to outsource complex buffers or buffers with hazardous components and prepare simple buffers internally. Others may prefer to be in control of the complex buffers and instead purchase large volumes of more simple fluids such as sodium chloride, sodium hydroxide, or ethanol. Still other manufacturers prefer having the main buffer preparation in house, while validating a vendor as a second supplier to reduce risk and gain extra capacity for buffer preparation when needed due to internal constraints.
Case studies
Accuracy in buffer formulations prepared from ready-made stock solutions using IC
This study describes a lean approach to buffer preparation by implementing IC. Buffers of different formulations for a mAb chromatography capture step were prepared in an automated, consecutive manner using our IC system. To further reduce the time and space required for buffer preparation, HyClone™ ready-made, highly concentrated, low-volume, single-component stock solutions were used.
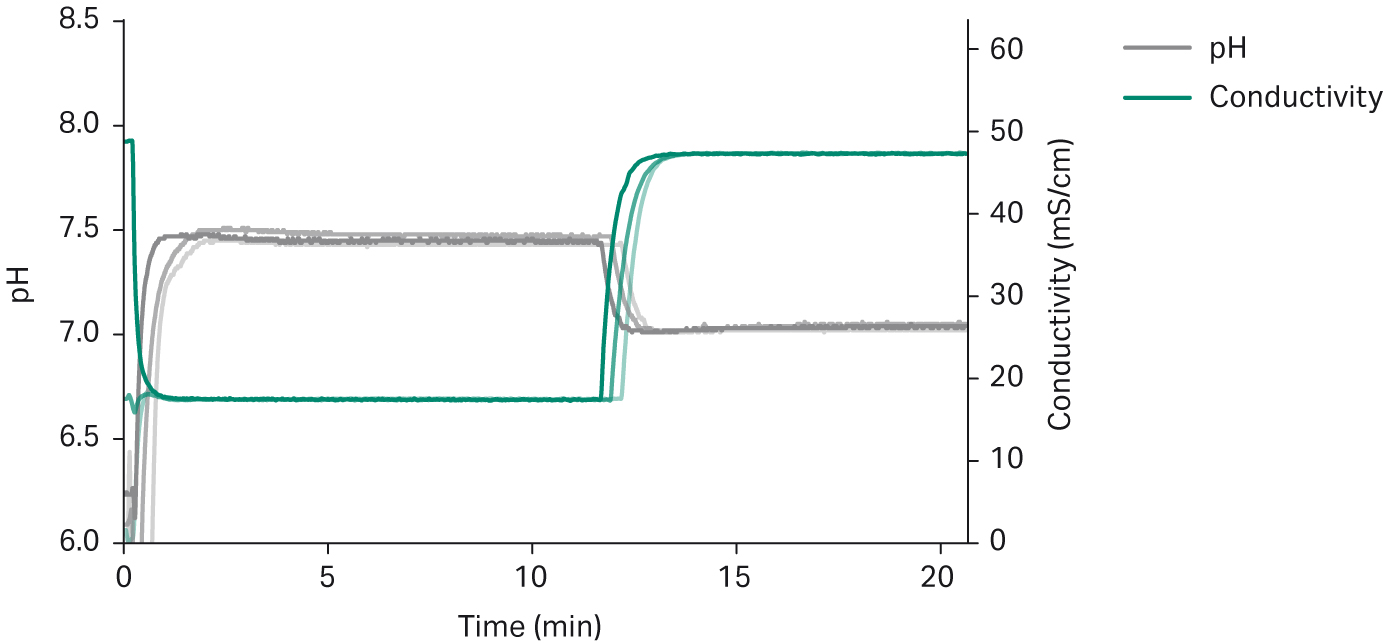
The dynamic control functionality of the UNICORN™ system control software was used for feedback regulation of the final buffers to ensure accurate formulations. Typically, in bioprocessing applications, buffer accuracy needs to be within ± 0.15 pH units. Conductivity specifications are typically within ± 2%. These requirements put high demands on the robustness of the system performance between batches and runs. In Figure 7, an overlay of three preparations of two buffers with an intermediate switch between buffers demonstrates consistency between the preparations.
More detailed information can be found in Automated in-line buffer preparation from readymade stock solutions in a mAb process step (4).
| Buffer | pH | Conductivity (mS/cm) |
| 20 mM sodium phosphate, 150 mM NaCI, pH 7 buffer | 7.41 ± 0.05 7.44 ± 0.05 7.40 ± 0.04 |
17.3 ± 0.3 17.3 ± 0.2 17.4 ± 0.2 |
| 20 mM sodium phosphate, 500 mM NaCI, pH 7 buffer | 7.00 ± 0.03 7.01 ± 0.04 6.99 ± 0.02 |
47.2 ± 0.2 47.3 ± 0.3 47.3 ± 0.3 |
Fig 7. Overlay of triplicate preparations of 20 mM sodium phosphate, 150 mM NaCl, pH 7.4 buffer, followed by preparation of 20 mM sodium phosphate 500 mM NaCl, pH 7 buffer, showing reproducibility of buffer formulation. The time to switch from one formulation to another is similar between preparations and takes about 2 min.
