Affinity resins - tagged proteins
Purify fusion proteins with his-tag, Cytiva Protein Select™, GST, and MBP affinity resins















Frequently asked questions
Protein tags
Protein tagging refers to the process of genetically grafting peptide sequences onto proteins such as antibodies. The tagged recombinant proteins can then be captured for analysis or for preparative purposes. These tags are often removable using chemical agents or enzymes like protease. You can add protein tags to either end of the protein of interest. Some tags are C-terminus or N-terminus specific, while others are both C-terminus and N-terminus specific. Appended to proteins, affinity tags allow protein purification. Common tags include maltose binding protein (MBP), glutathione-S-transferase (GST) and polyhistidine (his-tag). You can purify proteins through his-tag affinity chromatography or GST-tag affinity chromatography.
Gst tags and his-tags
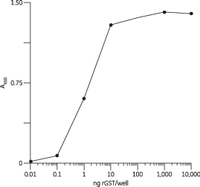
GST tags have high affinity for immobilized glutathione. Glutathione affinity is an effective method for single-step protein purification or as the first in a multistep purification procedure, depending on your requirements for the final protein purity. Cytiva offers a number of products for production and purification of GST-tagged proteins.
A his-tag is an amino acid motif that comprises six or more histidines added to the N- or C-terminus of a target protein. His-tagged proteins can be purified based on the his-tag’s affinity for certain metals or using an antibody ligand. The string of histidine residues binds to immobilized metal ions such as copper and nickel under appropriate buffer conditions. Use his-tag antibodies in assay methods.
Tagged protein purification
Protein tags can help simplify protein purification because protocols for isolation are standardized. Recombinant proteins of interest can be captured by a resin or matrix coupled to a binding ligand with high affinity to the tag. In addition to our bulk resins, we also offer prepacked columns for affinity capture of tagged proteins.
Immobilized metal affinity chromatography (IMAC)
Immobilized metal affinity chromatography is a common process of purifying his-tagged proteins, as they have binding affinity to certain metal ions under certain conditions. In this technique, metal ions are immobilized on a resin matrix through their interactions with chelating agents in the matrix. Resins charged with nickel (Ni) or cobalt (Co) ions are commonly used in his-tag affinity chromatography.
His-tag affinity chromatography FAQs
Here are answers to some frequently asked questions about his-tag affinity chromatography.
What does a his-tag do?
His-tags are added to proteins for detection or purification. His-tag affinity chromatography allows researchers and biomanufacturers to selectively extract target proteins from complex mixtures.
How is a his-tag removed?
Removal of affinity tags entails enzymatic cleavage of the tag.
Does a his-tag affect protein activity?
His-tags have low molecular weight and often have no effect on protein structure and function. However, for some recombinant proteins, his-tags can affect protein stability.
What type of protein tag should I use?
Our affinity tag selection guide can help you make sure you choose the best tag for your application.
What type of resin should I use to purify my recombinant protein?
Use the Purify app from Cytiva to find out which type of affinity column or resin would best suit your tagged protein purification.