Process economy using IC versus traditional manual buffer preparation
In a study we ran on the performance of the IC system, a process economy simulation was conducted, comparing traditional manual buffer preparation with buffer preparation using IC. The objective was to compare volumes needed to formulate buffers and the footprint required, as well as cumulative running costs for different approaches.
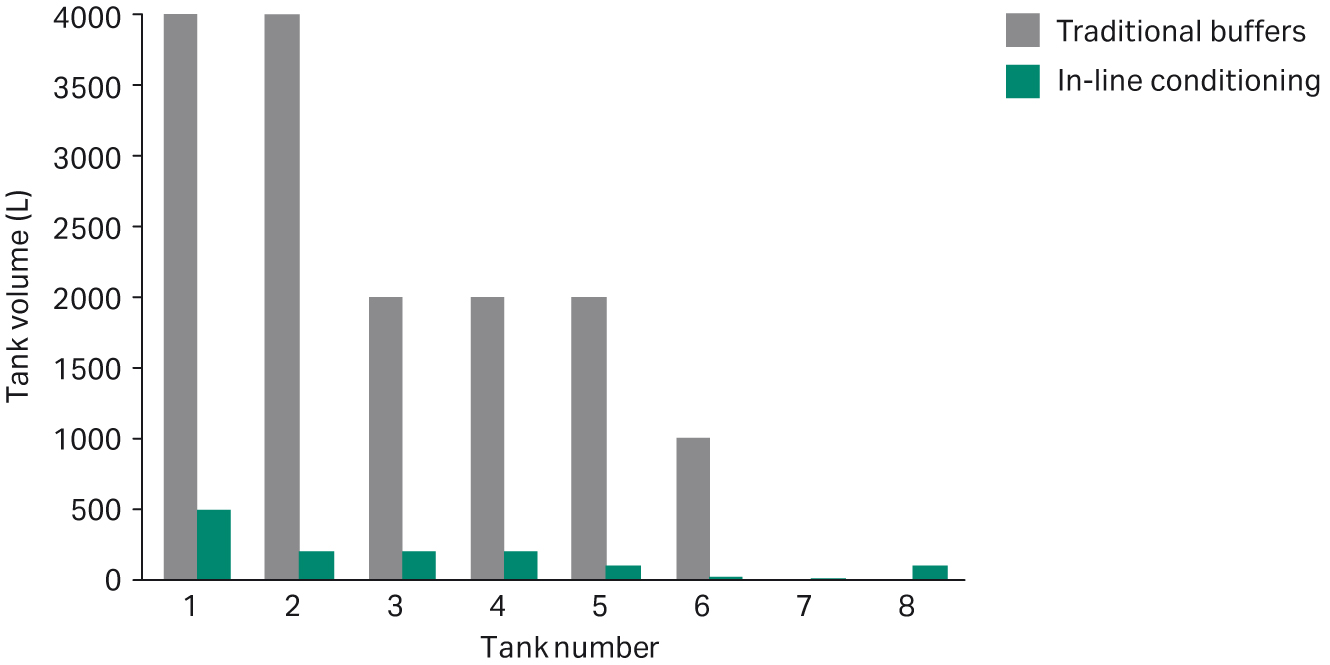
Compared with the traditional way of preparing buffers, the results show that the volume of the buffer holding tanks can be reduced by up to 90% by using IC (Fig 8). In addition, the total footprint of tanks and system can be reduced by 40% (Fig 9).
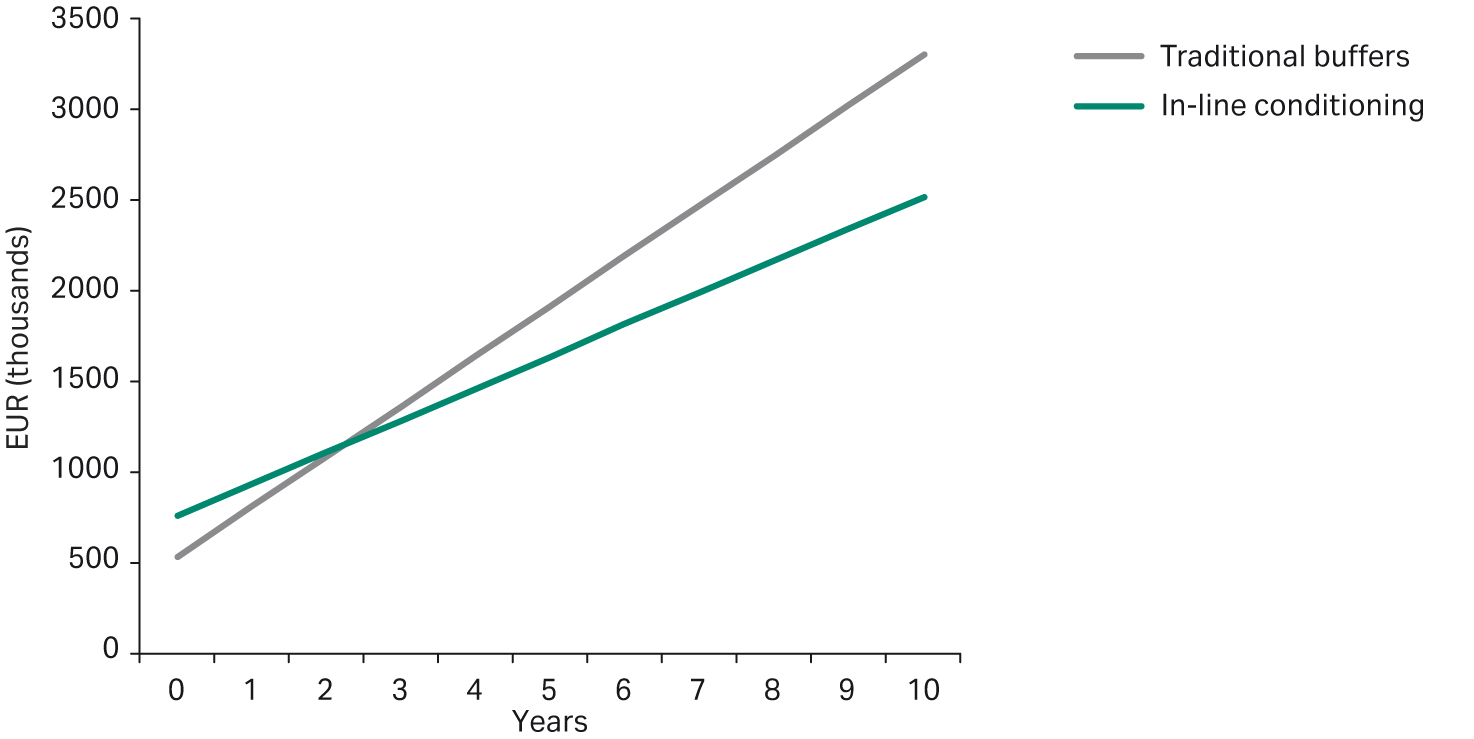
Although the initial investment, including tanks and system, is larger for IC, the savings in operating costs are significant. As shown in Figure 10, the higher investment cost for IC can be recouped after only a few years of operation. Additionally, costs associated with waste of buffers that have become obsolete, for example, during delays in production, can be prevented using IC, as single-component stock solutions are highly concentrated solutions of acid, base, or salt, and hence, can exhibit longer shelf life than the final buffer formulation. Automated buffer preparation not only reduces human error, but also allows for a more consistent buffer preparation, and by producing buffers just-in-time, the risk of buffers becoming obsolete can be avoided.
More detailed information can be found in Automated in-line buffer preparation from readymade stock solutions in a mAb process step (5).
Fig 8. Comparison of tank volumes between traditionally prepared buffers and buffers prepared using IC.
Fig 9. Comparison of footprint of system and tanks between traditionally prepared buffers and buffers prepared by IC.
Fig 10. Comparing accumulative running cost between traditionally prepared buffers and buffers prepared by IC.
Conclusion
When evaluating ways to save costs in buffer management, it is important to not only study the amount of time and cost spent on buffer preparation, but also to take a wider look at the productivity of the facility’s entire footprint. Space needed for in-house buffer preparation, inventory, and storage is disproportionally large, but does not add directly to the overall productivity. In addition, any bottleneck experienced in buffer preparation can reduce production capacity utilization.
By reducing footprint and relieving resources dedicated to buffers preparation, biomanufacturers can improve overall productivity. At a first glance, the purchase of ready-made buffers and other process liquids can appear more costly than in-house prepared equivalents. However, a mix of outsourced and in-house buffer preparation can often provide a better solution. In larger scale, technologies such as IC and ILD are efficient options for buffer preparation, and the use of ready-made, low-volume stock solutions can make the process even more efficient. By allowing for integrated, just-in-time buffer preparation, IC and ILD eliminate hold time as well as the need for intermediate holding tanks between process steps.
References
- BioPlan Associates. 21st Annual report and survey of biopharmaceutical manufacturing capacity and production. BioPlan Associates, Inc; 2024. https://www.bioplanassociates.com/21st/
- Application note: Efficient chromatography buffer prep with inline dilution
- Application note: Connected inline conditioning for better process efficiency
- Application note: Automated in-line buffer preparation from readymade stock solutions in a mAb process step. Cytiva, CY13953 (2017).
- Application note: Overcoming buffer challenges with in-line conditioning. Cytiva, CY13812 (2016).
- Download the related white paper
CY49362-06Feb25-AR